Given The Balanced Equation Representing A Reaction
Hey there, awesome science adventurers! Ever looked at a chemical reaction and thought, "Whoa, what's really going on there?" You know, those fancy scribbles with arrows and symbols that look like a secret code? Well, guess what? They're not that scary, especially when we talk about the super-important concept of a balanced equation. Think of it as the ultimate recipe for how atoms and molecules throw a party together and make new stuff!
So, you've got this chemical equation, right? It’s basically telling you what ingredients you’re starting with (we call those the reactants) and what delicious new dishes you end up with (the products). It’s like saying, "If I put flour, eggs, and sugar together, I get a cake!" (Though, chemistry is a little more complex than baking, but you get the drift). The arrow in the middle? That's the event horizon, the point of no return, where the magic happens!
Now, the "balanced" part is where things get really interesting. Imagine you're trying to make those miniature Lego castles. You need exactly two little grey bricks and one blue brick for each tiny castle tower. If you only have one grey brick, you can’t make a full tower, can you? Same deal with chemistry, but instead of Lego bricks, we're talking about atoms. Atoms are the universe's building blocks, and they're super-duper important.
The big rule, the one that makes the universe tick, is the Law of Conservation of Mass. Basically, it says that you can't just poof atoms into existence, and you can't make them vanish into thin air. They're just rearranging themselves, like a cosmic game of musical chairs. So, if you start with, say, four oxygen atoms on one side of your equation, you have to end up with four oxygen atoms on the other side. No exceptions, no cheating!
This is where our trusty balanced chemical equation comes in. It's like the universe's meticulous accountant, making sure every atom is accounted for. When an equation is balanced, it means that the number of atoms of each element is the same on both the reactant side and the product side. Ta-da! Mystery solved, or at least, the accounting is all there.
Let’s break it down with a super simple example. Imagine hydrogen gas (H₂) reacting with oxygen gas (O₂) to form water (H₂O). If we just wrote H₂ + O₂ → H₂O, it would look something like this:
H₂ + O₂ → H₂O
Let's count our atoms, shall we? On the left side (reactants):
- Hydrogen (H): 2 atoms
- Oxygen (O): 2 atoms
And on the right side (products):
- Hydrogen (H): 2 atoms
- Oxygen (O): 1 atom
Uh oh. See that? We've got the same amount of hydrogen, which is cool, but we've got two oxygens on the left and only one on the right. Where did the other oxygen atom go for a coffee break? It didn't! It's just that our initial equation wasn't balanced. It’s like trying to make two Lego towers with only one blue brick – you’re just not going to get there!
So, how do we fix this cosmic accounting error? We use coefficients. Think of coefficients as little numbers that we put in front of the chemical formulas. These numbers tell us how many molecules (or atoms) of that substance are involved. They’re like the multipliers in our Lego recipe. You never change the little numbers within the chemical formula itself (those are called subscripts) because that would change the actual substance. Changing H₂O to H₂O₂ would mean you've made hydrogen peroxide, which is a totally different beast (and can be used for cleaning or… well, you know).

We need to adjust the coefficients until our atom counts match up. Let's go back to our H₂ + O₂ → H₂O problem. We need more oxygen on the product side. If we put a '2' in front of H₂O, like this:
H₂ + O₂ → 2H₂O
Now, let's recount. On the left:
- Hydrogen (H): 2 atoms
- Oxygen (O): 2 atoms
On the right:
- Hydrogen (H): 2 * 2 = 4 atoms
- Oxygen (O): 2 * 1 = 2 atoms
Still not quite right! We've got enough oxygen now (yay!), but our hydrogen count is all out of whack. We started with 2 hydrogens and now we have 4. It’s like trying to balance a seesaw when one side is way heavier – you gotta adjust!
Let's try again. We need 4 hydrogens on the right, so let's put a '2' in front of the H₂ on the left. This gives us:
2H₂ + O₂ → 2H₂O
Now, let's do the grand total, the final audit, the moment of truth! On the left (reactants):

