Fluorine Is More Electronegative Than Oxygen Because

Hey there, science curious folks! Ever wonder about the tiny building blocks of everything around us? We're talking about atoms, the invisible wonders that make up our world. And today, we're going to spill the tea on a couple of particularly punchy characters: Fluorine and Oxygen. These guys are pretty important, and there's a little secret about them that's actually super cool. So, why is Fluorine such a bigger "grabber" than Oxygen? Let's dive in!
Imagine atoms as little social butterflies at a party. They all have this desire to get along with others, but some are just a bit more… well, clinging than others. This clinging power is something scientists call electronegativity. Think of it as an atom's "attraction power" for electrons, which are like tiny, negative-charged dancers swirling around the atom's center. When atoms bump into each other and decide to form a bond (like holding hands), they might share these dancers. But sometimes, one atom is just a little bit better at convincing the dancers to stay closer to its side. That's where our stars, Fluorine and Oxygen, come into play.
Now, Fluorine is like the ultimate socialite. It's incredibly, ridiculously good at pulling those electron dancers towards itself. It's practically a black hole for electrons in its little atomic neighborhood! On the other hand, Oxygen is pretty good too, a popular and well-liked guest at the atom party, but it just doesn't have that same, shall we say, intense grip as Fluorine.
So, what gives Fluorine its super-grabby superpower? It all comes down to its size and the way its "dancers" are arranged. Think of it this way: Fluorine is a smaller atom. It has fewer layers of these electron dancers, and the ones it has are closer to the atom's core. This core has a positive charge, and opposites attract, right? So, that positive core in Fluorine has a much stronger pull on the negatively charged electron dancers because they're so close.
Oxygen, while still popular, is a slightly bigger atom. It has more layers of electron dancers, and the outer ones are a bit further away from its core. Because of this distance, the positive pull from the core isn't quite as strong on those outer dancers. It's like trying to hold onto a balloon when it's right next to you versus holding onto one when it's a bit further away – the closer one is easier to keep a firm grip on.
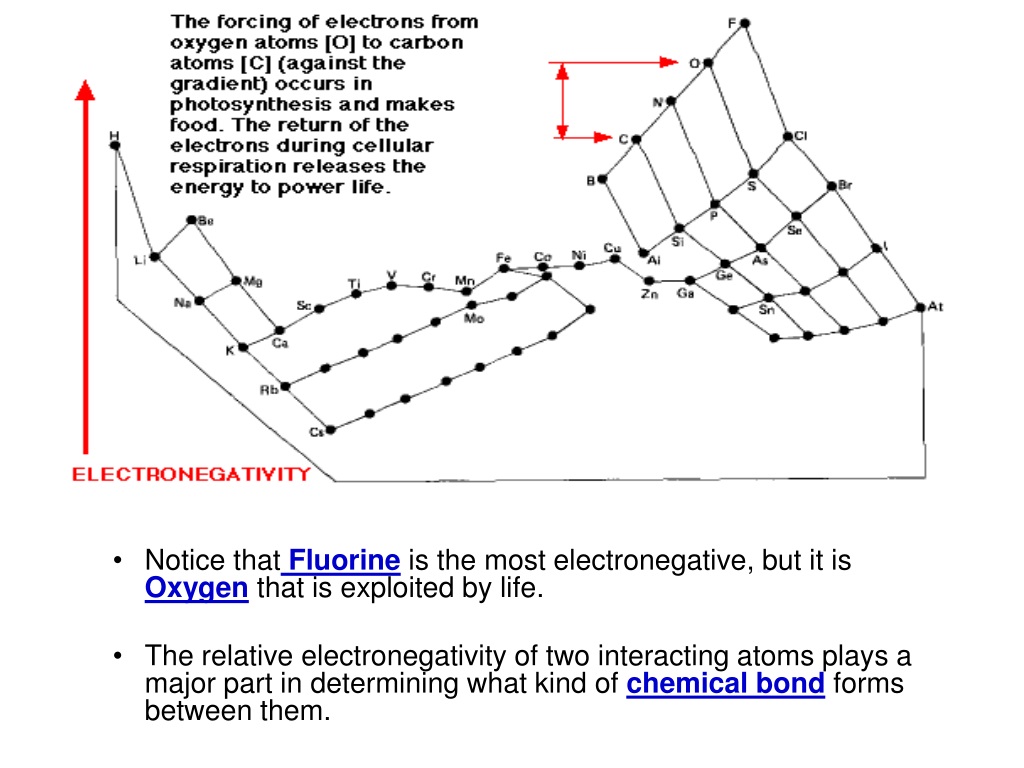
This difference in "clinging power" is what makes Fluorine the most electronegative element on the entire periodic table. It’s the undisputed champion! Oxygen comes in a very respectable second place, which is still incredibly important. This is why when Fluorine and Oxygen get together (which they sometimes do in certain chemical compounds, though it's a bit of a dramatic partnership!), the Fluorine atom pretty much hog all the electron dancers. It’s like Fluorine saying, "Mine!" and the electrons happily obliging.
Why should you care about this seemingly small detail in the world of atoms? Because this little difference in electronegativity has huge consequences for how molecules behave! Think about water, H₂O. That's Oxygen holding hands with two Hydrogens. Because Oxygen is quite electronegative, it pulls the electrons towards itself, making the water molecule a bit "uneven." One side is slightly negative, and the other side is slightly positive. This "unevenness" is what makes water such a fantastic solvent – it can dissolve so many things! It's like water has tiny little positive and negative ends that can attract and pull apart other molecules.

Now, imagine if you had a molecule with Fluorine involved. Because Fluorine is even more electronegative, it creates even more extreme "unevenness" in the molecules it bonds with. This leads to some really unique and powerful chemical properties. Many incredibly useful compounds, like the ones used in non-stick pans (think Teflon!) and refrigerants, are built around the incredible strength of the Carbon-Fluorine bond. That strong bond, driven by Fluorine's super-electronegativity, makes these materials very stable and resistant to breaking down.
"It's the little things that make the biggest differences, and Fluorine's extreme pull is one of those 'little things' with enormous impact!"
It’s fascinating to think that something as tiny as an atom’s attraction for its electron dancers can dictate the properties of everything from the water we drink to the materials we use every day. Fluorine, the element with the ultimate electron-grabbing superpower, and Oxygen, its slightly less intense but still powerful cousin, are constantly at work, shaping our world in ways we often don't even realize.
So, the next time you’re using a non-stick pan or even just taking a sip of water, you can give a little nod to these amazing elements and their electric personalities. Fluorine is more electronegative than Oxygen because of its smaller size and the resulting stronger pull its nucleus has on its electrons. It’s a fundamental principle in chemistry, but it’s also a fantastic little story about how the universe works, one atom at a time. Pretty neat, right? Keep exploring, and who knows what other atomic wonders you’ll uncover!
