Give An Example That Illustrates Lipids Being Waterproof

Ever notice how some things just refuse to get wet, no matter how much you try to soak 'em? We're talking about those everyday miracles that make life a little less... soggy. Think about it. You've probably had those moments where you're staring at something, utterly baffled by its ability to repel water like it's got a personal vendetta against it.
Well, guess what? There's a whole scientific reason behind this, and it all boils down to something called lipids. Sounds fancy, right? Like something you'd only hear about in a lab coat commercial. But trust me, lipids are the unsung heroes of waterproof-ness, and you interact with them more than you realize. They're basically nature's tiny, hydrophobic superheroes, zipping around and keeping water where it doesn't belong.
Let's break it down, shall we? Imagine you've got a greasy stain on your favorite shirt. You try to wash it with just water, and what happens? Nada. Zip. Zilch. The water just beads up and rolls off, leaving the stain stubbornly clinging on. That greasy stain? It's probably made up of lipids, or at least something that behaves very similarly. And water, bless its heart, just can't get cozy with it. It's like trying to mix oil and water – they just give each other the cold shoulder.
Think of it like this: water molecules are like a bunch of super social butterflies, always wanting to link up with other water molecules. They form these little huddles, these water parties. Lipids, on the other hand, are the introverts of the molecular world. They prefer their own company and give those water butterflies the side-eye. They'd rather stick to themselves and avoid any watery mingling.
The Great Water Escape: A Duck's Tale
Now, where do we see these lipid-powered waterproof wonders in action? Oh, everywhere! My absolute favorite example, the one that makes me chuckle every time I see it, is a duck. You've seen them, right? Waddling around, diving into ponds, emerging looking like they just stepped out of a high-end spa. Not a single drop seems to have penetrated their fluffy exterior.
How do they do it? They've got a secret weapon: preen oil. Ducks have a little gland near their tail that squirts out this oily substance. And what do you think that oil is made of? You guessed it – lipids! They meticulously spread this oil all over their feathers with their beaks. It’s like they’re giving themselves a daily hydrophobic manicure.
Imagine a duck going for a swim. The water hits its feathers, and instead of getting soaked like a sad, waterlogged sponge, the water just boings right off. The feathers stay dry and fluffy, keeping the duck warm and buoyant. It's a masterclass in staying dry, and all thanks to lipids acting like tiny, invisible raincoats for every single feather.
I always picture a duck saying, "Oh, water? Nah, I'm good. Got my lipid armor on. You guys have fun with your little splash party, I'll be over here, dry and fabulous." It's genuinely impressive. And frankly, a little bit envy-inducing on a rainy Tuesday.
Our Own Built-in Waterproofing: The Skin You're In
But it's not just about the birds and the bees (or rather, the ducks and the lipids). We humans are surprisingly waterproof too, thanks to our very own lipid buddies. Our skin, this amazing barrier that keeps everything in and everything gross out, is packed with lipids.
Think about your skin after you've been in the shower for a while. Does it feel like a giant, soggy sponge? For the most part, no! Our skin cells have these lipid layers sandwiched between them, forming a pretty formidable barrier. This is called the stratum corneum, and it's like the bouncer at the club of your body, deciding who gets in and who stays out. And lipids are the bouncers' best friends, making sure the watery riff-raff don't sneak in.
This lipid barrier is crucial. It prevents us from drying out like a forgotten raisin in the desert. It stops harmful things from entering our bodies. It's working 24/7, silently and effectively, thanks to our internal lipid production. So next time you splash around and don't end up pruney after five minutes (though prolonged soaking can still get to you, of course!), give a little nod to those hardworking lipids.
It’s kind of like how your phone case keeps your phone safe from accidental drops. Your skin's lipid barrier is like a high-tech, self-healing, completely natural phone case for your entire body. Pretty neat, huh?
The Kitchen Countertop Challenge: Greasy Goodness
Let’s bring it back to the kitchen, because that’s where many of our lipid-related encounters happen. Think about cooking. What’s one of the most common ingredients we use? Oils and fats. And what are oils and fats? Bingo. Lipids!

Ever tried to clean up a greasy mess after frying up some bacon or sautéing some onions? Water alone just skates over that oily residue, leaving a shiny, stubborn film. You need something with a bit more oomph, like soap or a degreaser, to break down those lipids. That’s because water molecules are too busy holding hands with each other to bother with the oil molecules.
Imagine you’re trying to wash your hands after handling a greasy piece of fried chicken. You just run them under the tap. The water slides right off, leaving your hands still feeling… well, greasy. It's like the water is saying, "Nope, not my problem!" Then you grab some soap, lather it up, and poof – the grease is gone. That soap molecules are clever little things; they’re able to bridge the gap between the oily lipids and the watery environment, helping to wash them away.
It’s a classic illustration of the waterproof nature of lipids. They just don’t play well with water. This is why oil and water don’t mix in salad dressings unless you’ve got an emulsifier (which is like a molecular matchmaker). It's a fundamental property of lipids that we see play out every single day.
Nature's Paint Job: Waxy Wonders
Look at plants. They're constantly battling the elements, and they have their own lipid-based defenses. Many plants have a waxy coating on their leaves. This isn't just for show; it's a crucial survival mechanism.
Think about the leaves of a succulent plant, or the waxy sheen on a shiny apple. That's a lipid-based substance called cutin or waxes. This coating acts like a miniature umbrella for the leaf. It prevents the plant from drying out too quickly, especially in hot, sunny weather. It also helps to prevent water from pooling on the leaf surface, which could lead to fungal infections.

