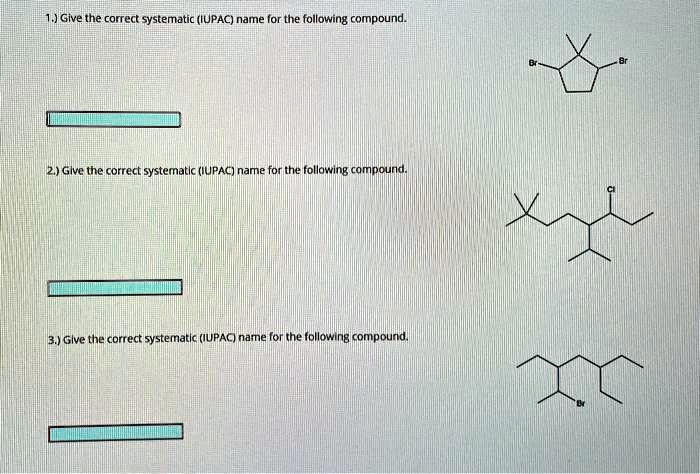
Give The Systematic Iupac Name For The Following
Ever found yourself staring at a complex string of letters and numbers, perhaps on a bottle of medicine, a food label, or even in a science textbook, and wondered, "What is that?" You're not alone! For many, the world of chemical nomenclature, particularly the systematic IUPAC naming convention, can feel a bit like deciphering an ancient code. But far from being a dry academic exercise, understanding these names is surprisingly useful and can even be a source of… dare we say… fun!
Why do people engage with this? It’s all about clarity and precision. Imagine trying to order a specific type of cake without being able to describe its ingredients accurately. You might end up with something completely unexpected! Similarly, IUPAC (International Union of Pure and Applied Chemistry) names provide a universal language for chemists and scientists worldwide. This ensures that when someone refers to, say, "2-methylpropan-1-ol," everyone knows they're talking about the same exact molecule, not some similar-looking but functionally different substance. This eliminates ambiguity and is crucial for safety, research, and commerce.
The benefits for everyday life might seem subtle, but they're significant. Think about the medications you take. The active ingredient often has a long, systematic IUPAC name. Knowing this name, or at least understanding its structure through its name, can empower you to understand what you're ingesting and why. It's also vital in the food industry; imagine the chaos if different manufacturers used different names for the same preservative or flavoring! Even in your own kitchen, understanding the names of common organic compounds (which often follow IUPAC rules) can be a gateway to understanding cooking chemistry – why bread rises, why oil and water don't mix, and so on.
Common examples are all around us, even if we don't always recognize the systematic name. That simple table salt? Its IUPAC name is sodium chloride. The alcohol in your hand sanitizer? Often propan-2-ol (also known as isopropyl alcohol). Even common plastics like polyethylene have systematic names related to their repeating monomer units. The challenge, and indeed the enjoyment for some, lies in breaking down these complex names into their constituent parts to understand the molecule's structure. It’s like a molecular puzzle!
So, how can you enjoy this activity more effectively? Start small! Don't try to memorize the entire IUPAC rulebook overnight. Pick a common molecule you encounter, like aspirin (acetylsalicylic acid) or caffeine (1,3,7-trimethyl-1H-purine-2,6(3H,7H)-dione), and try to understand how its name reflects its structure. There are fantastic online resources and apps that can help you visualize molecules and break down their names. Think of it as a mental workout. It sharpens your analytical skills and provides a deeper appreciation for the intricate world of chemistry that surrounds us. Embrace the challenge, and you might find yourself unlocking a new level of understanding about the materials that make up our world.
