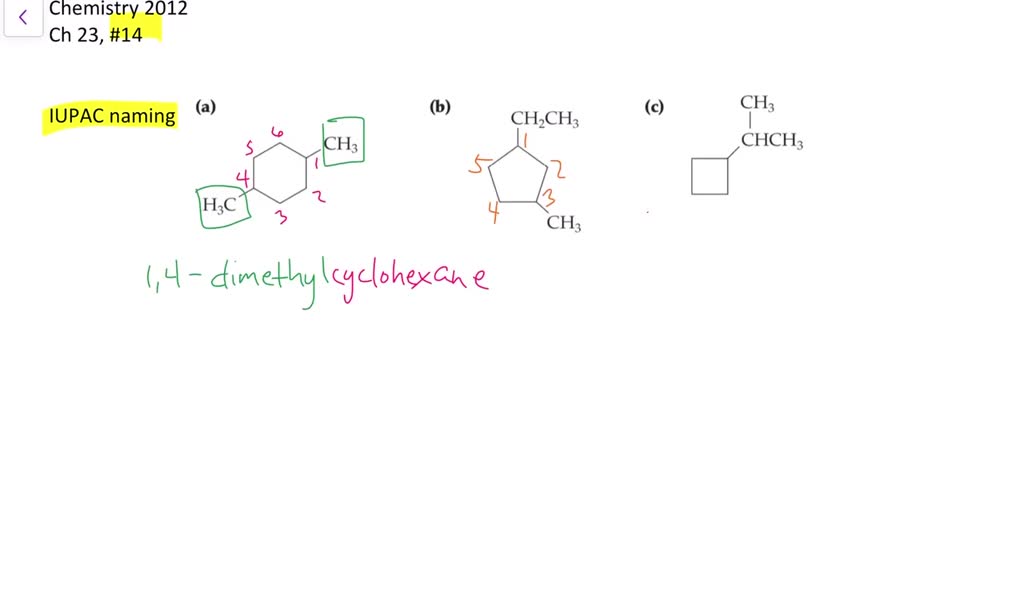
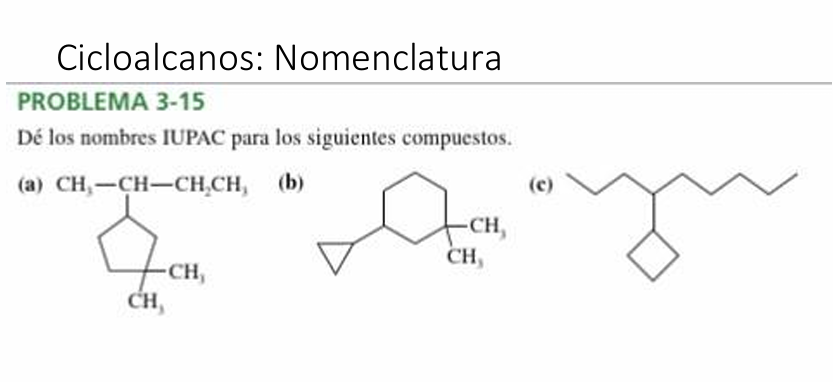
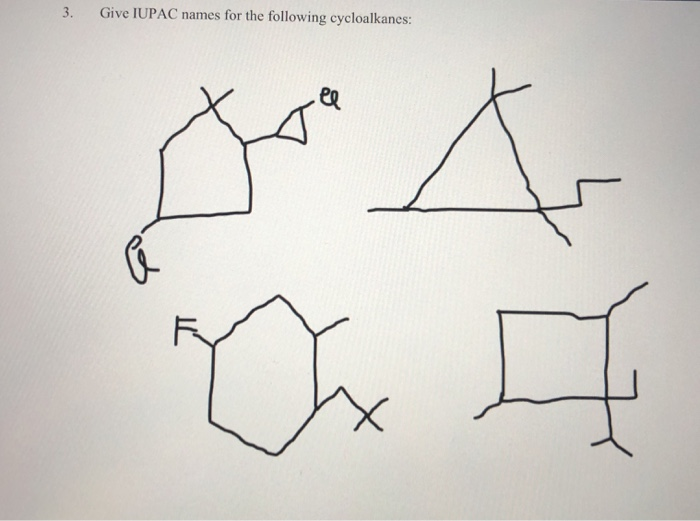
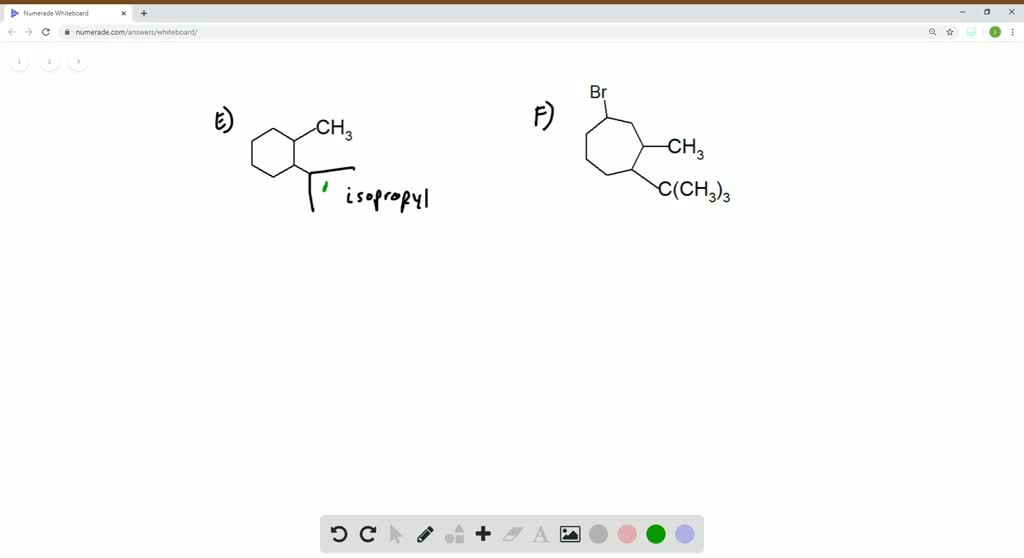
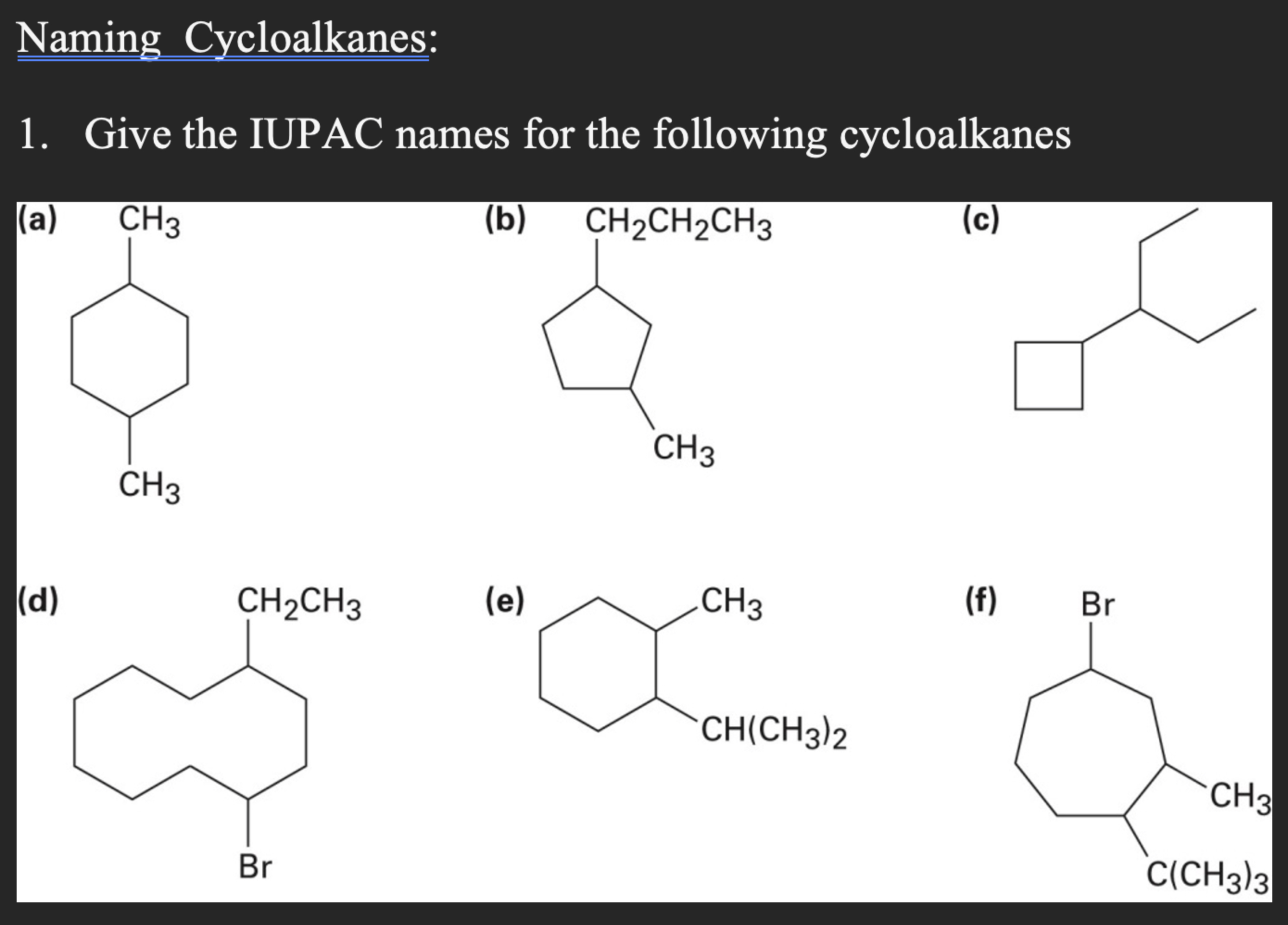
Give Iupac Names For The Following Cycloalkanes
Alright, fellow Earthlings, let's dive into the whimsical world of naming things. You know, the kind of naming that makes you scratch your head and wonder if someone just spilled alphabet soup onto a chemistry textbook. We're talking about cycloalkanes today, which sound super fancy, like a secret society of bike enthusiasts or maybe a type of fancy cheese.
But don't worry, it's not all serious business. Think of it as a friendly game of "Guess the Weird Name." We've got some innocent-looking rings of carbon atoms, just chilling, and our job is to give them their proper, albeit sometimes baffling, IUPAC names.
