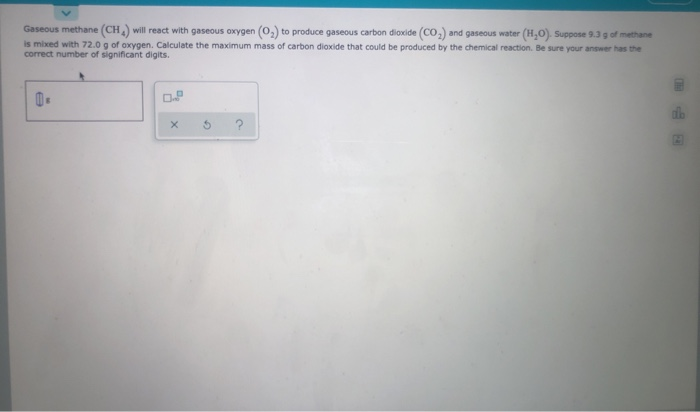
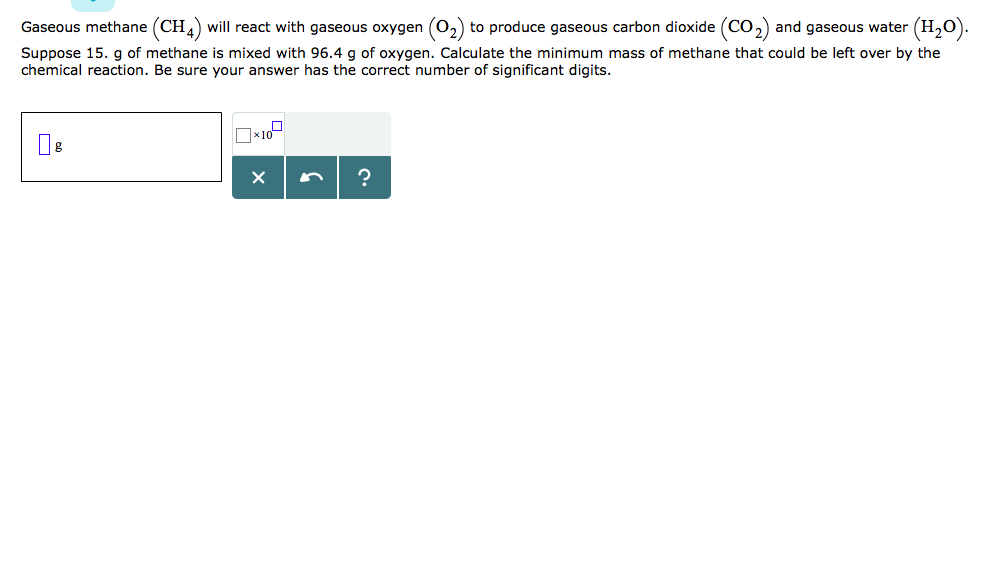
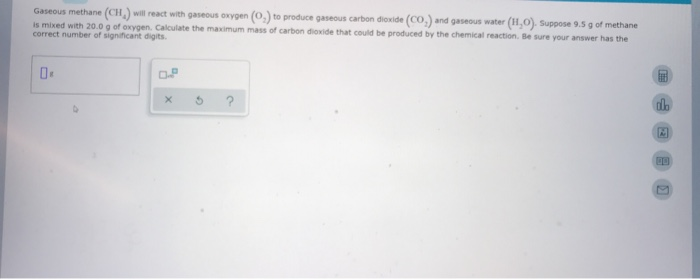
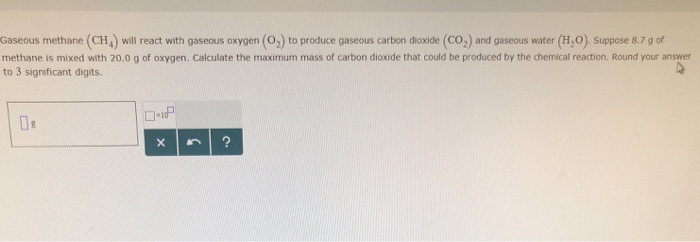
Gaseous Methane Will React With Gaseous Oxygen
Okay, so picture this: I’m a kid, probably around seven or eight, and my dad’s trying to teach me something about… well, something vaguely scientific. He pulls out this old, dusty box of matches, the kind you strike on the side of the box. He lights one, and for a glorious, flickering second, there’s this tiny, dancing flame. Then, poof, it’s gone. He hands me a match, a little nervous because, you know, fire, and I strike it. Same thing. Flame, then darkness. He smiles and says, “See? It burned out. It used up all its fuel.”
