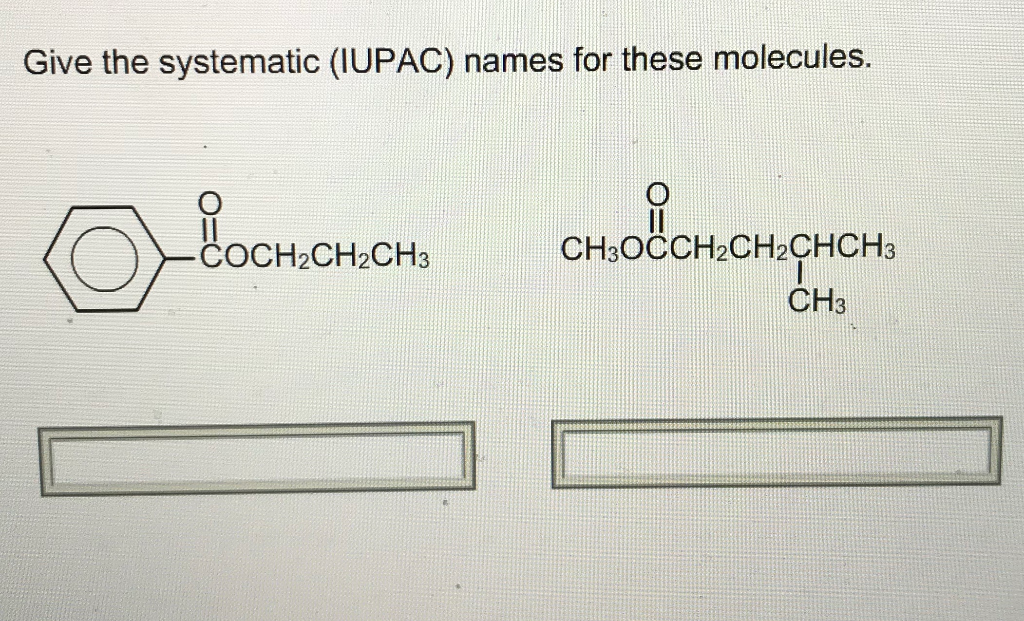
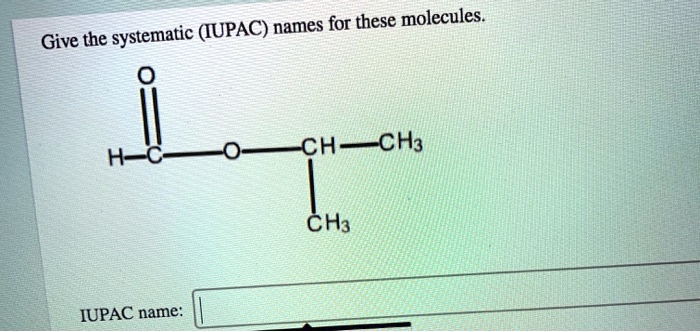
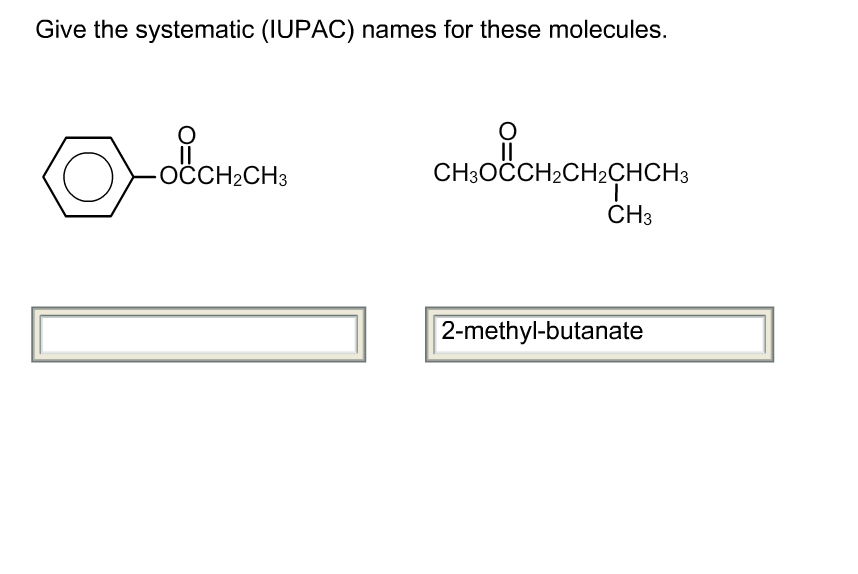
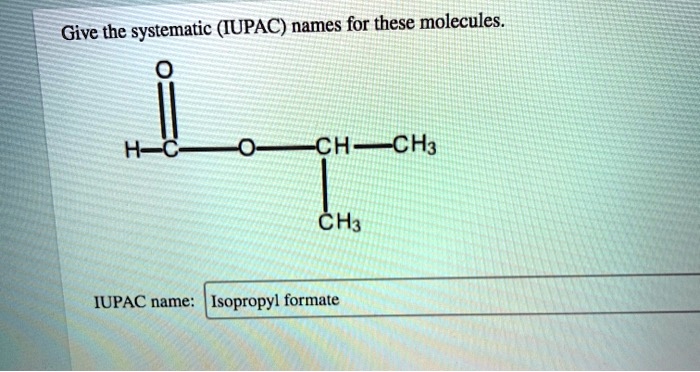
Give The Systematic Iupac Names For These Molecules
Hey there, fellow humans navigating the delightful chaos of modern life! Ever glance at a cool molecule in a science documentary or a quirky art piece and wonder what its real name is? We're talking about the official, the universally recognized, the fancy names that chemists use. No, not just "that one with the rings" or "the bubbly one." We're diving into the world of IUPAC nomenclature – think of it as the ultimate style guide for chemical compounds. It sounds intimidating, but trust me, it's more like learning a cool new language that unlocks a secret world. Today, we’re breaking down the systematic IUPAC names for a few intriguing molecules, making it as easy-breezy as finding the perfect avocado. Get ready to flex those brain muscles, but keep it chill, because learning should be fun, right? It’s like mastering the art of the perfectly brewed iced coffee – a little effort, a lot of reward.
Unlocking the Secrets: Why IUPAC Names Matter
