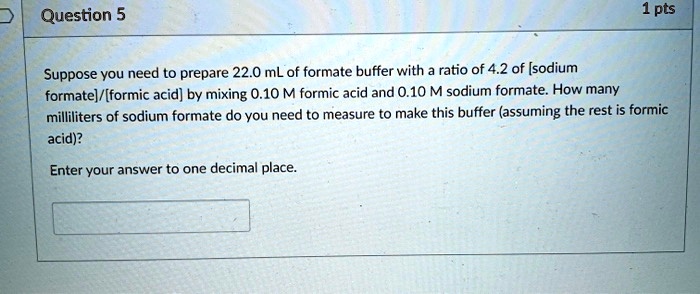
Formic Acid And Sodium Formate Buffer Equation

Ever feel like your morning coffee is just a tad too acidic, or maybe your lemonade is heading towards "pucker-up-so-hard-you-might-turn-inside-out" territory? Well, guess what? The universe, in its infinite wisdom (and sometimes slightly chaotic glory), has built-in little helpers to keep things chill. Today, we're diving headfirst into the wonderful world of Formic Acid and Sodium Formate buffers, and trust me, it's way more exciting than it sounds! Think of it as the unsung hero of stable pH, the chill dude at the party who keeps everyone from getting too wild.
Imagine you're having a picnic, and the weather is just…perfect. Not too hot, not too cold. The sandwiches are delicious, the company is great. Suddenly, a rogue cloud threatens to unleash a downpour, ruining your perfect afternoon. What do you do? You whip out your trusty umbrella, right? That umbrella is like our Formic Acid and Sodium Formate buffer system. It’s there to catch those "rainy day" pH drops and keep everything smooth sailing. Without it, things could get messy, just like a soggy sandwich.
So, what are these magical ingredients? We've got Formic Acid, which is, you guessed it, an acid. Think of it as the energetic, slightly excitable friend. It’s got a little zing to it. Then we have Sodium Formate. This is the salt of formic acid. It’s like the calm, collected cousin who’s always got your back. When these two pals hang out together in a solution, they form a dynamic duo, a pH-stabilizing dream team that works wonders.
Now, let's talk about the magic behind the curtain – the Henderson-Hasselbalch equation. Don't let the fancy name scare you! It’s like a secret recipe for keeping your solution’s pH perfectly balanced. For our formic acid and sodium formate friends, this equation looks like this:
pH = pKa + log ([Formate Ion] / [Formic Acid])
SOLVED: Question 5 1 pts Suppose you need to prepare 22.0 mL of formate
See? It’s not brain surgery! The pKa is just a number that tells us how strong our formic acid is when it's hanging out with its buddy, water. The real stars of the show here are the concentrations of the formate ion (from the sodium formate) and the formic acid itself. This equation is basically telling us: "Hey, as long as you’ve got a good amount of both these guys around, your pH will stay pretty darn stable!" It’s like having a thermostat for your liquid. You can crank up the heat (add acid), or turn down the cool (add base), and this buffer system will bravely resist, trying its best to keep things at the temperature you want.
Why is this so cool? Well, imagine you're trying to make that perfect batch of homemade ice cream. If the acidity is all over the place, you might end up with something that tastes like frozen toothpaste. Blech! But with a good buffer, like our formic acid and sodium formate pals, you can keep that creamy, delicious pH just right, ensuring your ice cream is a triumph, not a tragedy. It’s the difference between a delightful dessert and a science experiment gone wrong!
Let’s get a little playful with it. Imagine your solution is a bustling city. The formic acid is like the enthusiastic police force, always ready to react to any unwanted troublemakers (acids or bases). The sodium formate is like the solid infrastructure – the roads, the buildings – providing the necessary environment for the police to do their job effectively. When a new, aggressive acid tries to storm into town, the formic acid steps in, neutralizing it with all its might. If a sneaky base tries to sneak in, the formic acid’s trusty sidekick, the formate ion, jumps into action. It’s a well-oiled machine, keeping the city (your solution) peaceful and orderly.
This isn't just for food, either! Think about the amazing world of pharmaceuticals. When scientists are making life-saving medicines, they need to be super precise. The pH of a drug can dramatically affect how it works in your body. A good buffer ensures that the medicine stays potent and effective, doing exactly what it’s supposed to do. So, in a way, formic acid and sodium formate are contributing to keeping us healthy and happy. Pretty neat, huh?
Even in the humble world of your car's battery, pH control is crucial. Keeping things within the right range prevents corrosion and ensures your battery performs optimally. It's like giving your battery a nice, stable home environment so it can keep powering your tunes and your drives. Formic acid and sodium formate are out there, quietly doing their job, making sure things run smoothly.
So, the next time you’re enjoying a perfectly brewed cup of tea, or marveling at the stability of a scientific process, give a little nod to the unsung heroes: Formic Acid and Sodium Formate. They’re the ultimate chill-out crew, the pH superheroes, working tirelessly behind the scenes to make sure our world, and our experiments, stay beautifully balanced. And honestly, who doesn't love a good balance?