Give The Numerical Value Of N Corresponding To 3p

Alright, gather 'round, you magnificent humans! Have I got a story for you. It involves numbers, tiny invisible things, and a journey so epic it makes Indiana Jones look like he’s looking for his car keys. We're talking about a quest to find the numerical value of something called '3p'. Now, before you picture a tiny, three-legged penguin trying to pay for his fish dinner, let me assure you, it's a little more complicated, but way, way cooler. Think of it as the secret handshake of the universe. And like most secret handshakes, it's surprisingly simple once you know it, and frankly, a bit embarrassing if you get it wrong.
So, imagine you’re a cosmic detective, right? You’ve got your trench coat, your fedora, and a magnifying glass so powerful it can see into the very soul of matter. Your mission, should you choose to accept it (and let's be honest, who wouldn't want to crack the universe's biggest secrets?), is to understand the building blocks of everything. And by 'building blocks,' I mean atoms. Yes, those things we learned about in school that were probably as exciting as watching paint dry, but are actually the rockstars of existence. They’re like the ultimate Lego bricks, except way smaller and infinitely more impressive.
Now, within these atoms, there are even smaller dudes. We’re talking subatomic particles. Think of atoms as bustling cities, and these particles are the citizens. Some are the grumpy old men in their rocking chairs (neutrons, but we'll get to that), some are the energetic teenagers zipping around (electrons, maybe?), and then there are the guys in the middle, the heart of the city, the real VIPs: the protons. Protons are like the mayors of the atomic city, and they carry a very important ID badge: a positive charge. They’re the life of the party, always broadcasting their positivity. You can’t miss ‘em.
So, we’ve got protons, the positively charged VIPs. And then we have their polar opposites, the brooding, negative types: the electrons. Electrons are like the rebellious teenagers of the atomic world, constantly orbiting the nucleus (where the protons and neutrons hang out) like they’re trying to escape their parents. They have a negative charge. It’s like a cosmic battle of good versus evil, except it’s happening trillions of times a second in every single thing you can see, touch, or even smell. Smells are just atoms doing their thing, you know?
And then there are the neutrons. Oh, the neutrons. These guys are the silent types. They’re in the nucleus, chilling with the protons, but they’re neutral. No charge. They’re like that friend who’s always at the party but never says anything, just nods along. They’re the glue holding the nucleus together, preventing the positive protons from saying, "Peace out!" and flying away from each other. Imagine trying to herd cats, but the cats are all positively charged and actively want to repel each other. That’s where neutrons come in, the ultimate cat whisperers of the subatomic realm.

Now, here’s where things get a little spicy. These particles, these protons, neutrons, and electrons, they’re not just randomly floating around. They have these things called quantum numbers. Think of quantum numbers as the unique ID numbers for each particle. It's like a cosmic social security system, but way more complex and with fewer forms to fill out. Each electron in an atom gets a special set of these numbers that describes its exact location, energy, and spin. It’s like a very, very, very specific address and personality profile for each tiny particle.
We’ve got four main quantum numbers. The first is the principal quantum number (n). This one is like the apartment building number. It tells you how far from the nucleus an electron generally is, and how much energy it has. Higher 'n' means further away and more energy. Think of it as the floor number in our atomic skyscraper. Then there’s the angular momentum quantum number (l). This one is like the apartment number within the building. It describes the shape of the electron's orbital – is it a round ball, a dumbbell shape, or something even weirder? It dictates the subshell within the main energy level. And then… and then comes the one we’re interested in, in a way. It's part of a pair, actually.
We have the magnetic quantum number (ml). This one is like the specific apartment number on the floor. It tells you the orientation of the orbital in space. So, if you have a dumbbell shape (l=1), it can point along the x-axis, y-axis, or z-axis. Ml distinguishes between these orientations. It’s like having three identical apartments on the same floor, but one faces north, one east, and one south. They’re all the same size and shape, but their spatial arrangement is different. Pretty neat, huh? It’s like the universe giving us options for where our electrons can chill.

And finally, the last little nugget of information about an electron is its spin quantum number (ms). This one is super cool. Electrons have this property called 'spin', which is kind of like them spinning on their own axis, either clockwise or counterclockwise. It’s not literally spinning like a top, it’s a fundamental property, but thinking of it that way helps. So, ms can be either +1/2 or -1/2, representing these two opposite spin directions. Think of it as an electron saying, "Up yours!" or "Down with this!" It's like the final detail that makes each electron unique. And get this, two electrons can share the same orbital (same n, l, and ml) only if they have opposite spins. This is the famous Pauli Exclusion Principle, the ultimate "no shirt, no shoes, no service" rule of the atomic world. No two electrons can have the exact same set of all four quantum numbers.
Okay, deep breaths. We've covered n, l, ml, and ms. Now, you asked about '3p'. This is where the magic happens, where the shorthand of chemistry and physics comes into play. The '3' in '3p' is our principal quantum number (n). So, n = 3. Simple enough, right? That means we’re talking about electrons that are in the third energy level, further out from the nucleus, with more energy than those in the first or second levels. They’re like the residents of the third floor of our atomic building.