So, why bother with these seemingly complex names? Well, imagine trying to order a specific latte without knowing the difference between a macchiato and a cortado. Chaos! Similarly, in chemistry, having a standardized naming system, the IUPAC (International Union of Pure and Applied Chemistry) system, ensures everyone, everywhere, is talking about the exact same chemical. It's like the universal handshake of the science world. This system is built on logic, a bit like how a great playlist is built on genre and tempo. It tells you a molecule's structure, its backbone, and its functional groups – all from a single name. Pretty neat, huh? It's the behind-the-scenes magic that keeps scientific discovery humming along smoothly.
The Building Blocks: Hydrocarbons are Our Friends
Let's start with the basics, the OG building blocks of organic chemistry: hydrocarbons. These are molecules made up only of hydrogen and carbon atoms. Simple, right? They're the foundational notes in the symphony of organic molecules. Think of them as the plain white tee of your wardrobe – versatile and essential. The IUPAC naming for hydrocarbons is all about prefixes that tell you how many carbon atoms are in the main chain, and a suffix that tells you what kind of bonds are between them. Easy peasy.
Molecule #1: CH4 – The Simplest Star
First up, the superstar of simplicity: CH4. This little guy is everywhere. It's what natural gas is made of, and it plays a role in everything from the atmosphere to our digestive systems (yep, that's right!).
