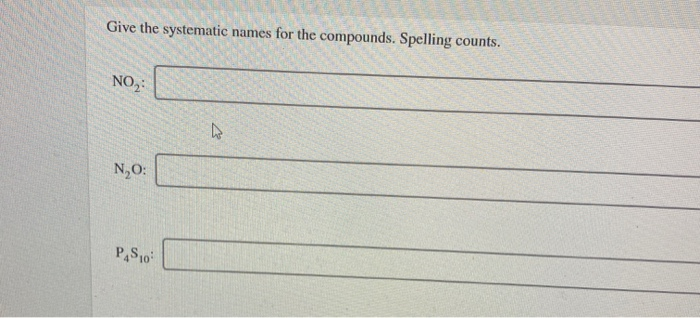
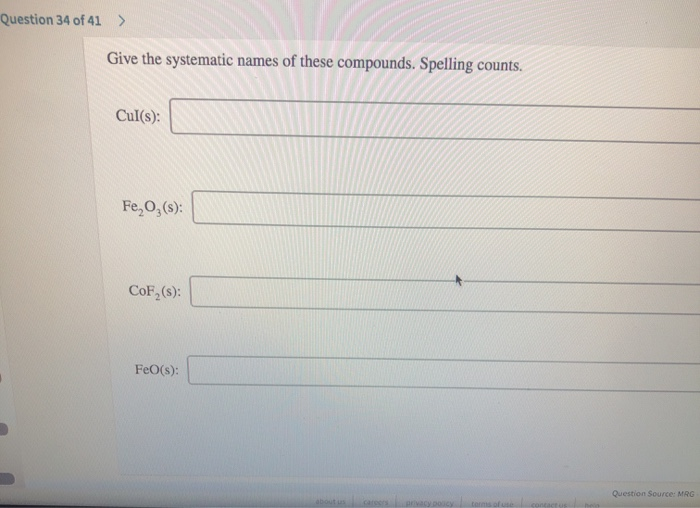
Give The Systematic Names Of These Compounds. Spelling Counts

Hey there, style mavens and curious minds! Ever find yourself staring at a fancy bottle of something – maybe a skincare serum, a cleaning product, or even a gourmet ingredient – and wondering what on earth all those long, complex names mean? You know, the ones that sound like they were invented by a wizard after a particularly challenging spellcasting session? We're talking about those systematic chemical names, the official monikers that tell us exactly what’s inside. And while they might seem intimidating, understanding them can be a seriously empowering and, dare I say, chic thing to do. Think of it as decoding the secrets of your everyday world, one syllable at a time. It’s like knowing the designer behind your favorite handbag; it adds a whole new layer of appreciation, right?
Today, we're diving headfirst into the intriguing universe of systematic chemical nomenclature. It’s not about becoming a chemistry professor overnight, but rather about gaining a little insight, a touch of knowledge that can make your everyday choices – from what you slather on your face to what you use to polish your kitchen counter – feel a lot more informed and, frankly, more intentional. We’ll keep it light, breezy, and sprinkled with fun facts, so grab your favorite beverage (mine’s a lavender latte, very calming!), get comfy, and let’s get this naming party started!
Unlocking the Code: Why Systematic Names Matter
So, why bother with these mouthfuls? Well, common names can be a bit of a wild west. Take, for example, "water." We all know it’s H₂O, but imagine if every single chemical compound had a unique, catchy nickname. Chaos! Systematic names, on the other hand, are like a universal language. They’re built on a logical system, and once you grasp the basics, you can actually deduce the structure of a molecule just from its name. How cool is that? It’s like a secret handshake in the world of science, and knowing it gives you a little insider cred.
Think about it like this: if you’re trying to order a specific coffee, you wouldn’t just say "coffee." You’d specify "iced latte with oat milk and an extra shot of espresso." Systematic names are the scientific equivalent of that precision. They leave no room for ambiguity, ensuring that everyone, everywhere, is talking about the exact same substance. This is crucial for safety, for innovation, and for simply understanding the ingredients that make up our modern lives.
The Building Blocks: Prefixes, Suffixes, and Roots
The beauty of systematic naming lies in its predictability. It’s like Lego bricks, but for molecules! Most systematic names are built from a few key components:
- Roots: These usually tell you the main part of the molecule, often the longest carbon chain in organic chemistry.
- Prefixes: These come before the root and tell you about things attached to the main structure – like extra groups of atoms or where they're located.
- Suffixes: These come after the root and tell you about the type of molecule it is – is it an alcohol? A ketone? An acid?