Fuel. That’s the word that stuck with me. Like the gas in the car, or the stuff we use to cook. But then he goes on, and this is where my little brain started to get all buzzed up, “Everything that burns needs two things: fuel and oxygen.” He pointed to the air around us. “This air,” he said, “is full of oxygen. It’s what lets things burn.”
Fast forward a couple of decades, and I’m still thinking about that. Turns out, my dad wasn’t just talking about wooden matches. He was hinting at this fundamental, and honestly, pretty awesome, chemical reaction that happens all around us, all the time. We’re talking about the fiery dance between gaseous methane and gaseous oxygen.
Methane, right? Sounds a bit science-y, but you’ve definitely encountered it. It’s the main ingredient in natural gas, the stuff that heats our homes, cooks our food, and, if you’ve ever been unlucky enough to get stuck behind a particularly gassy cow, you’ve experienced it in its… less refined forms. It’s everywhere! And it’s a gas. And it loves to party with oxygen.
So, when we’re talking about gaseous methane (that’s CH4 for you chemistry buffs out there, but let’s stick with the fun names for now) and gaseous oxygen (O2, our friendly air-breather), we’re not just talking about two gases hanging out. We’re talking about potential energy just waiting to be unleashed. Think of methane as the eager dancer, and oxygen as the enthusiastic partner. They’re just waiting for the right moment to twirl.
The Spark of Life (or Combustion, Anyway)
You can have methane all day long, and you can have oxygen all day long, and nothing much happens. They’re like two shy people at a party, standing in opposite corners. But introduce a little something… a spark, a bit of heat, a tiny bit of energy to get the ball rolling, and BAM! Things get interesting. This is what we call activation energy, and it’s crucial. It’s like the DJ dropping the beat and saying, “Okay, dancers, it’s time to MOVE!”
Once that initial spark happens, once that first bond breaks and things start rearranging, the reaction between methane and oxygen kicks into high gear. It’s a chain reaction, a cascade of molecular rearrangements that releases a heck of a lot of energy. And this is where my dad’s match analogy really comes into play. The matchstick was the methane, the air was the oxygen, and the strike was the spark.
The general idea, the big picture, is this: methane (CH4) gets together with oxygen (O2), and with a little nudge, they transform into something entirely new. They become carbon dioxide (CO2) and water (H2O). Sounds simple, right? But the way it happens is where the magic (and the heat) lies.
Imagine methane as a little molecule with one carbon atom in the middle, all snuggled up with four hydrogen atoms. Oxygen is a bit different; it’s two oxygen atoms linked together. When they react, those bonds that hold them together have to break. And breaking bonds? That takes energy. But here's the twist: the new bonds that form between carbon and oxygen, and between hydrogen and oxygen to make water, are much, much stronger and release way more energy than was needed to break the original bonds. It’s like a give-and-take, but the 'take' is way bigger than the 'give'. That excess energy? That’s what we see as heat and light. That’s fire!
The Chemical Ballet: What's Really Going On?
Let’s get a tiny bit nerdy for a second, okay? Don’t worry, I’ll keep it light. The balanced chemical equation for this whole shebang looks like this: CH4 + 2O2 → CO2 + 2H2O.