So, buckle up your metaphorical seatbelts, because we're about to embark on a naming adventure. Prepare for some chuckles, maybe a few groans, and definitely an appreciation for the sheer, unadulterated joy of making things sound complicated.
Let's Start with the Basics!
Imagine a simple ring. Just a few carbon atoms holding hands in a circle. Pretty straightforward, right? Well, in the grand scheme of IUPAC nomenclature, even the simplest things get a title. Think of it as giving your pet rock a formal designation. "Rock McStoneface, Esquire."
Our first little ring is the smallest one we can make. It’s got three carbon atoms, all huddled together like a tiny, geometric family reunion. It’s a triangle, if you will, but a carbon triangle. And its IUPAC name? Drumroll, please…
The Tiny Triangle of Terror (Not Really)
This little fella is called cyclopropane. See? It’s not so bad! "Cyclo" because it’s a circle, and "propane" because it has three carbons, like propane gas you use for your BBQ. Simple, right? Almost too simple. It’s like calling a cat "Meow."
But here’s where it gets fun. What if this little cyclopropane decides to spice things up? What if it decides to hang a party decoration on one of its carbon atoms? Let’s say it’s a tiny little methane molecule, just chilling there.
When Rings Get Companions
Now, this is where IUPAC’s love for order really kicks in. We’ve got our cyclopropane ring, and then we’ve got our little accessory. We need to decide which part is more important. It’s like choosing between the wedding cake and the bride. (Spoiler: the ring is usually the bride in these situations).
So, we have methylcyclopropane. "Methyl" is what we call a single carbon group hanging off something. And "cyclopropane" is our trusty ring. It’s like saying "Fluffy with a Hat." Adorable, and very specific.
What if our cyclopropane decides to go all out and have two party decorations? This is where things could get chaotic. But IUPAC has a system, bless its organized heart.
