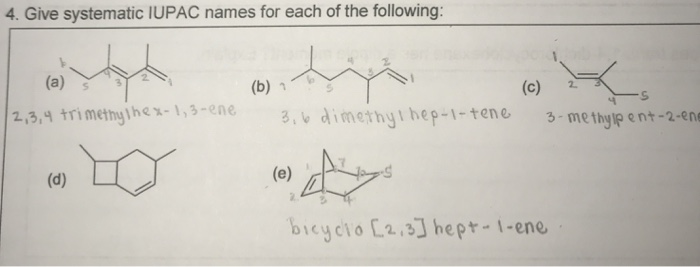
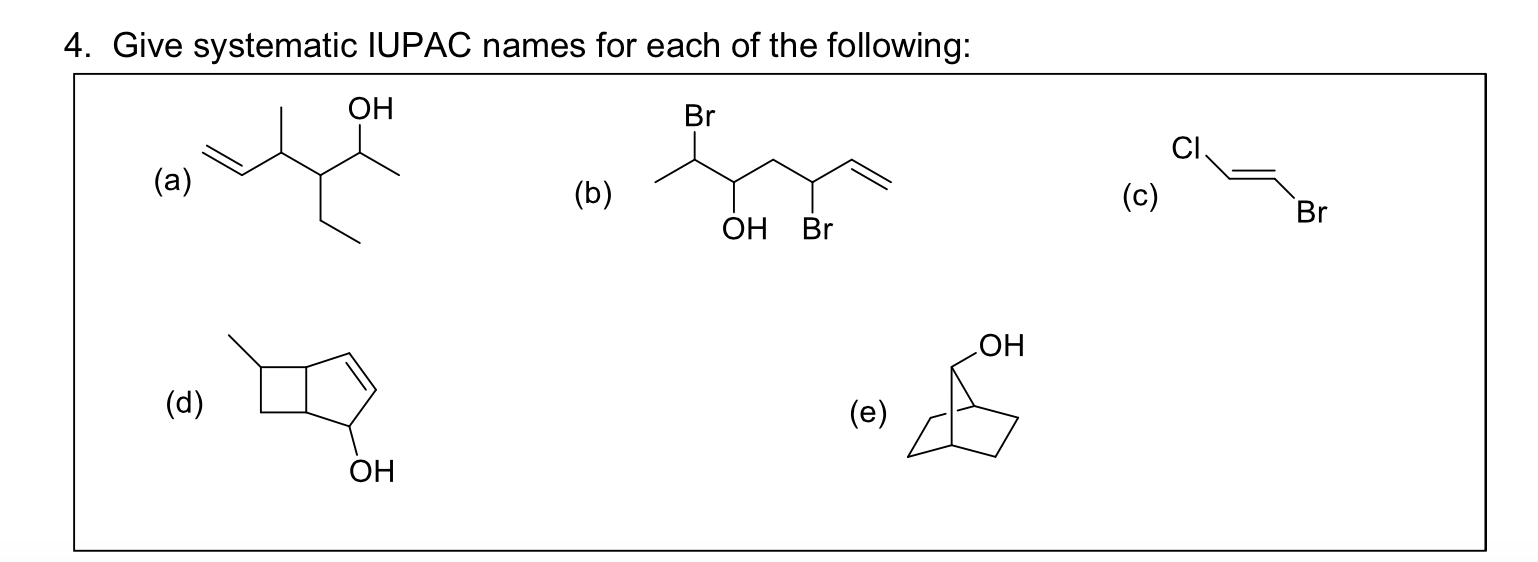
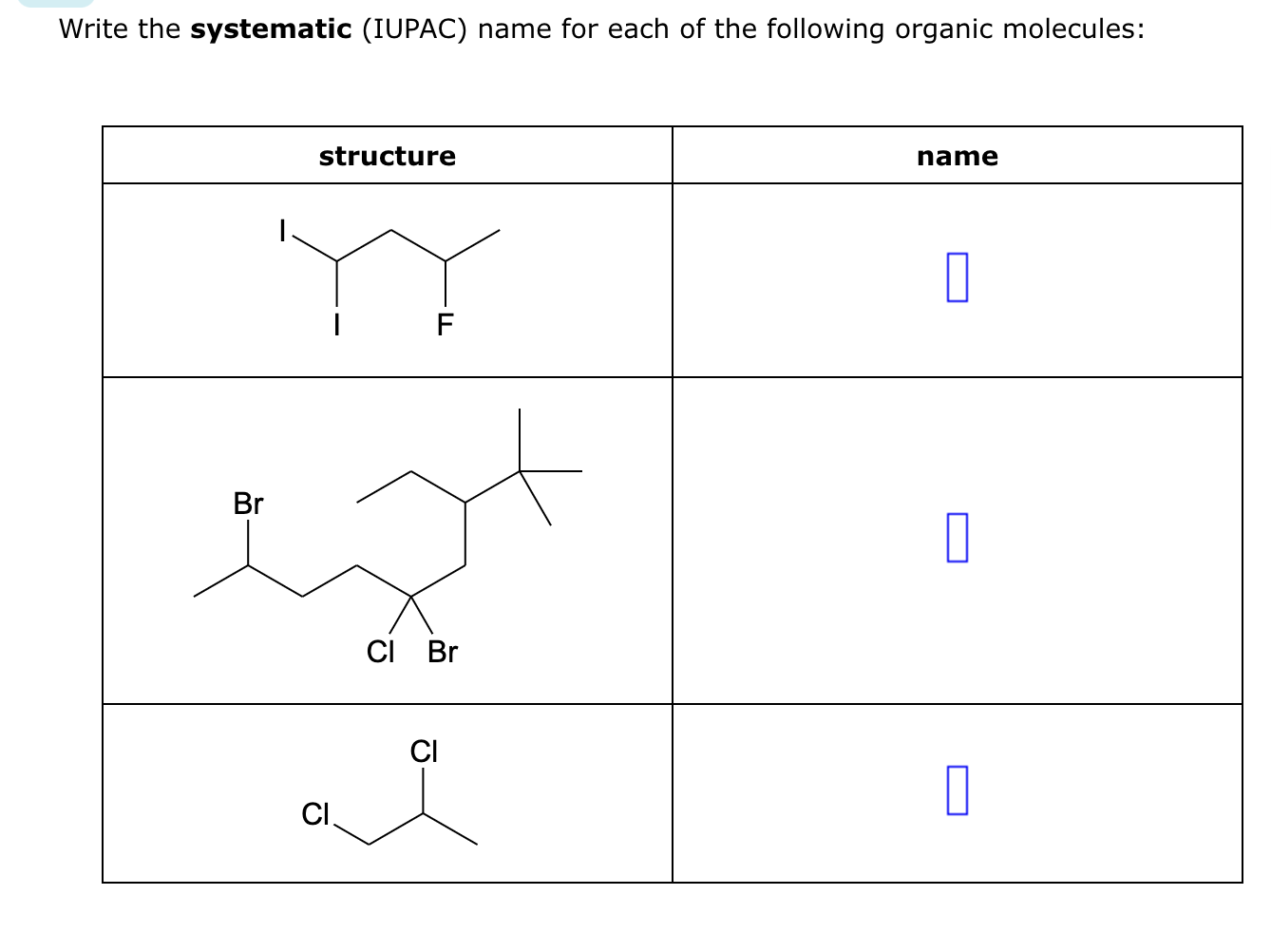
Give Systematic Iupac Names For Each Of The Following

Alright, my chemistry adventurers! Ever feel like you’re staring at a chemical name and it’s just a jumbled mess of letters and numbers? Like trying to decipher an alien language after a particularly loud concert? Well, fear not! Today, we’re going to tame those wild, unruly chemical names and give them some proper, official (and surprisingly fun!) IUPAC monikers. Think of IUPAC (that's the International Union of Pure and Applied Chemistry, by the way – a bunch of super smart people who decided to bring order to the chemical chaos) as the ultimate naming convention guru. They’ve got this system that’s like a secret decoder ring for molecules. And honestly, once you get the hang of it, it’s more satisfying than finally finding that missing sock!
