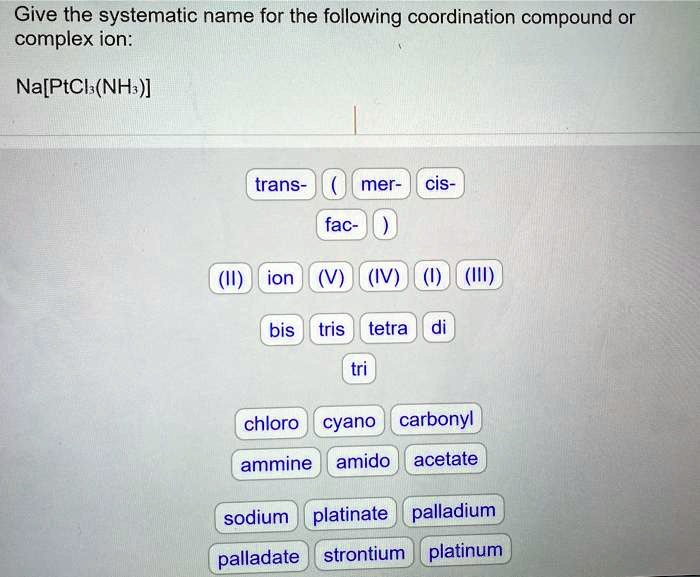
Give The Systematic Name For This Coordination Compound
Hey there, science enthusiasts and curious minds! Ever glance at a complex chemical formula and think, "Whoa, that looks like a tiny, intricate city made of atoms!" Well, you're not wrong! Today, we're diving into the wonderfully quirky world of coordination compounds and, specifically, tackling a naming challenge that might seem a little intimidating at first. But trust me, by the end of this, you'll be feeling like a chemistry detective, ready to crack any molecular code!
So, what exactly is a coordination compound? Think of it like a fancy molecular handshake. You have a central metal atom (or ion) acting as the host, and around it, you have a bunch of other molecules or ions, called ligands, holding on tight. These ligands are like loyal companions, donating their electrons to the metal. It's a beautiful dance of attraction and electron sharing that creates some truly amazing substances!
Now, the universe of chemistry has a super-organized way of naming things. It's like giving every single person on Earth a unique, specific name so we don't get confused. And for these cool coordination compounds, there’s a systematic name that tells you exactly what’s going on inside. It’s not just a random jumble of letters and numbers; it’s a descriptive masterpiece!
Let's get to the good stuff, shall we? The challenge! We're going to tackle this gem: [Co(NH₃)₆]Cl₃. Ooh, fancy! At first glance, it might look like a secret code, but fear not! We’re going to break it down piece by piece, and you'll see it's not as scary as it seems. In fact, it’s kinda fun, like solving a delicious riddle.
First things first, let’s identify our players. See those brackets? [...] That whole section inside is our coordination sphere. It’s the main event, the star of the show. Inside our coordination sphere, we have a central metal: Co. Do you recognize that? Yep, that’s cobalt! Cobalt is a pretty cool element, often found in pigments that give glass and ceramics those vibrant blues we love. Imagine, your coffee mug might have a bit of cobalt in it!
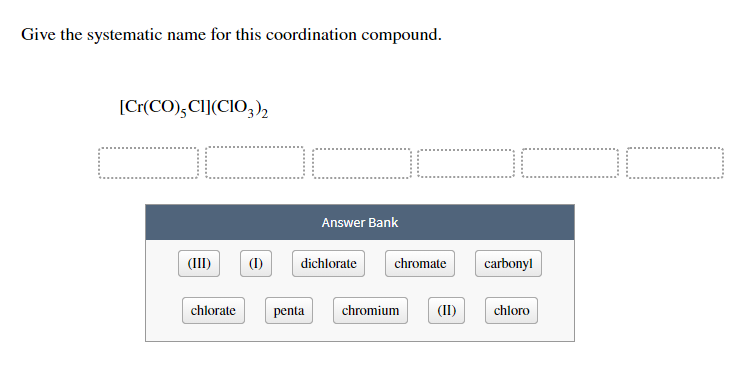
Now, look at what's hanging out with our cobalt. We have (NH₃)₆. The NH₃ part is ammonia. In the world of coordination chemistry, when ammonia acts as a ligand, we give it a special name: ammine. And that little number 6? It tells us there are six of these ammine ligands surrounding our cobalt. So, within the coordination sphere, we have hexaammine (hexa meaning six!). Pretty straightforward, right? It’s like saying "six companions."