To name CH4 systematically using IUPAC, we look at the number of carbon atoms. There's only one. The prefix for one carbon atom is "meth-". Then, we look at the bonds. In CH4, the carbon is bonded to four hydrogen atoms with single bonds. Single bonds in hydrocarbons mean the suffix is "-ane".
Put it together, and you get: methane.
Fun Fact: Methane is the simplest alkane, and it's also a potent greenhouse gas. So, while it’s fundamental, its prevalence has real-world implications. It’s like that friend who’s super chill but also surprisingly influential in group decisions!
Molecule #2: C2H6 – The Next Step Up
Moving on to our next molecule, C2H6. This one has two carbon atoms. The prefix for two carbons is "eth-". Again, all the bonds between the carbons and hydrogens are single bonds, so the suffix remains "-ane".
This gives us: ethane.
Practical Tip: Ethane is a component of natural gas and is used in the petrochemical industry to produce ethylene. Think of it as the slightly older, more established sibling to methane, still essential but with a bit more to offer.
Molecule #3: C3H8 – The Trio
Now for C3H8. We have three carbon atoms. The prefix for three carbons is "prop-". And, you guessed it, it's all single bonds, so the suffix is still "-ane".
The IUPAC name? Propane.
Cultural Connection: Propane is the fuel that powers many backyard barbecues and camping stoves. It's the taste of summer cookouts and the warmth of a cozy campsite. When you smell that familiar scent at a grill, you're smelling propane at work! It’s the unsung hero of outdoor gatherings.
Molecule #4: C4H10 – Branching Out (Slightly!)
Okay, things are getting a tiny bit more interesting with C4H10. We have four carbon atoms. The prefix for four carbons is "but-". And still, we have single bonds, so the suffix is "-ane".
So, the name is butane.
The Plot Twist: Now, here’s where IUPAC really shines. For C4H10, there’s actually more than one way these atoms can be arranged. They can be in a straight line, or they can have a little branch. The straight-chain version is called n-butane (the 'n' stands for normal, meaning unbranched). If it has a branch, it gets a different, more specific name (which we’ll touch on briefly later, but don't worry, it’s not a test!). This ability to distinguish between isomers (molecules with the same formula but different structures) is crucial in chemistry.
Fun Little Fact: Butane is commonly used as a fuel in lighter fluid and aerosol propellants. It’s the invisible force that helps you light your candle or spray your hairspray.

Beyond the Straight and Narrow: Introducing Alkenes and Alkynes
So far, we've been talking about alkanes – hydrocarbons with only single bonds. But what happens when there are double or triple bonds? This is where the suffix changes, and we enter the worlds of alkenes (double bonds) and alkynes (triple bonds).
For alkenes, the suffix changes from "-ane" to "-ene". For alkynes, it changes to "-yne". The position of these double or triple bonds also needs to be indicated in more complex molecules, but for our simple examples, we’ll keep it straightforward.
Molecule #5: C2H4 – The Double Deal
Let's look at C2H4. We have two carbon atoms, so the prefix is "eth-". But this time, there's a double bond between the two carbon atoms. This means it’s an alkene.
The suffix becomes "-ene". So, the name is ethene.
Practical Tip: Ethene (also commonly known as ethylene) is a vital industrial chemical. It's used to produce polyethylene, the plastic found in everything from grocery bags to water bottles. It’s the silent workhorse behind much of our modern packaging.
Molecule #6: C2H2 – The Triple Threat
Now for C2H2. Two carbon atoms, prefix "eth-". This molecule has a triple bond between the carbon atoms. This makes it an alkyne.
The suffix changes to "-yne". Therefore, the name is ethyne.
Cultural Connection: Ethyne, often called acetylene, is famously used in oxy-acetylene torches for welding and cutting metal. It generates incredibly high temperatures. Think of the sparks flying in a blacksmith's shop or a construction site – that’s ethyne in action! It’s the powerful punch of the hydrocarbon family.