It’s a bit like building a sentence. You have your subject (the root), your descriptive adjectives (the prefixes), and your verb (the suffix) that tells you what’s happening. And just like in a sentence, the order matters!
A Peek into Organic Chemistry: The Carbon Crew
A huge chunk of the substances we encounter daily are organic compounds, meaning they are based on carbon atoms. The way these carbon atoms link up is what gives molecules their unique properties. And the systematic names for these often start by identifying the carbon backbone.
For example, let’s look at the names for simple alkanes, which are hydrocarbons with only single bonds between carbon atoms. This is where those prefixes come into play:
- Meth- (1 carbon)
- Eth- (2 carbons)
- Prop- (3 carbons)
- But- (4 carbons)
- Pent- (5 carbons)
- Hex- (6 carbons)
- Hept- (7 carbons)
- Oct- (8 carbons)
- Non- (9 carbons)
- Dec- (10 carbons)
See? Already, you're building a vocabulary. Add the suffix "-ane" (which signifies a saturated hydrocarbon), and you've got yourself some basic building blocks. So, a molecule with 5 carbons linked by single bonds is pentane. Simple, right? It’s like learning the first few chords on a guitar – suddenly, you can play a whole song!
Fun Fact: The smell of freshly cut grass is largely due to a group of organic compounds called green leaf volatiles (GLVs). Their names? Often tongue-twisters like (Z)-3-hexenol! But hey, the science behind that lovely aroma is pretty neat.
Introducing Functional Groups: The Personality Injectors
But molecules aren't just chains of carbon. They often have special groups of atoms called functional groups, which dramatically change their behavior. This is where the suffixes really shine.
Let’s take some common ones:
- -ol: Indicates an alcohol, meaning there's an -OH group attached. Think of ethanol (C₂H₅OH), the alcohol in your favorite adult beverages (in moderation, of course!). The "eth-" tells you it has two carbons, and "-ol" tells you it's an alcohol.
- -one: Indicates a ketone, which has a carbon atom double-bonded to an oxygen atom (C=O) within a carbon chain. Acetone, a common solvent found in nail polish remover, is a great example. Its systematic name is propanone (CH₃COCH₃), with three carbons and the ketone group on the middle one.
- -oic acid: Indicates a carboxylic acid, which has a -COOH group. Acetic acid, the key component that gives vinegar its tang, is systematically named ethanoic acid (CH₃COOH). Two carbons, and the characteristic acid group.
It's like giving characters to a story. Each functional group adds a distinct personality to the molecule, influencing everything from its smell and taste to its reactivity.
Putting It All Together: A Mini-Challenge
Let’s try naming a slightly more complex one. Imagine a molecule with a chain of four carbons, and an -OH group attached to the second carbon. The root for four carbons is "but-". The "-ol" suffix tells us it's an alcohol. And the "2-" prefix tells us where that alcohol group is attached.
So, its systematic name would be butan-2-ol. See? You just decoded a chemical name! This is the kind of molecule you might find in some rubbing alcohols or as a solvent in certain industrial applications. It’s all about following the rules and building the name piece by piece.
Cultural Snippet: The French, known for their sophisticated palates and culinary prowess, are masters of utilizing ingredients whose properties are dictated by their chemical structures. Think of the fermentation processes that create esters responsible for the fruity notes in wine, or the complex reactions that develop the rich flavors in cheese. Understanding these compounds, even at a basic level, adds an appreciation for the science behind the sensory delight.
Beyond Organic: Inorganic Wonders
While organic chemistry gets a lot of the spotlight, inorganic compounds have their own systematic naming conventions, and they’re equally important. These are the compounds that generally don’t contain carbon (with a few exceptions, like carbonates and cyanides). The rules here are a little different, often involving the names of the elements involved and their charges.
Salts: A Tale of Two Ions
Many inorganic compounds are salts, formed when a metal cation (positively charged ion) combines with an anion (negatively charged ion). The naming is pretty straightforward:

- You take the name of the metal.
- Then you take the root of the non-metal and add the suffix "-ide".
Let's look at some common ones:
- Sodium chloride (NaCl): The "sodium" part is the metal. The "chlor-" root from chlorine, with the "-ide" suffix, gives you "chloride". So, sodium chloride. This is, of course, common table salt!
- Magnesium sulfate (MgSO₄): "Magnesium" is the metal. "Sulfate" is a polyatomic anion (an ion made of multiple atoms). This compound is often found in Epsom salts, used for soothing baths.
- Calcium carbonate (CaCO₃): "Calcium" is the metal. "Carbonate" is another polyatomic anion. This is the main ingredient in chalk and seashells!
