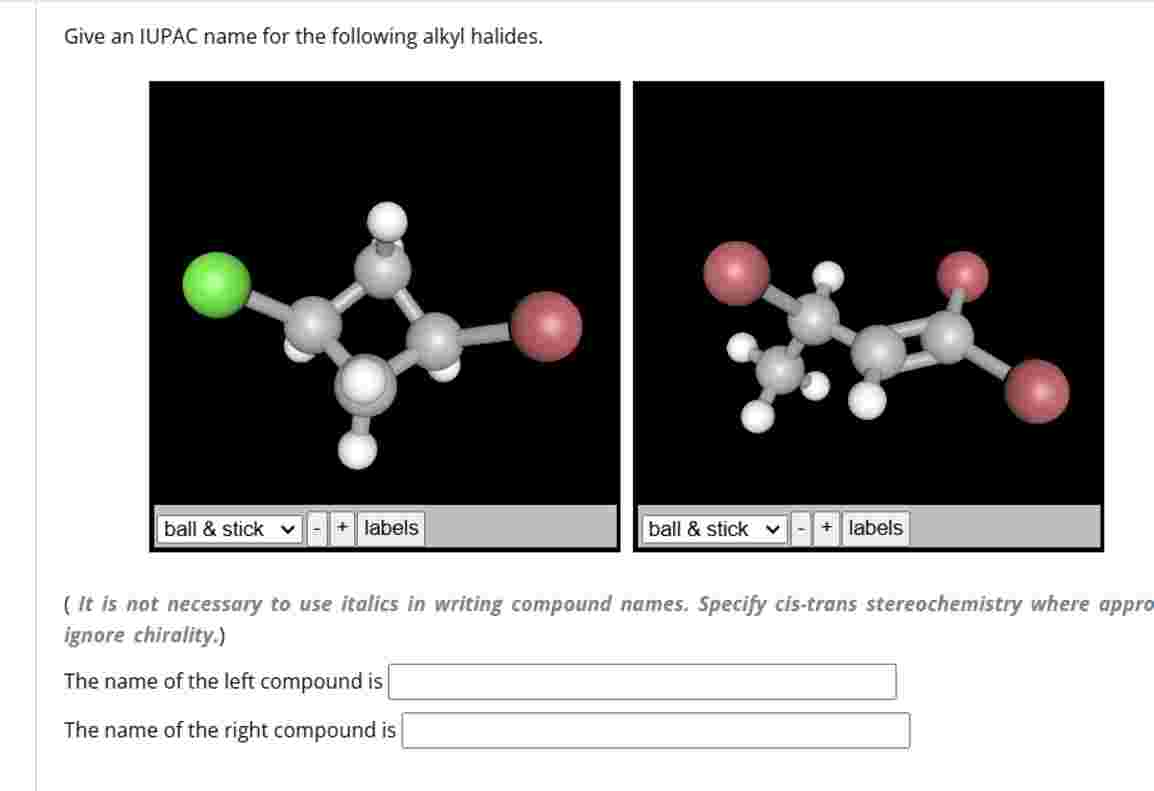
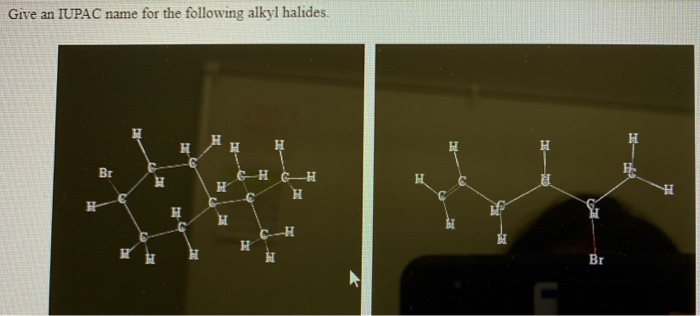
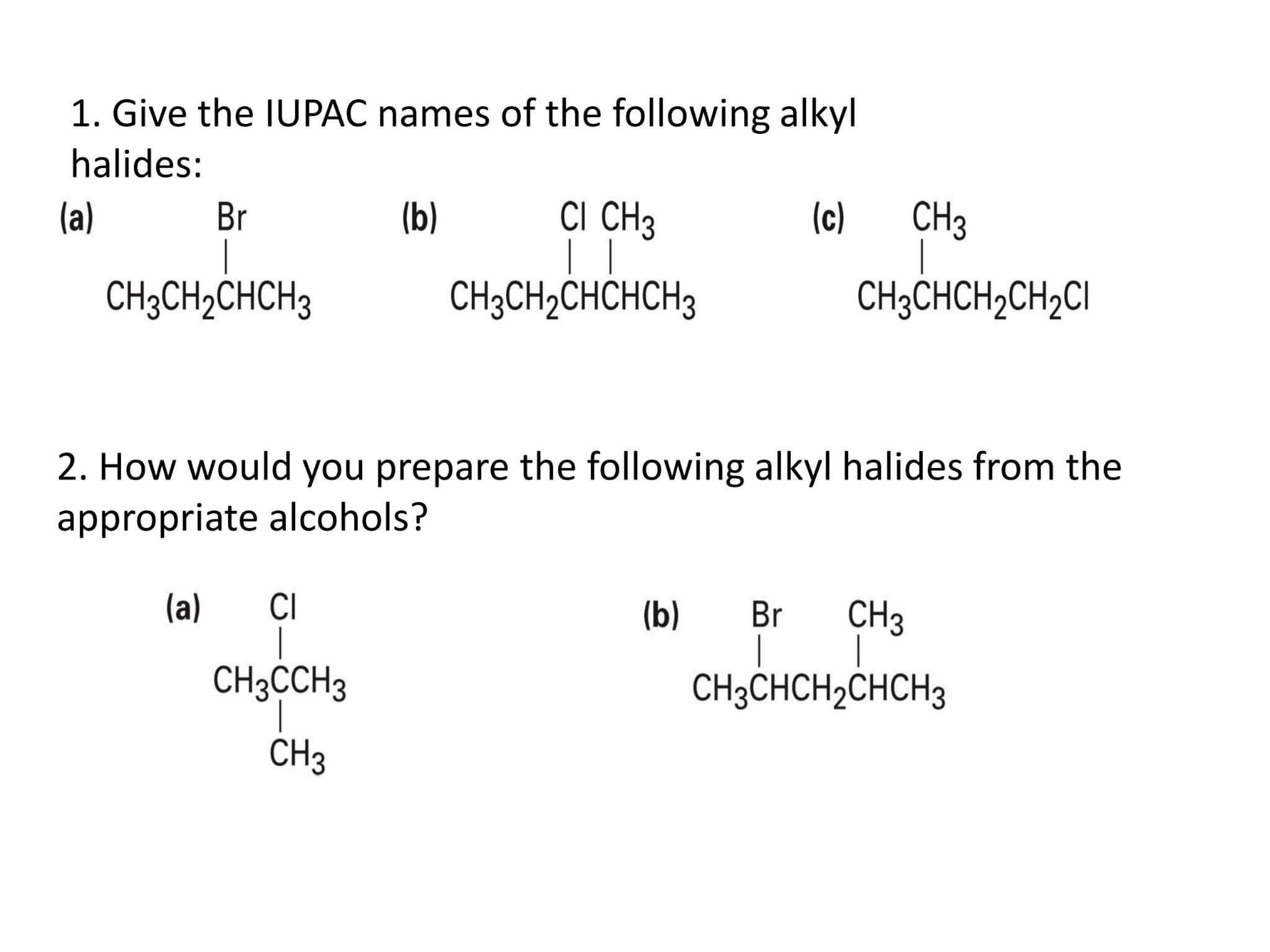
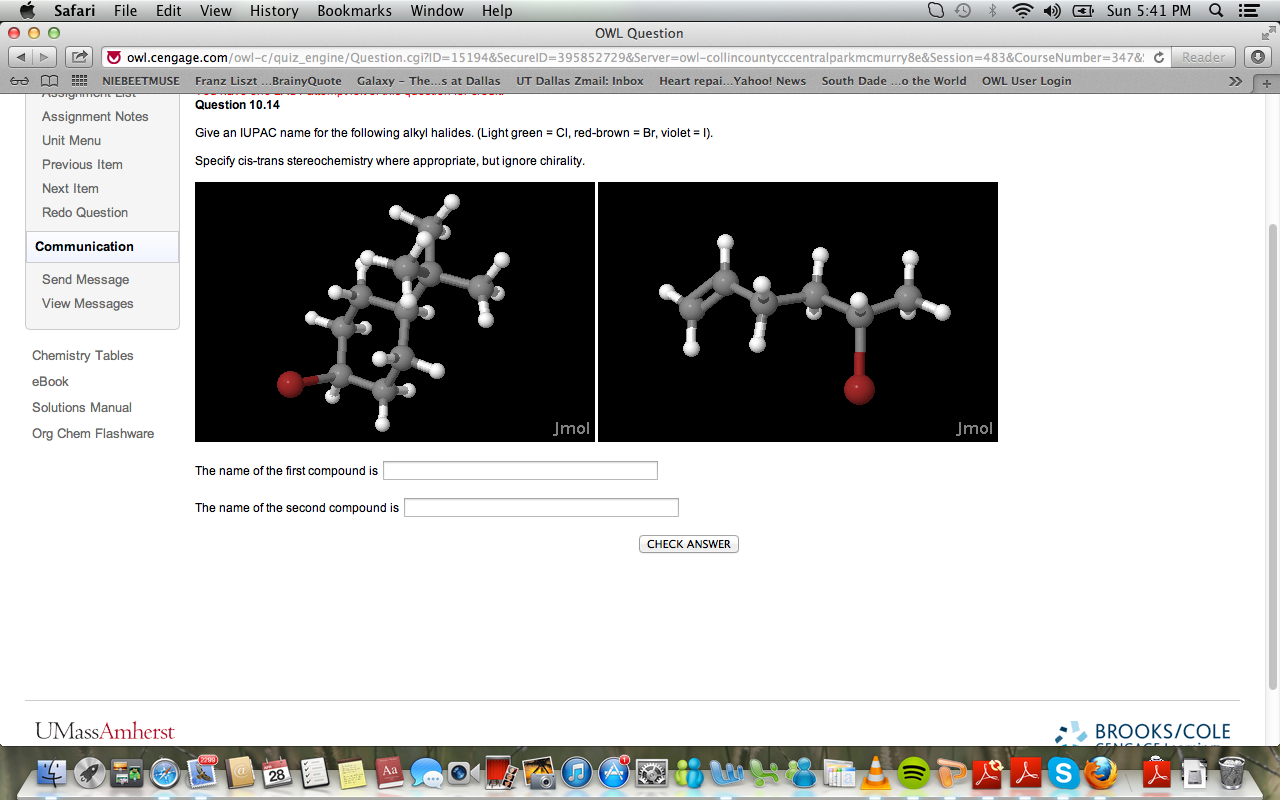
Give An Iupac Name For The Following Alkyl Halides
Hey there, science curious folks! Ever looked at those wacky-sounding chemical names and thought, "What in the world is that all about?" Well, buckle up, because we're about to dive into the super fun, surprisingly logical, and totally captivating world of naming things in chemistry. It's like a secret code, but once you crack it, everything makes sense!
Today, we're focusing on a special group of molecules called alkyl halides. Think of them as the rockstars of the chemical world. They've got a carbon chain, which is like their stage, and then a halogen atom, which is their flashy accessory. These halogens are fluorine, chlorine, bromine, and iodine – kind of like the elemental glitter and glam of organic chemistry!
But here's the really cool part: they all have official, super-organized nicknames. These aren't just random jumbles of letters. Nope, these are IUPAC names. IUPAC stands for the International Union of Pure and Applied Chemistry. Imagine them as the ultimate naming committee for all things chemical. They've got rules, they've got systems, and they make sure everyone, everywhere, is talking about the same molecule when they use its official name.
Why is this so entertaining? Because it’s like a puzzle! You're given a chemical structure, and your job is to figure out its proper, official title. It’s like being a detective, but instead of solving crimes, you’re solving chemical identities. And trust me, there's a certain thrill in deciphering these codes.
So, how do we get these awesome names? It’s all about following a few simple, yet powerful, rules. The first step is to find the longest continuous chain of carbon atoms. This is your parent chain, the foundation of the molecule. Think of it as the main stage where all the action happens. The longer the chain, the more impressive the molecule!