Double the Fun, Double the Names
If we have two methyl groups on our cyclopropane, we need to tell everyone where they are. We use numbers! Imagine numbers on a clock face, guiding us around the ring. We start numbering at one of the carbons with a methyl group, and then we count around.
If both methyls are on the same carbon, it's easy-peasy. We just say 1,1-dimethylcyclopropane. It’s like saying "The house with two doorknobs on the front door." Very descriptive. And slightly peculiar.
But what if they’re on different carbons? This is where the real naming prowess of IUPAC shines. If they are on adjacent carbons, we get 1,2-dimethylcyclopropane. It’s like saying "The house with doorknobs on the first and second steps." Still a house, still doorknobs, just strategically placed.
And if they’re on opposite sides of the tiny triangle? You guessed it: 1,3-dimethylcyclopropane. "The house with doorknobs on the first and third steps." You can practically picture the architect’s meticulous notes.
Moving Up to Bigger Rings
Okay, the three-membered ring is a bit intense. Let’s move on to something a bit more… chill. A four-membered ring. This one looks like a square, or maybe a slightly lopsided picture frame. It’s got four carbon atoms in a loop.
The Square Deal
This four-membered wonder is called cyclobutane. "Cyclo" for the circle, "butane" because it has four carbons. It’s like calling a dog "Woof." Basic, but effective.
Now, let’s add some flair to our cyclobutane. What if we put a little ethyl group (that's two carbons) on one of the corners? Just a simple dangling chain.
We’d get ethylcyclobutane. It's like saying "Squarey with a scarf." It just adds a little something extra. And IUPAC, ever the stylist, has a name for it.
What if we have two ethyl groups? Oh boy, here we go again with the numbering! If they’re on the same carbon, it’s 1,1-diethylcyclobutane. If they’re on adjacent carbons, it’s 1,2-diethylcyclobutane. And if they’re across from each other, it’s 1,3-diethylcyclobutane.
It’s like ordering a very specific kind of sandwich. "Yes, I'll have the sourdough, with two ham slices on the first layer, and two cheese slices on the second." Everyone knows exactly what you're getting.
The Six-Membered Superstar
Now, let’s talk about the king of the simple rings. The one you’ll see everywhere. It's a six-membered ring, looking like a perfectly formed hexagon. It’s the supermodel of the cycloalkane world.
The Classic Hexagon
This magnificent creature is called cyclohexane. You’ve probably heard of it, or at least its cousins in the world of everyday chemicals. "Cyclo" for the circle, "hexane" for the six carbons. It's elegant, it's stable, it's the darling of organic chemistry.
Imagine giving cyclohexane a little makeover. What if we attach a chlorine atom to one of the corners? Chlorine sounds a bit sinister, but in chemistry, it's just another atom looking for a friend.
We get chlorocyclohexane. It's like saying "Hexagon with a tiny superhero cape." It’s all about the accessories.
What if we get really wild and put a whole bunch of methyl groups on it? Let’s say we have three methyl groups. IUPAC, in its infinite wisdom, has us numbering the ring and specifying the positions.
We could have 1,2,3-trimethylcyclohexane. Or maybe 1,3,5-trimethylcyclohexane. It’s like describing a very intricate knitting pattern. Row 1, knit 2 purl 1, row 2, purl 3…
Why So Many Names?
Now, you might be thinking, "Why all the fuss? Can't we just call them 'ringy things' and be done with it?" And to that, I say, "You and I would get along famously." But alas, scientists need to be precise.
These detailed names ensure that when you say "1,1,2-trimethylcyclopropane," everyone, everywhere, knows exactly which specific, quirky little molecule you’re talking about. No room for misinterpretation, even if the name itself sounds like a tongue twister invented by a mischievous gnome.
It’s like having a secret handshake for every single carbon ring out there. A unique identifier that tells its story. And honestly, while it can be a bit much, there’s a certain charm to it. A knowing wink from the universe of molecules.
So, the next time you see a chemical name that looks like it was generated by a random word generator, take a moment to appreciate the effort. It’s a testament to our human desire to categorize, to understand, and to, of course, give everything a ridiculously long and specific name. Cheers to cycloalkanes, and the delightful chaos of their IUPAC monikers!