What does that mean in plain English? One molecule of methane needs two molecules of oxygen to have a complete and utter combustion party. And the result? One molecule of carbon dioxide and two molecules of water. Easy peasy, lemon squeezy, as my grandma used to say. Though I’m not sure she was talking about chemical reactions.
The methane molecule (CH4) has its carbon atom bonded to four hydrogen atoms. Oxygen molecules (O2) are made of two oxygen atoms double-bonded together. To get to CO2 and H2O, all those original bonds have to be broken. Think of it like deconstructing a LEGO set. You have to take all the pieces apart before you can build something new.
First, the bonds within the methane molecule start to break. Then, the double bonds in the oxygen molecules break. This requires that initial energy input – the spark, the heat. Once those bonds are broken, the atoms are free to recombine in new ways. The carbon atom, which is quite happy to bond with oxygen (it’s a very social atom), finds two oxygen atoms and forms carbon dioxide (CO2). Each hydrogen atom, now freed from the methane, finds an oxygen atom, and they team up to form water (H2O).
And remember all that energy we talked about? When the carbon-oxygen and hydrogen-oxygen bonds form, they release a tremendous amount of energy. This is what powers our stoves, our boilers, and, unfortunately, can contribute to environmental issues when we burn fossil fuels.
It's a bit like taking a bunch of building blocks and rearranging them. You break down the old structure, which takes some effort, but the new structure you build is more stable and releases energy in the process. It's a fundamental concept in chemistry, and it's happening constantly, from the smallest flame to massive industrial processes.
Why Should We Care About Methane and Oxygen Hugging?
Well, for starters, it’s the basis of a huge chunk of our energy production. When we burn natural gas in our homes, that’s methane reacting with oxygen. When power plants generate electricity using natural gas, same thing. It’s a relatively clean-burning fuel compared to others, producing mostly CO2 and water, but that CO2 is still a greenhouse gas, so it’s a bit of a double-edged sword. We get warmth and power, but we also contribute to climate change.
But it’s not just about our stoves. Methane is also a potent greenhouse gas in its own right, even before it reacts. It’s released from natural sources like wetlands and from human activities like agriculture (hello, cow burps!) and fossil fuel extraction. When methane is in the atmosphere, it traps heat. It’s like a cozy blanket for the Earth, but we’ve been adding a lot of blankets lately, and things are getting a bit too warm.
So, understanding how methane reacts with oxygen is not just an academic exercise. It’s about understanding our energy sources, our impact on the environment, and the fundamental processes that govern our planet.
Think about it: that little match flame my dad showed me? It was a miniature version of the same reaction that powers cities. It’s a chemical transformation driven by the need for things to become more stable, releasing energy in the process. And it all starts with a little spark and the willingness of methane and oxygen to get together and make something new.
It’s pretty wild when you stop and think about it, isn't it? We’re surrounded by invisible gases, and with just a little nudge, they can put on a spectacular show of energy release. It’s a reminder that even the most ordinary things, like the air we breathe, are part of incredibly complex and dynamic processes. So next time you turn on your stove or see a flame, you can think, “Ah, yes. That's the glorious union of gaseous methane and gaseous oxygen at work!” And maybe, just maybe, you’ll feel a tiny bit of that eight-year-old wonder about the world around you.
It's a constant cycle, really. The Earth generates methane, and the atmosphere provides the oxygen for it to potentially react. We humans have just learned to harness that reaction for our own purposes, for better or worse. It’s a powerful reminder of the interconnectedness of everything, from the smallest molecule to the global climate.
And the beauty of it, in a scientific sense, is its predictability. We know that given the right conditions, methane will react with oxygen. It’s a fundamental law of nature. It doesn't get much more reliable than that. Unlike, say, predicting the stock market, which is a whole other ballgame!
So, yeah. Methane and oxygen. Not the most glamorous duo in the chemical world, perhaps, but undeniably important. They’re the unsung heroes (or villains, depending on your perspective) behind so much of our modern life and a key factor in the ongoing conversation about our planet's future. Pretty cool, right?