Once you’ve identified that longest carbon chain, you need to count the carbon atoms in it. This counting gives you the base name. For example, if you have a chain of two carbons, it's called ethane. Three carbons? That's propane. Four? That’s butane. It’s like learning a new language, but this one is super useful for understanding the building blocks of pretty much everything around you.
Now, where do those flashy halogen accessories come in? They get attached to the carbon chain. And they get their own little prefixes. If you've got a fluorine atom, it becomes fluoro. Chlorine? It's chloro. Bromine becomes bromo, and iodine is iodo. See? It’s starting to sound a little like a catchy song, isn't it?
But here’s where the detective work really kicks in. You need to tell the IUPAC committee where these halogen accessories are located on the carbon chain. So, you number the carbon atoms in your parent chain. The trick is, you want to give the halogen the lowest possible number. This is like giving your superstar a front-row seat on the stage – it’s the most important spot!
Let’s imagine a simple case. You have a two-carbon chain (ethane) and a chlorine atom attached to one of the carbons. If you number the carbons from left to right, the chlorine might be on carbon number 1. If you number from right to left, it might also be on carbon number 1. So, the name would be 1-chloroethane. It's precise, it's clear, and everyone knows exactly what you're talking about.
What if you have a longer chain, like three carbons (propane), and a bromine atom? If the bromine is on the middle carbon, no matter which way you number, it'll be on carbon number 2. So, you'd call it 2-bromopropane. It’s like giving directions: “Go down the main road, turn at the second house, and you’ll find it!”
But what if there are multiple halogen accessories? This is where things get even more exciting! If you have two chlorine atoms, you use the prefix di-. Three chlorines? That's tri-. And so on, with tetra-, penta-, and so forth. You just stack them up like building blocks!
And you have to specify the location of each halogen. So, if you have a four-carbon chain (butane) with two chlorine atoms, and one is on carbon 1 and the other is on carbon 2, you'd have to list both numbers. The name would become 1,2-dichlorobutane. It’s like a meticulously organized inventory.

The order in which you list the halogens also matters, especially if you have different types. You list them in alphabetical order. So, if you have a chlorine and a bromine on the same molecule, the bromo part comes before the chloro part in the name. It's a bit like lining up your rockstars for a photo op – everyone in their designated spot.
Let's take a slightly more complex example to really get the adrenaline pumping. Imagine a six-carbon chain (hexane). And let’s say you have a chlorine atom on carbon number 2 and a bromine atom on carbon number 4. Following the rules, you find the longest chain (hexane). Then you identify your halogens: chloro and bromo. Alphabetically, bromo comes first. Then you check the numbering to ensure the lowest possible numbers. In this case, numbering from one end gives you positions 2 and 4. Numbering from the other end would give you positions 3 and 5, which are higher numbers. So, you stick with the 2 and 4. The IUPAC name? 4-bromo-2-chlorohexane.
Isn't that fascinating? It’s like solving a miniature chemical riddle. Every part of the name tells you something specific about the molecule's structure. You can literally draw the molecule just by reading its IUPAC name. That’s the power of this system! It’s a universal language for chemists, ensuring that when they talk about, say, 1,1-difluoroethane, everyone knows they’re discussing a very specific compound.
And this isn’t just abstract stuff. These alkyl halides are everywhere! They are used in making plastics, refrigerants, solvents, and even some medicines. Understanding their names is like getting a backstage pass to how so much of our modern world is made.
The beauty of IUPAC naming for alkyl halides is its elegance and logic. There are no exceptions, no quirks, just a systematic approach that works every time. It’s like learning to play a musical instrument; at first, it might seem a bit daunting, but with practice, you start to see the patterns and can create beautiful melodies.
So, the next time you encounter a chemical name that looks like a tongue twister, remember the IUPAC system. It’s there to guide you, to demystify the molecule, and to reveal its hidden structure. It’s a rewarding journey, full of discovery, and it makes chemistry feel a lot less intimidating and a whole lot more like an exciting adventure.
It’s a system that brings order to the vast universe of molecules. And in that order, there’s a special kind of beauty and satisfaction. So go ahead, give it a try! See if you can name some of these chemical rockstars. You might just discover a hidden talent for chemical nomenclature and find yourself utterly captivated by the organized world of IUPAC names. It’s a fun challenge, and the reward is a deeper understanding of the molecules that shape our lives. Keep exploring, keep naming, and most importantly, keep being curious!