- Hydrogen (H): 2 * 2 = 4 atoms
- Oxygen (O): 2 atoms
And on the right (products):
- Hydrogen (H): 2 * 2 = 4 atoms
- Oxygen (O): 2 * 1 = 2 atoms
BINGO! We have 4 hydrogens on both sides and 2 oxygens on both sides. The equation is balanced! This means that for every two molecules of hydrogen gas reacting with one molecule of oxygen gas, you get exactly two molecules of water. It’s like the perfect, harmonious dance of atoms, all accounted for and happily transformed!
Why is this whole balancing act so important, you ask? Well, imagine you're a chemist trying to make a specific amount of a new medicine. If your equation isn't balanced, you won't know the correct ratio of ingredients you need. You might end up with too much of one reactant and not enough of another, leading to wasted materials or even an impure product. It’s like trying to bake a cake and accidentally using a whole bag of salt instead of a teaspoon – definitely not the yummy result you were hoping for!
Balancing equations also helps us understand the stoichiometry of a reaction. Fancy word, right? Stoichiometry is basically the study of the quantitative relationships between reactants and products in a chemical reaction. In simpler terms, it tells you how much stuff reacts and how much stuff is produced. If your equation is balanced, you can use those coefficients as a roadmap to calculate exactly how many grams or moles of a substance you need or will get.
Let's think about it this way: If you have a recipe that calls for 2 cups of flour and 1 egg to make 12 cookies, and you want to make 24 cookies, you know you need to double everything – 4 cups of flour and 2 eggs. The balanced equation is your recipe! The coefficients are your measurements. They tell you the precise proportions needed for a successful chemical "recipe."
Sometimes, balancing equations can feel like a little detective game. You’re looking for clues (the atoms!) and trying to make everything add up. It’s a logic puzzle where the universe provides the pieces. Don’t get discouraged if it takes a few tries. We’ve all been there, staring at an unbalanced equation, muttering to ourselves, "Where did that carbon atom run off to?"
There are some handy tips and tricks to make the process smoother:
Tips for Balancing Like a Boss:
- Start with the elements that appear in only one reactant and one product. These are usually the easiest to balance first.
- Leave elements that appear in multiple places (like oxygen or hydrogen in some cases) for later. They can be trickier to manage initially.
- Balance polyatomic ions (like sulfate (SO₄²⁻) or nitrate (NO₃⁻)) as a single unit if they appear unchanged on both sides of the equation. This can save you a lot of hassle counting individual atoms.
- If you end up with fractions, multiply the entire equation by the denominator of the fraction. This will clear the fractions and give you whole numbers.
- Always, always, always double-check your final count! One misplaced atom can throw off the whole cosmic balance.
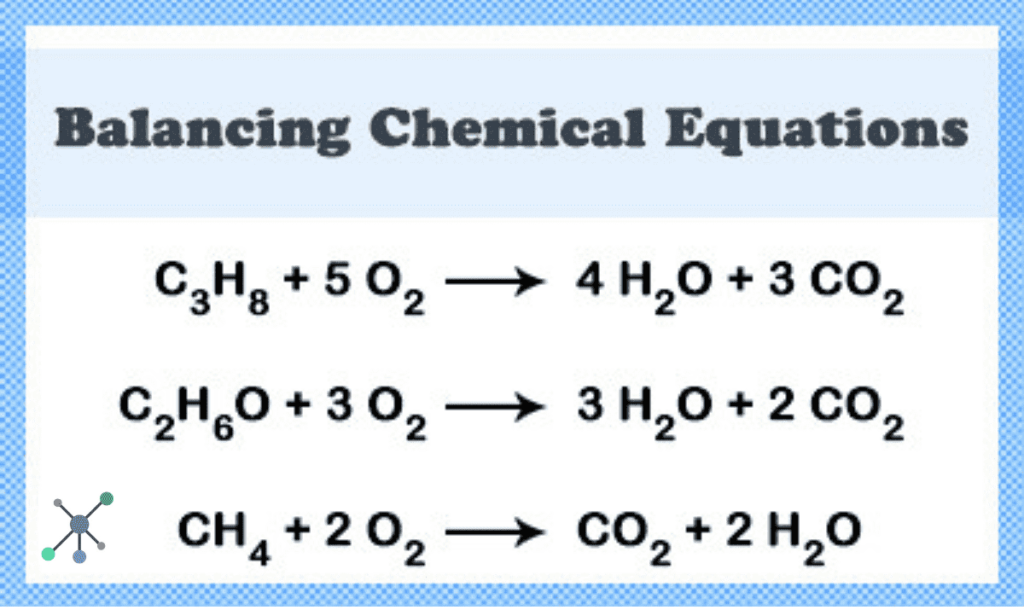
Let’s try another one, just for kicks. How about the reaction of methane (CH₄) with oxygen (O₂) to produce carbon dioxide (CO₂) and water (H₂O)? This is like the combustion of natural gas. The unbalanced equation looks like this:

CH₄ + O₂ → CO₂ + H₂O
Let’s count:
Reactants:
- C: 1
- H: 4
- O: 2
Products:
- C: 1
- H: 2
- O: 2 + 1 = 3
Okay, carbon is balanced (score!). Hydrogen needs help – 4 on the left, 2 on the right. Oxygen is a bit of a mess – 2 on the left, 3 on the right. Let's tackle hydrogen first. To get 4 hydrogens on the right, we need a '2' in front of H₂O:
CH₄ + O₂ → CO₂ + 2H₂O
Now, let’s re-count:
Reactants:

- C: 1
- H: 4
- O: 2
Products:
- C: 1
- H: 2 * 2 = 4
- O: 2 + (2 * 1) = 4
Carbon and hydrogen are looking good! Now for oxygen. We have 2 on the left and 4 on the right. To get 4 oxygens on the left, we need a '2' in front of O₂:
CH₄ + 2O₂ → CO₂ + 2H₂O
Final check!
Reactants:
- C: 1
- H: 4
- O: 2 * 2 = 4
Products:
- C: 1
- H: 2 * 2 = 4
- O: 2 + (2 * 1) = 4
Hooray! It’s balanced! This equation tells us that one molecule of methane reacts with two molecules of oxygen to produce one molecule of carbon dioxide and two molecules of water. See? With a little patience and a systematic approach, you can master the art of balancing equations!
It’s not just about passing a chemistry test; understanding balanced equations is a fundamental skill that opens doors to understanding so much more about the world around us. From the air we breathe to the fuel that powers our cars, chemical reactions are happening all the time, and balanced equations are our key to understanding their intricate dance. They are the silent symphony conductors of the molecular world, ensuring that every atom plays its part perfectly.
So, the next time you see a chemical equation, don't just see a jumble of letters and numbers. See a story, a recipe, a perfectly orchestrated event where matter transforms in fascinating ways. Embrace the challenge of balancing, and you’ll find a sense of satisfaction knowing you've mastered a fundamental principle of chemistry. And remember, even the most complex reactions start with a simple principle: making sure everything adds up! Keep exploring, keep questioning, and keep those equations balanced. You've got this, and the universe is full of amazing chemical wonders waiting for you to discover them!
