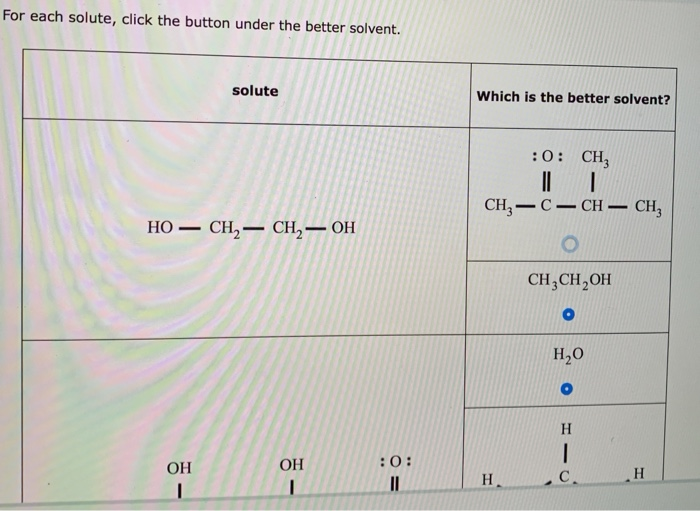
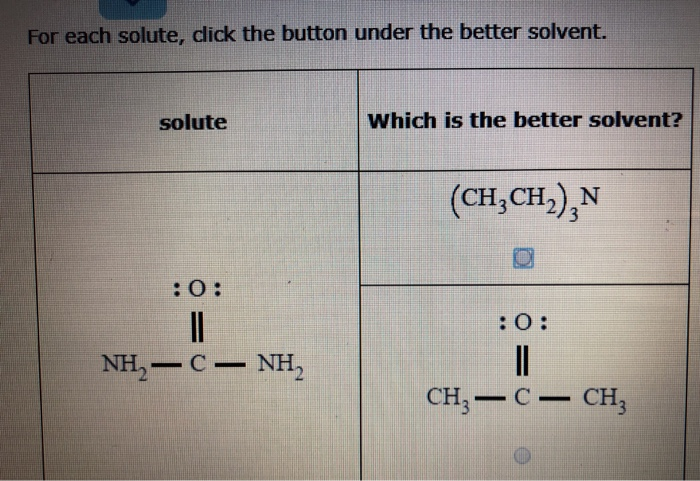
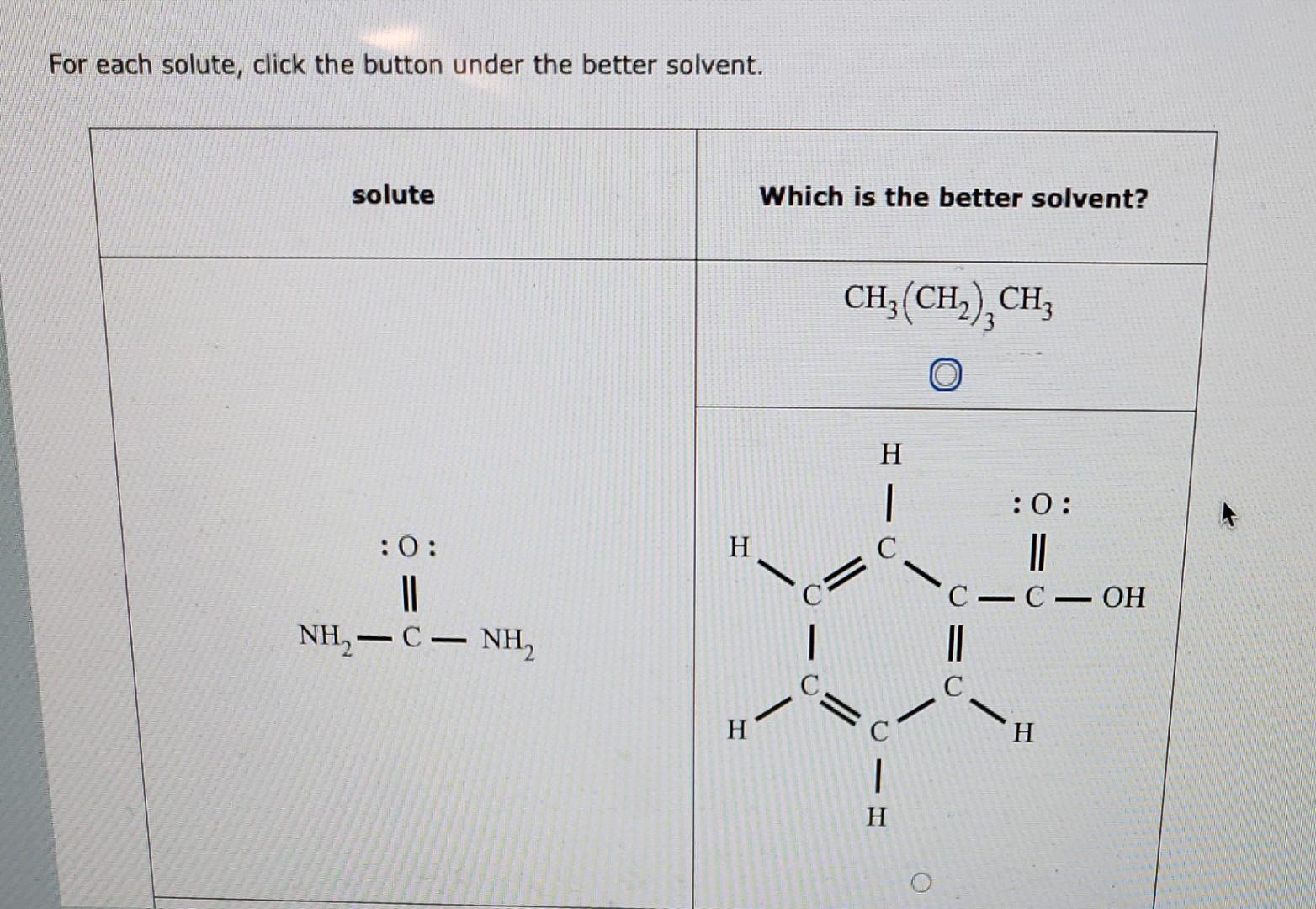
For Each Solute Click The Button Under The Better Solvent
Ever find yourself staring at a cup of coffee, a glass of juice, or maybe even a science experiment kit (hey, no judgment!) and wonder, "Why does this dissolve in that?" Well, get ready for a little peek behind the curtain of the everyday magic that makes our world go 'round. It's like a giant, invisible "Which One Will It Be?" game, and the players are pretty darn interesting.