Let’s dive into the wonderful world of naming these little building blocks of everything around us. It's like giving nicknames, but with a whole lot more precision and a touch of scientific flair. Forget those common names that are sometimes as confusing as a politician’s promise. IUPAC is here to give us clarity, and let me tell you, clarity is beautiful!
First up, we have a molecule that’s basically the lifeblood of… well, life! It’s got a super simple structure, like a tiny, happy chain. We’re talking about something that’s a fundamental building block for so many things. When you look at it, you see a core of carbon atoms, all lined up like little soldiers, and then some hydrogens chilling around them. The IUPAC name for this straightforward fellow is incredibly fitting. It’s so fundamental, it’s almost like it deserves a crown! The name we’re bestowing upon this essential chemical is, drumroll please… Methane!
Yep, that’s right. Methane. Simple, classic, and gets the job done. It’s the smallest of its kind, a real minimalist when it comes to carbon chains. Think of it as the tiny seed from which larger, more complex molecular plants can grow. It’s like the number "1" in the world of numbers – foundational and utterly essential.
Now, let’s crank it up a notch. Imagine our little methane friend found a buddy. Now we have two carbon atoms linked together, with hydrogens happily attached all around. It’s like our previous molecule decided to go on a little road trip with a twin. This slightly longer molecule is still pretty simple, but it's a step up in complexity. It's got a bit more personality, a bit more… presence. And its IUPAC name? It’s equally as straightforward, reflecting its two-carbon structure. Prepare yourselves for the grand reveal: it’s called Ethane!
Ethane! See? Not so scary, right? It’s like upgrading from a solo act to a dynamic duo. This molecule is a bit more substantial, a bit more robust. It’s still a fundamental part of many organic processes, like a reliable workhorse in the molecular world. It’s the dependable friend who’s always there.
Moving on, let's picture another variation. This time, we’re looking at three carbon atoms all linked up in a nice, neat row. Imagine a tiny, perfectly aligned train. This molecule is getting a little more substantial, a little more interesting. It's the "three's company" of the simple alkane world. And its official IUPAC title? It’s a name that just rolls off the tongue, a testament to its clean structure. Brace yourselves for the elegance of… Propane!
Propane! So good, so clean, so… propane-y! This is the stuff you might find in your barbecue grill, ready to bring the heat to your summer cookouts. It’s a fantastic example of how these seemingly complex names are actually just descriptive. It’s like saying, “Hey, this thing has three carbons in a row, so we’ll call it propane!” No mystery, just pure, unadulterated chemical logic. It’s the backbone of many everyday conveniences, and its name is as straightforward as its function.
Let’s keep this naming party going! What if we have four carbon atoms all lined up, like a perfectly formed row of dominoes just waiting to be tipped? This molecule is getting a bit longer, a bit more… substantial. It’s no longer the shy beginner; it’s a confident participant in the molecular dance. Its IUPAC name perfectly captures its four-carbon essence. Get ready for the… Butane!
Butane! Think of the lighter you use to ignite a candle – chances are, it’s powered by butane! This is a molecule that’s become a staple in our lives, and its name is as familiar as its utility. It’s the workhorse of the four-carbon straight chains, a reliable performer in countless applications. It’s the sturdy, dependable choice, and its name is a simple reflection of its structure.
And there you have it! Just a little taste of the delightful simplicity that IUPAC brings to the chemical world. These names aren't meant to intimidate; they're designed to inform and impress. So next time you see a chemical formula, remember the fun of naming! It’s like giving your molecules the perfect, official, and entirely understandable nicknames. Embrace the names, understand the structures, and feel the pure joy of chemical clarity!