The 'p' in '3p' refers to the angular momentum quantum number (l). In the quantum world, different values of 'l' are represented by letters. When l = 0, it's an 's' orbital (think of a spherical room). When l = 1, it's a 'p' orbital (think of a dumbbell-shaped room). When l = 2, it's a 'd' orbital (think of a more complex shape, like a four-leaf clover). And when l = 3, it's an 'f' orbital (which gets really wild, like a multidimensional origami project). So, for 'p', we know that l = 1.
So, when someone says '3p', they're essentially saying: "We're looking at electrons in the third energy level (n=3) that are in p-orbitals (l=1)." It’s a descriptor, a label, a coded message from the quantum universe. It’s not a single numerical value for '3p' itself, but rather a description of the state of an electron. It's like saying "a luxury apartment on the third floor." You wouldn't give "luxury apartment on the third floor" a single number; it's a description.
Now, if we’re talking about the possible values associated with this '3p' description, we need to consider the other quantum numbers. For n=3 and l=1, what are the possible values for ml? Remember, ml tells us the orientation of the p-orbital. Since l=1, ml can take on integer values from -l to +l. So, for l=1, ml can be -1, 0, or +1. That means there are three different 'p' orbitals in the third energy level, each with a different spatial orientation. They're like three distinct rooms on the third floor, all shaped like dumbbells.
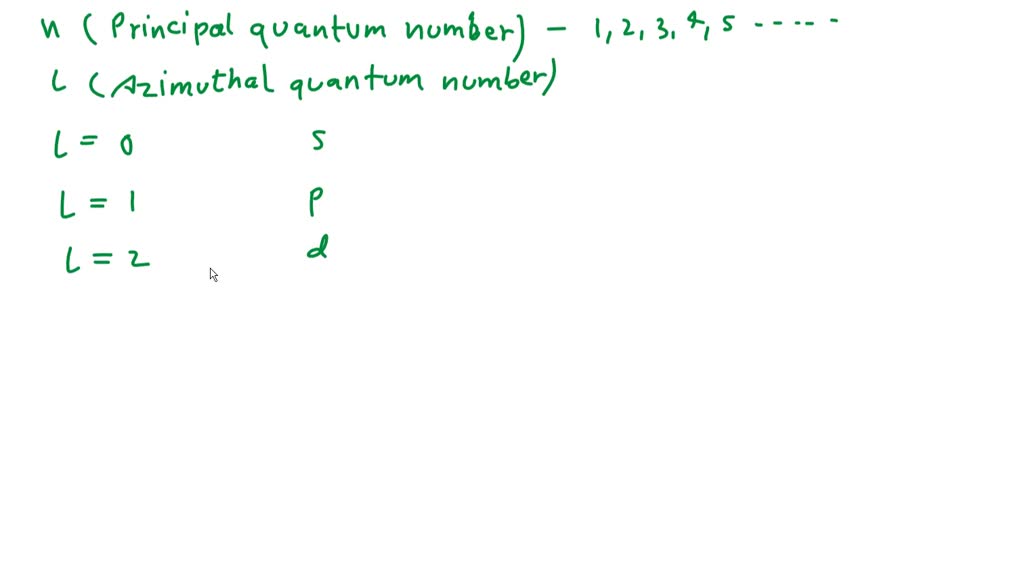
And within each of those ml orbitals, we can have two electrons, each with a different spin. So, for each of the ml values (-1, 0, +1), we can have an electron with ms = +1/2 and another with ms = -1/2. This means that in the '3p' subshell (which comprises all the orbitals where n=3 and l=1), there’s a maximum of 2 electrons * 3 orbitals = 6 electrons. So, the '3p' description actually encompasses a whole group of possibilities, not a single number. It's like asking for the numerical value of "all the red cars in a parking lot." There are many red cars, each with its own number of wheels and horsepower, but they all share the characteristic of being red.
So, to circle back to your question, "Give the numerical value of N corresponding to 3p." The '3' in '3p' is the numerical value of the principal quantum number, n = 3. The 'p' tells us about the shape, corresponding to l = 1. There isn't a single number that is '3p' itself. It’s more like a label for a set of states. Think of it like this: if you have a password that's "secret_agent_007", the '007' is the numerical part. In '3p', the '3' is the numerical part for the energy level.
It’s like the universe has its own secret code, and '3p' is a little snippet of that code. It’s not a price tag, it’s a description of where an electron is chilling in its atomic neighborhood. And knowing this '3p' helps us understand everything from how atoms bond to form molecules (which make up you, me, and that questionable burrito you had for lunch) to how stars shine. So next time you hear '3p', don't think of a sad, three-legged penguin. Think of the electrifying, yet incredibly ordered, dance of particles that make up reality itself. And maybe, just maybe, you'll feel a little bit like a cosmic detective yourself. Now, who wants a refill?