So, our coordination sphere is shaping up to be hexaamminecobalt. But wait, there’s a little twist! The name of the metal in the coordination sphere changes if the compound is an anion (negatively charged). In this case, our coordination sphere is a cation (positively charged), so we stick with the metal's normal name: cobalt. Phew! One hurdle cleared!
Now, let’s look outside the brackets. We have Cl₃. These are our counterions. They're there to balance the charge of the coordination sphere. Think of them as the helpful balancing act that keeps the whole thing stable. In this case, we have three chloride ions. If there were just one, we’d call it chloride. But since there are three, we use the prefix tri-. So, we have trichloride.
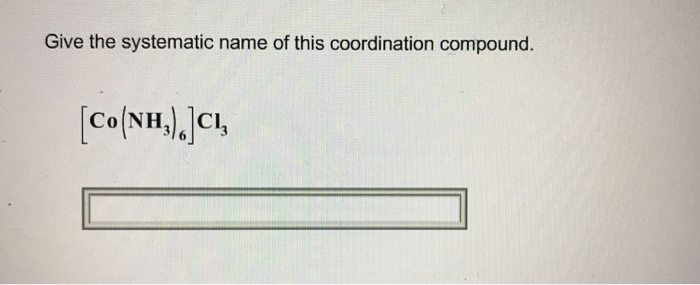
Putting it all together, we have the coordination sphere first, followed by the counterions. So, it’s hexaamminecobalt… and then… trichloride!
But hold on a sec! We need to be super precise. What’s the charge on our cobalt? This is where things get a little more detective-y. We know chloride has a charge of -1. Since we have three chlorides, the total negative charge from the counterions is 3 x (-1) = -3. To make the entire compound neutral, our coordination sphere (hexaamminecobalt) must have a positive charge of +3 to balance that -3. So, our cobalt is actually cobalt(III)! That little Roman numeral in parentheses tells us the oxidation state, or the "charge," of the metal. It’s a crucial detail!

So, let’s refine our name. We have hexaamminecobalt(III) as our coordination sphere, and then our counterion: trichloride. Combined, we get: Hexaamminecobalt(III) chloride.
Ta-da! Doesn't that sound so much more impressive than just a jumble of letters? You’ve just deciphered a complex chemical name! This systematic naming system is a marvel of organization. It allows chemists all over the world to communicate precisely about the molecules they're working with. It’s like a universal language for the microscopic world.
Why does this matter? Well, imagine the possibilities! These coordination compounds aren’t just abstract ideas. They have real-world applications that can make our lives better and more interesting. They’re used in everything from medicines and vitamins to catalysts that speed up industrial processes, and even in the pigments that give your favorite jeans their color!

Think about it. The next time you see a vibrant blue pigment, you can wonder about the coordination compounds that create that hue. Or when you take a vitamin, you can appreciate the intricate molecular structures that are helping your body thrive. It adds a whole new layer of appreciation to the everyday!
Learning to name these compounds might seem like a small step, but it’s a gateway to understanding a vast and fascinating field. It's a reminder that even the most complex systems have an underlying logic and order. And once you grasp that logic, the seemingly impenetrable becomes beautifully understandable, even fun!
So, don't shy away from those chemical formulas! Dive in! Ask questions! Explore the systematic names of other coordination compounds. You might just discover a new passion, a new way of seeing the world, and a newfound appreciation for the incredible artistry of chemistry. Keep exploring, keep learning, and keep being amazed by the wonders of the molecular universe!
