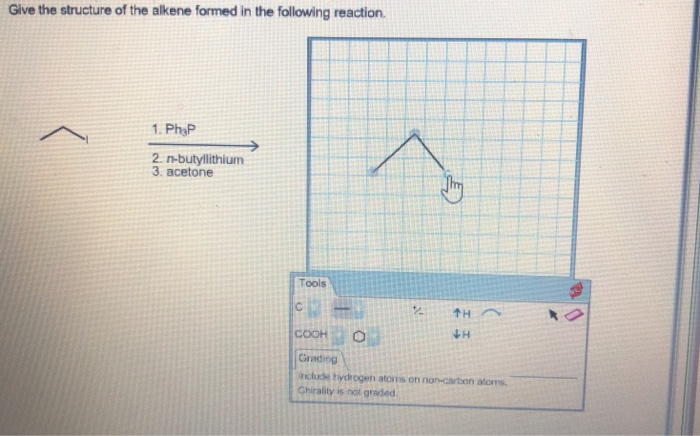
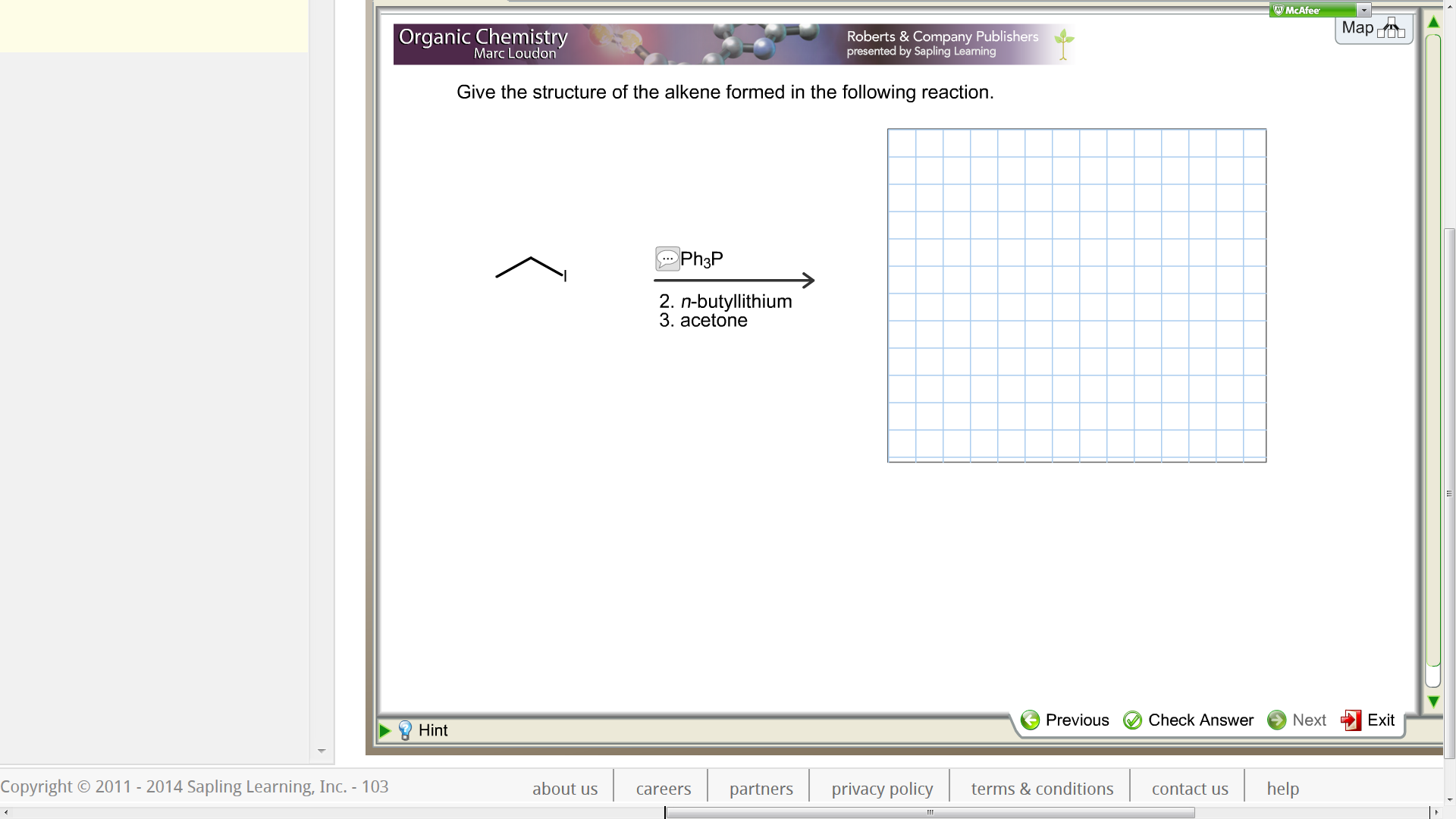
Give The Structure Of The Alkene Formed In The Reaction.
