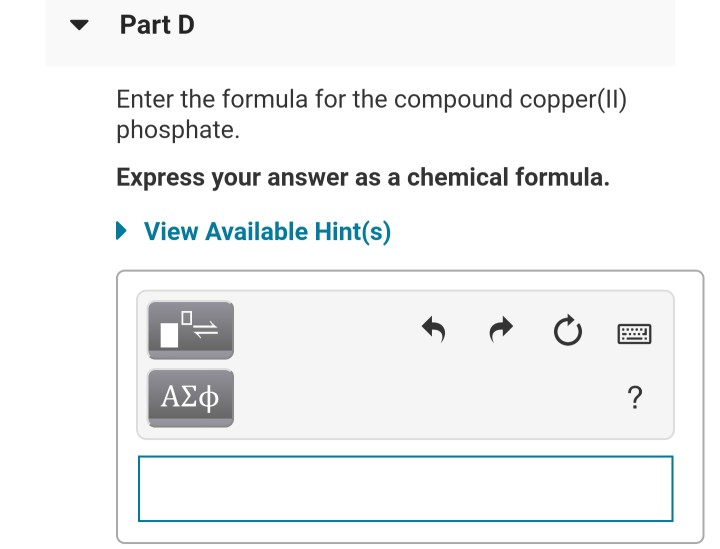
Give The Systematic Name For The Compound Al No3 3
You know, sometimes the most ordinary things around us have the most fascinating, and dare I say, downright quirky names hidden beneath the surface. We’ve all seen it, right? That little white powder that pops up in unexpected places, often playing a starring role in science experiments that fizz and pop in a glorious, albeit sometimes sticky, way. Today, we’re going to shine a spotlight on one of these chemical celebrities, a compound that might just have you saying, "Wait, that's what it's called?" We're talking about the one and only Aluminum Nitrate, or as it's known in the wonderfully verbose world of chemistry, Al(NO₃)₃. Now, I know what you're thinking. "Aluminum Nitrate? Sounds a bit… plain." And honestly, you wouldn't be entirely wrong. But behind this straightforward moniker lies a little tale that’s surprisingly sweet, a bit mysterious, and definitely worth a chuckle.
Imagine a shy, unassuming character in a play. That’s a bit like how Aluminum Nitrate often acts. It’s not the flashy, attention-grabbing villain or the dramatic hero. It’s more of a supporting actor, present and important, but rarely hogging the spotlight. This compound is made up of two main parts: the ever-so-sturdy Aluminum, and the rather zippy Nitrate group. Think of Aluminum as the reliable friend, always there to lend a hand, and Nitrate as the energetic spark, bringing a bit of zest to the party. Together, they form a bond that's quite useful, though not quite as exciting as, say, a volcano erupting or a rocket blasting off. Still, its usefulness is undeniable, which is why we encounter it in a variety of places, from fancy fireworks that light up the night sky with a dazzling display, to fertilizers that help our gardens grow into lush, green oases. It’s the quiet achiever of the chemical world, working diligently behind the scenes.
Now, let’s talk about the name itself, the systematic name. In the grand ballroom of chemical nomenclature, where every element and compound has its official dance card, Aluminum Nitrate is actually just the common name. The more formal, the one you’d see in a chemistry textbook or on a scientific journal’s cover, is Aluminum(III) nitrate. Yes, that little Roman numeral three is a crucial detail. It tells us that the Aluminum atom is in a particularly positive mood, having decided to share three of its electrons. This little detail is like a secret handshake among chemists, a quick way to understand the compound's precise personality. It’s a bit like knowing someone’s full name versus just their nickname. Both refer to the same person, but one carries a little more official weight and precision.
The journey to naming compounds like this is a fascinating one, born out of a need for clarity and consistency. Imagine if everyone just called their favorite stuff by a different name! It would be utter chaos. So, the chemists, bless their organized hearts, sat down and created a system, a universal language for these molecular marvels. This systematic naming ensures that no matter where you are in the world, if you say Aluminum(III) nitrate, everyone knows exactly which molecule you’re talking about. It’s like the difference between saying "soda," "pop," or "fizzy drink" – all referring to the same bubbly beverage, but the systematic name is like the brand name that’s universally recognized.
It's a beautiful dance of elements, orchestrated by rules that ensure everyone knows their place and their role. Even for something as seemingly simple as Al(NO₃)₃, there's a whole story of organization and clarity behind its designation.
Solved Part A Give the systematic name for the compound | Chegg.com
What’s truly heartwarming, though, is how this seemingly mundane compound can contribute to things that bring us joy. Think about those breathtaking fireworks displays that punctuate summer nights and celebrations. Aluminum Nitrate, in a supporting role of course, can help create those brilliant flashes and vibrant colors. It’s a tiny piece of a much larger, more spectacular puzzle. Or consider the humble tomato plant in your garden, reaching for the sun. Fertilizers containing nitrates, often in forms derived from compounds like Aluminum Nitrate, provide the essential nutrients for that plant to flourish, eventually leading to those juicy, sun-ripened tomatoes that taste like pure summer. It’s a full circle of goodness, from a carefully named chemical to the simple pleasures of life.
So, the next time you see a flash of color in the night sky or bite into a delicious homegrown vegetable, you can have a little secret smile. You know that behind the scenes, the well-named Aluminum Nitrate, or Aluminum(III) nitrate to be precise, played its part. It’s a reminder that even in the often-complex world of science, there’s beauty, order, and a touch of everyday magic. And that, my friends, is a pretty sweet deal for a simple white powder.