Pro-Tip: When you see "-ate" or "-ite" at the end of an inorganic name, it usually means you’re dealing with a polyatomic ion containing oxygen. For instance, chlorate (ClO₃⁻) and chlorite (ClO₂⁻). Think of them as special units that act like single elements when forming salts.
Oxides: The Oxygen Affair
Compounds containing oxygen and another element are called oxides. The naming here often involves prefixes to indicate how many oxygen atoms there are.
- Carbon dioxide (CO₂): "Carbon" is the element, and "di-" means two, referring to the two oxygen atoms. So, carbon dioxide. This is the stuff we exhale and that plants use for photosynthesis.
- Sulfur dioxide (SO₂): Similar logic, sulfur dioxide. A gas often associated with volcanic activity and pollution, but also used in winemaking as a preservative.
- Iron(III) oxide (Fe₂O₃): Here, we have a Roman numeral! This indicates the charge of the iron ion. Iron can have different charges (oxidation states), and it's important to specify which one. "Iron(III)" means the iron has a +3 charge. "Oxide" is for oxygen. So, iron(III) oxide, commonly known as rust.
The Roman numerals are like a subtle accent mark, telling you exactly which version of an element you’re dealing with. It’s the scientific equivalent of distinguishing between a simple espresso and a ristretto – both are coffee, but with crucial differences.
Acids and Bases: The pH Pals
Acids and bases have their own naming conventions too. Acids typically start with "hydro-" if they only contain hydrogen and one other element, or they end in "-ic acid" if they contain oxygen.
- Hydrochloric acid (HCl): This is the "hydro-" version because it's just hydrogen and chlorine.
- Sulfuric acid (H₂SO₄): This is the "-ic acid" version, derived from the sulfate ion.
Bases often contain a metal and a hydroxide group (-OH). Their names are formed like salts:
- Sodium hydroxide (NaOH): "Sodium" and "hydroxide". This is a strong base, also known as lye, used in soap making and drain cleaners. Handle with care!
- Potassium hydroxide (KOH): "Potassium" and "hydroxide". Another strong base, often used in batteries.
Fun Fact: The ancient Romans used vinegar (acetic acid) to clean their marbles and statues, recognizing its mild acidic properties even without knowing the exact systematic name!
Practical Tips for the Curious Consumer
So, how can you use this newfound knowledge in your daily life? It’s not about memorizing a chemical dictionary, but about developing an awareness.
- Skincare Savvy: Look at the ingredient list on your favorite moisturizer or serum. If you see names like hyaluronic acid (a humectant that keeps skin hydrated) or tocopherol (which is Vitamin E), you’ll know they’re there for a reason! Understanding these can help you choose products that truly benefit your skin.
- Cleaning Consciously: When picking up a household cleaner, you might see names like sodium hypochlorite (the active ingredient in bleach) or ammonia (often used in glass cleaners). Knowing these basic components can help you understand their purpose and potential hazards. Always follow label instructions!
- Foodie Fun: In gourmet ingredients or even packaged foods, you might encounter names like lecithin (an emulsifier often derived from soy or sunflower) or citric acid (a common food additive for tartness and preservation). It’s a way to peek behind the culinary curtain.
- DIY Smarts: If you're into DIY projects, whether it's making your own cleaning solutions or crafting soaps, understanding the basic chemistry of your ingredients is key to safety and success. For example, knowing that sodium hydroxide is a strong base is essential for safe soap making.
Inspiration Station: Think of the minimalist aesthetic. It’s about stripping away the unnecessary to reveal the essential. Similarly, systematic names strip away ambiguity to reveal the exact chemical identity. It’s a form of clarity, and there’s a certain beauty in that precision, wouldn’t you agree?
A Final Thought: The Art of Understanding
Navigating the world of systematic chemical names might seem like a deep dive, but it’s more like dipping your toes in. The more you engage with these names, the more familiar they become. They transform from intimidating strings of letters into informative labels, telling a story about the substance’s composition and properties.
In our everyday lives, we’re constantly interacting with a complex chemical ballet. From the air we breathe to the food we eat, the clothes we wear, and the products we use, chemistry is everywhere. By taking a moment to appreciate the systematic names, we’re not just learning science; we’re gaining a deeper understanding of the world around us. It’s about being more mindful consumers, more informed individuals, and perhaps, just a little bit more amazed by the intricate, elegant workings of the universe. So next time you see a long chemical name, don’t shy away. Embrace it, decode it, and enjoy the little spark of knowledge it brings. It’s a small step, but it opens up a whole new perspective, and that, my friends, is always in vogue.