Imagine you have a spoonful of sugar. You know, the sweet stuff that makes everything better. Now, what do you pour into it? Water, right? Of course! It’s like the ultimate best friend for sugar. They just click. The sugar molecules, like tiny energetic dancers, just love to boogie around in the water. They spread out, get all cozy, and voilà! You have sweet, sweet tea or lemonade. But what if you tried to dissolve sugar in, say, cooking oil? Nope. Nada. The sugar just sits there, stubbornly refusing to join the oil party. It's like the oil is too much of a smooth operator, and the sugar is just too, well, sugary to fit in. It's a classic case of opposites not attracting when it comes to these two.
Think of it like this: Water is the ultimate party host, always ready to welcome and mingle with all sorts of guests. Oil, on the other hand, is more of a… exclusive club. It prefers its own kind.
Now, let's talk about salt. That little white crystal that makes popcorn pop and fries taste divine. You know what’s great for salt? Yep, you guessed it, good old water again! It’s like salt and water have a secret handshake. The water molecules grab onto the salt bits and pull them apart, spreading them out so evenly you can’t even see them anymore. It’s an invisible uncoupling, a tiny molecular breakup that happens so fast and so completely. But try to mix salt with something like sand? They just sort of sit next to each other, like awkward strangers at a party who haven't been introduced. Sand, bless its heart, is just too big and too stubborn to be dissolved by water. It prefers to stay in its sandy little clump.
This is where it gets really fun. Let’s say you have some food coloring. Those vibrant little drops that turn your milk into a rainbow! What do you think they’ll dissolve in best? If you’re thinking water, you’re spot on! Food coloring is designed to spread its colorful cheer throughout water, making everything look festive. Imagine your kids' faces when you turn their milk blue or pink! It’s pure delight. But if you were to try and mix food coloring into something like rubbing alcohol (the kind you might use for cleaning a small cut), it wouldn't mix nearly as well. The colors would probably clump up, looking like tiny, sad blobs. The water is just the much better dance floor for those colorful molecules.
What about something a little more… oily? Like grease from that delicious burger you had? You can't just splash water on that and expect it to disappear. That's where dish soap comes in! Dish soap is like a superhero for grease. It has a special trick: one part of the soap molecule loves water, and the other part loves oil. So, it acts as a bridge, grabbing onto the greasy bits and then letting the water wash them all away. It’s a beautiful collaboration, a testament to how different things can work together. If you tried to use just water on the grease, it would slide right off, leaving you with a greasy mess and a sad, uncleaned pan. Water, in this case, is a bit clueless about how to handle the oily situation on its own.
And then there are those moments when you might be making something a bit more… sophisticated. Like if you’re working with certain types of glues or paints. Some of these are designed to work with water, making them easy to clean up with a wet cloth. It’s a parent’s dream come true, right? You’ve got little artists at work, and a spill happens. No panic! A quick wipe, and it's like it never happened. But then there are other kinds of glues and paints that are made with different ingredients. They might need a special kind of solvent, like mineral spirits, to clean them up. Trying to use water on those would be like trying to paint a masterpiece with a damp sponge – it just wouldn’t do the job. The mineral spirits are the true allies for those particular sticky situations.
It's a fascinating dance, isn't it? The tiny world of molecules, playing out these little dramas of attraction and repulsion all around us. From the simplest cup of coffee to the most complex cleaning task, there's a whole universe of "who likes who" happening. So, the next time you’re dissolving something, take a moment to appreciate the unsung heroes: the solvents! They're the real stars of the show, making our drinks sweet, our dishes clean, and our world just a little bit more wonderfully mixed.