Adding Some Flair: Functional Groups Enter the Chat
Hydrocarbons are fantastic, but the real party starts when we add other atoms or groups of atoms, called functional groups. These groups give molecules their unique properties and reactivity, like adding spices to a dish. They’re the personality traits of the chemical world.
Molecule #7: CH3OH – The Alcohol Aha! Moment
Meet CH3OH. This looks a bit different. We have a carbon atom bonded to three hydrogens and then to an oxygen atom, which is itself bonded to a hydrogen. This -OH group is a hydroxyl group, and when attached to a carbon chain, it makes the molecule an alcohol.
Let's break it down: The hydrocarbon part has one carbon, so it starts with "meth-". Since the primary chain is like methane and there are no double or triple bonds within the carbon structure, we might think of the "-ane" base. However, the IUPAC system has specific rules for naming compounds with functional groups. For simple alcohols, we replace the final "-e" of the parent alkane name with "-ol".
So, from methane (CH4), we remove the "-e" to get "meth-" and add "-ol", resulting in: methanol.
Practical Tip: Methanol is a versatile chemical used as a solvent and a fuel additive. It’s also known as wood alcohol. Historically, it was produced by the destructive distillation of wood. Just a heads-up: it's toxic, so admire it from afar!
Molecule #8: CH3COOH – The Sour Note
Now for CH3COOH. This molecule has a carbon atom with three hydrogens, attached to another carbon atom. This second carbon is bonded to an oxygen atom with a double bond, and also to a hydroxyl (-OH) group. This whole -COOH group is a carboxyl group, and molecules containing it are called carboxylic acids.
Let's analyze: We have a two-carbon chain. The parent alkane would be ethane. However, the presence of the carboxyl group dictates the naming. We take the alkane name corresponding to the number of carbons (ethane), drop the final "-e", and add "-oic acid".
So, from ethane, we get "ethan-" and add "-oic acid", giving us: ethanoic acid.
Cultural Connection: Ethanoic acid is the key component that gives vinegar its characteristic sour taste and pungent smell. Diluted, it's a common household staple. Think of salad dressings, pickling, and that distinctive aroma when you open a jar of pickles. It’s the tangy zest of life!
Molecule #9: C6H6 – The Ring of Power
Finally, let's tackle C6H6. This one is a classic and a bit of a celebrity in the chemical world: benzene. While its formula is simple, its structure is quite unique. It's a ring of six carbon atoms, each bonded to one hydrogen, with alternating double and single bonds within the ring. This arrangement creates a stable, aromatic system.
IUPAC officially recognizes benzene as a root name for many related compounds. While you could describe it in a more complex way based on its structure, its common name, benzene, is widely accepted and used by IUPAC for simplicity and historical reasons.
Fun Little Fact: Benzene is a foundational molecule in organic chemistry and is the parent compound of many aromatic compounds. It's the basis for many dyes, plastics, and even some pharmaceuticals. It’s like the sophisticated, well-traveled aunt of the hydrocarbon family.
Embracing the Nomenclature Lifestyle
Learning IUPAC names might seem like a chore at first, like trying to remember all the different coffee brewing methods. But once you get the hang of the prefixes, suffixes, and the logic behind them, it opens up a whole new way of understanding the world around you. Every molecule, from the air you breathe to the food you eat, has a story told by its name. It’s a beautiful, organized language that connects us to the fundamental building blocks of existence. So, the next time you see a chemical formula, don't be intimidated. Think of it as a puzzle waiting to be solved, a secret code to be deciphered. Embrace the nomenclature, and you'll find a new appreciation for the intricate dance of atoms and molecules that shapes our reality.
In the grand scheme of things, understanding these names is like learning to read the ingredients list on your favorite snack. It might not change your life overnight, but it adds a layer of awareness and appreciation. It’s a small step towards understanding the complex tapestry of the universe, one systematic name at a time. And isn't that, in its own quiet way, a rather satisfying feeling?