Imagine a plant’s leaf as a tiny little boat. The waxy coating is like a protective layer of varnish. When rain falls, it just beads up and rolls off, keeping the delicate leaf underneath safe and dry. Without this lipid barrier, plants would be much more vulnerable to dehydration and disease.
It’s a beautiful example of how lipids are nature’s original waterproofing agents, ensuring that life can thrive even in challenging environments. It’s like the plant is saying, "Bring on the sun, bring on the rain, I’ve got my lipid shield ready!"
The Science Behind the Shake: Why Oil and Water Don't Hug
So, what’s the actual scientific reason behind this lipid-water standoff? It all comes down to polarity. Water molecules are polar. This means they have a slight positive charge on one end and a slight negative charge on the other. Because of this, they love to stick together, forming those hydrogen bonds we talked about – like little molecular besties holding hands.
Lipid molecules, on the other hand, are generally nonpolar. They don’t have these distinct positive and negative ends. They’re more like a bunch of neutral observers at a party. Because they lack this polarity, they don’t have any strong attraction to water molecules. They’d rather hang out with other nonpolar molecules.
It’s like trying to get two people who are completely different in their social styles to become best friends. One loves big, boisterous group activities (water), and the other prefers quiet, one-on-one conversations or just chilling alone (lipids). They can coexist in the same space, but they’re not going to be sharing secrets and making plans anytime soon.

This difference in polarity is why oil and water separate. The water molecules are so busy interacting with each other that they essentially push the nonpolar lipid molecules away. The lipids, in turn, are happier clumped together, away from the water's constant molecular embrace. It’s a fundamental principle that governs so many things we see and experience.
Beyond the Obvious: Even Your Cells Get in on the Act
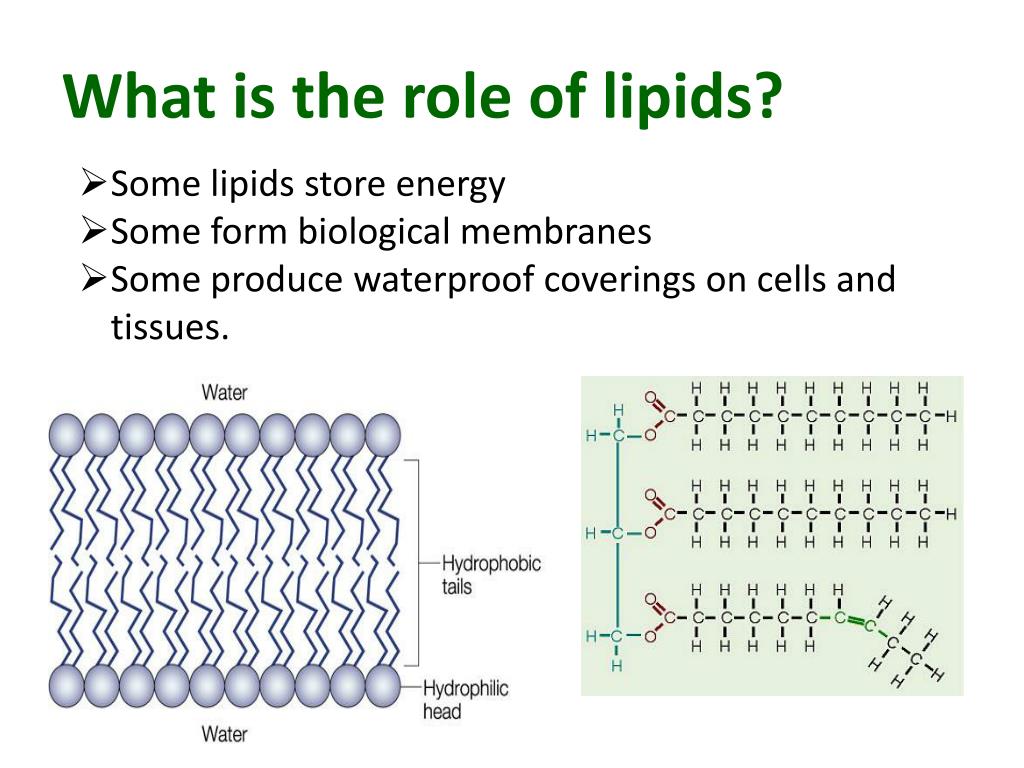
We’ve talked about ducks, skin, and cooking, but lipids are also vital for the very structure of our cells. The outer boundary of every single cell in your body is made up of something called a phospholipid bilayer. And guess what’s in a phospholipid? You got it – lipids!
These phospholipids have a bit of a dual personality. They have a head that's attracted to water (hydrophilic) and a tail that's repelled by water (hydrophobic). So, when they arrange themselves to form cell membranes, the hydrophilic heads face outwards, towards the watery environment inside and outside the cell, and the hydrophobic tails point inwards, creating a waterproof barrier in the middle.
Think of it like building a fortress. The outer walls (the hydrophilic heads) are designed to interact with the outside world, but the inner core of the walls (the hydrophobic tails) is dense and impenetrable, keeping invaders (like unwanted water molecules) out. This is how our cells maintain their integrity and control what goes in and out.
Without this lipid barrier, our cells would essentially dissolve in the watery environment of our bodies. It’s a testament to the genius of biological design, all powered by these amazing waterproof molecules.
So, the next time you see a duck gliding effortlessly on the water, or you notice how your skin doesn't instantly turn into a raisin, or you're wrestling with a greasy pan, take a moment to appreciate the incredible world of lipids. They are the silent, unsung heroes of waterproof-ness, making our lives drier, healthier, and a whole lot less soggy, one molecule at a time. They’re basically the body’s and nature’s original Teflon coating, keeping things running smoothly and, well, dry!
