Give The Full Electron Configuration For Sulfur

Hey there, fellow curious minds! Ever wondered what goes on inside the tiny, invisible world of atoms? It's pretty wild, right? Today, we're going to peek at a particular element, sulfur, and unravel its full electron configuration. Don't let the fancy words scare you off – it's actually a super neat puzzle to solve, and once you get the hang of it, you'll see atoms in a whole new light.
So, what's this "electron configuration" thing all about? Think of an atom like a tiny apartment building. The nucleus is the ground floor, where all the heavy stuff is. And then you've got these "floors" or "shells" where the electrons, the much lighter residents, hang out. Electron configuration is basically the address system for these electrons – telling us exactly which "apartment" on which "floor" each electron calls home.
Let's Talk Sulfur!
Sulfur! You probably know it as that stuff that smells like rotten eggs. Yep, that's our guy. But sulfur is way more than just a pungent odor. It's a super important element that's found in everything from volcanic gases to the proteins in your own body. Pretty cool, huh?
When we talk about the full electron configuration for sulfur, we're essentially listing out where all of its electrons are located within its atomic "apartment building." And to do that, we first need to know how many electrons sulfur actually has. This is where the atomic number comes in.
The Atomic Number: Our Starting Point
Every element has a unique atomic number, which is basically the count of protons in its nucleus. For a neutral atom, the number of electrons is the same as the number of protons. Sulfur's atomic number is 16. So, a neutral sulfur atom has 16 electrons zipping around its nucleus.
Now, these electrons aren't just randomly scattered. They follow some pretty strict rules, filling up specific "rooms" or "orbitals" in a predictable order. These orbitals have different shapes and energy levels, like different types of rooms in our apartment building.
The Orbital Dance: s, p, and d
We have different types of orbitals, and they're often described by letters: 's', 'p', and 'd' (and 'f', but we won't need that for sulfur). Think of 's' orbitals as cozy little studios – they can hold a maximum of 2 electrons. 'p' orbitals are like slightly larger one-bedroom apartments – they can house up to 6 electrons. And 'd' orbitals are like spacious three-bedroom apartments – they can fit up to 10 electrons.

These orbitals are also organized into energy levels, or "shells." Shell 1 is the closest to the nucleus and has the lowest energy. Shell 2 is a bit further out and has more energy, and so on. Electrons, being the organized critters they are, tend to fill up the lowest energy levels first, just like we'd fill the ground floor apartments before heading upstairs.
Filling Up the Shells for Sulfur
Let's start filling those orbitals for our 16 sulfur electrons. We'll go in order of increasing energy.
Shell 1: The Ground Floor
The first shell only has one type of orbital: the 's' orbital. It's super small and cozy, and can hold a maximum of 2 electrons. So, the first 2 electrons of sulfur go into the 1s orbital. We write this as 1s2. That little superscript '2' means there are 2 electrons in that orbital.
We've now placed 2 out of our 16 electrons. We've got 14 more to go!
Shell 2: The First Floor Up
Moving up to the second shell, we have a 's' orbital and three 'p' orbitals. Remember, the 's' orbital can hold 2 electrons, and the 'p' orbitals can hold up to 6 electrons each. So, the second shell can hold a total of 2 + 6 = 8 electrons.
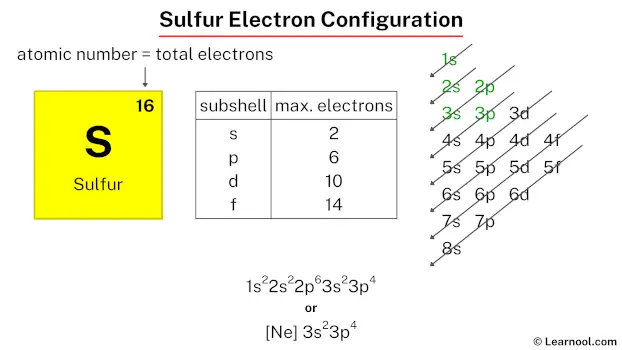
First, we fill the 's' orbital in this shell. So, we have the 2s orbital, which will take 2 electrons. That brings our total to 2 (from shell 1) + 2 (from the 2s orbital) = 4 electrons placed.
We still have 12 electrons left. Next, we fill the 'p' orbitals in the second shell. There are three 'p' orbitals, and each can hold 2 electrons, for a total of 6 electrons. So, the 2p orbitals get filled with 6 electrons. We write this as 2p6.
Now, let's add them up: 2 (from 1s) + 2 (from 2s) + 6 (from 2p) = 10 electrons placed. We're getting closer! We have 6 more electrons to account for.
Shell 3: The Second Floor Up
On to the third shell! This shell is a bit more crowded. It has an 's' orbital, three 'p' orbitals, and even five 'd' orbitals. However, electrons like to fill orbitals in order of energy, and it turns out that for sulfur, the '3d' orbitals are higher in energy than the '4s' orbital (which is a bit of a quirk of atomic structure, but that's for another chat!). So, we'll fill the 's' and 'p' orbitals in the third shell before we even think about the 'd' orbitals.

First, we fill the 3s orbital with 2 electrons. That brings our total electrons placed to 10 + 2 = 12 electrons.
We have 4 more electrons to go. These will fill the 3p orbitals. Since the 3p orbitals can hold up to 6 electrons, we'll put our remaining 4 electrons there. We write this as 3p4.
And there you have it! We've placed all 16 electrons.
Putting It All Together: The Full Electron Configuration
So, let's string all those orbital fillings together in order. The full electron configuration for sulfur is:
1s2 2s2 2p6 3s2 3p4

Pretty neat, right? It's like a step-by-step guide to where every single one of sulfur's 16 electrons is chilling. Each part of that string tells us about a specific "room" and how many electrons are in it.
Why Is This Even Cool?
You might be thinking, "Okay, but why should I care about where electrons are?" Well, it's because these little guys are the real puppet masters of chemistry! The way electrons are arranged dictates how an atom will interact with other atoms. It determines whether sulfur will bond with other elements, what kind of compounds it will form, and ultimately, why it behaves the way it does.
Think of it like this: the electron configuration is the personality of the atom. A sulfur atom with this configuration is going to react differently than, say, an oxygen atom or a neon atom. It's all about those outer electrons, the ones in the highest energy levels, because they're the ones most likely to get involved in bonding.
For sulfur, those 3p4 electrons are its valence electrons – its "social" electrons, if you will. They're looking for partners to form stable bonds. This is why sulfur is so versatile and can be found in so many different chemical compounds. It's got that perfect little setup to make connections!
So, next time you encounter something with sulfur in it, whether it's the eggy smell of natural gas or a vital protein in your body, you can impress yourself (and maybe a friend!) by knowing a little bit about the secret lives of its electrons. It’s a tiny glimpse into the incredible, intricate dance of the universe, one electron configuration at a time!