Ever wondered what makes things smell so sweet, or how we get those vibrant colors in paints and plastics? Well, get ready for a behind-the-scenes peek into the molecular world, because today we're diving into the fascinating realm of alkenes! These clever little molecules are the building blocks for so many everyday wonders, and understanding their structure is like unlocking a secret code to how nature and industry create amazing things. Forget boring textbooks; we're going to explore why these molecules are not only scientifically significant but also incredibly cool.
Think of alkenes as nature's versatile connectors. They are a class of hydrocarbons, meaning they're made up of just carbon and hydrogen atoms. What makes them special, and frankly, quite fun to study, is their defining feature: a carbon-carbon double bond. This double bond isn't just an extra bond; it's a hub of reactivity and a key determinant of their shape and how they interact with other molecules. This means alkenes are often involved in chemical reactions that create more complex and useful compounds. It's this inherent 'eagerness' to react that makes them so valuable in so many different applications.
The Magic of the Double Bond: Why It Matters
The carbon-carbon double bond (C=C) is the star of the show when it comes to alkenes. In a typical single bond between two carbon atoms, electrons are shared equally in a straightforward, flexible connection. But in a double bond, there are two pairs of electrons being shared. This creates a stronger, more rigid connection than a single bond. It also means that the region around the double bond is electron-rich, making it a prime target for other molecules looking to react.
This electron density is what gives alkenes their characteristic reactivity. They readily undergo addition reactions, where other atoms or groups can 'add' across the double bond, breaking one of the bonds and forming new single bonds. This is super useful because it allows chemists to transform simple alkenes into a vast array of more complex molecules with specific properties. It’s like having a Lego brick with a special connection point that lets you build almost anything!
The double bond dictates not only reactivity but also influences the shape of the alkene molecule. The atoms attached to the double-bonded carbons tend to lie in the same plane, giving these molecules a degree of rigidity.
From Simple to Sophisticated: The Purpose and Benefits of Alkenes
So, why do we care about the structure of alkenes? Because their structure directly dictates their function! The presence and position of the double bond, along with the types of groups attached to the carbon atoms involved in the double bond, determine how an alkene will behave in a chemical reaction. This predictability is crucial for chemists designing new materials and processes.

One of the most significant benefits of understanding alkene structure is its role in polymerization. Many plastics we use every day, like polyethylene (used in plastic bags and bottles) and polypropylene (used in containers and textiles), are made from repeating alkene units called monomers. Under specific conditions, these alkene monomers link together end-to-end to form long chains, creating these versatile polymers. The structure of the original alkene monomer directly influences the properties of the resulting polymer – its flexibility, strength, transparency, and resistance to heat or chemicals.
Beyond plastics, alkenes are vital in the synthesis of countless other important chemicals. They are precursors to alcohols, which are used as solvents and fuels. They can be converted into epoxides, highly reactive compounds used in adhesives and coatings. The fragrance industry relies heavily on alkene chemistry to create artificial flavors and scents, often by building complex carbon skeletons that originate from simpler alkenes.
Putting It All Together: The Structure of the Formed Alkene
When an alkene is formed in a reaction, the key aspect to identify is the location of that crucial carbon-carbon double bond. We need to know which carbon atoms are sharing those two pairs of electrons. Additionally, we look at what other atoms or groups are attached to these double-bonded carbon atoms. This information allows us to predict the alkene's physical and chemical properties.
For instance, if we have a simple alkene like ethene (CH₂=CH₂), it has the simplest possible double bond with no other groups attached to the double-bonded carbons. But if we have propene (CH₃CH=CH₂), one of the double-bonded carbons has a methyl group (CH₃) and a hydrogen atom attached, while the other has two hydrogen atoms. This seemingly small difference in attached groups can influence how the propene reacts and the properties of any polymers it might form.
The arrangement of atoms around the double bond can also lead to stereoisomers. If the groups attached to each double-bonded carbon are different, there can be two possible spatial arrangements of these groups, known as cis and trans isomers. This is analogous to two people sitting on either side of a rigid table; their relative positions can be different. These different spatial arrangements can have a significant impact on how the molecule interacts with other molecules, affecting its biological activity or its behavior in chemical processes. So, when we describe the structure of an alkene formed in a reaction, we're not just drawing a simple formula; we're describing its precise molecular architecture, which holds the key to its identity and potential.
