Find The Ph Of A 0.0075 M Sulfuric Acid Solution.
:max_bytes(150000):strip_icc()/sulfuric-acid-molecule-147219841-58a0d0253df78c47580373b5.jpg)
Hey there, ever found yourself wondering about the secret life of liquids? You know, the stuff that makes your morning coffee just right, or that cleans your kitchen counters with a satisfying sparkle? Well, today we're diving into a slightly more technical, but still super-duper interesting, topic: the pH of a 0.0075 M sulfuric acid solution.
Now, before you picture a mad scientist in a bubbling lab coat, let's take a deep breath. We're going to break this down like we're figuring out the perfect pizza toppings – easy peasy! Think of pH as a liquid's personality. Some liquids are friendly and mellow, like plain ol' water (pH of 7, right in the middle of the road). Others can be a bit feisty, like lemon juice or vinegar, which are acidic (lower pH). And then there are the super-friendly ones, like baking soda paste, which are alkaline or basic (higher pH).
So, why should you, a perfectly normal human being who probably has more important things to worry about than the precise pH of a chemical, actually care about this? Well, it’s like knowing why your sourdough starter needs a specific temperature to bubble happily, or why your cat prefers a certain brand of food. Understanding pH helps us understand the world around us, and how things behave. It’s the invisible force that influences everything from the taste of your food to the health of our oceans.
Today, we’re looking at sulfuric acid. Now, sulfuric acid sounds a bit intimidating, doesn’t it? Like something you’d see in a cartoon villain’s lair. And it is a powerful acid. But even powerful things can be understood and handled with care. Imagine a really strong cup of coffee. It can wake you up, right? But if you drink too much, it can make you jittery. Sulfuric acid is kind of like that, but on a much grander scale. It's used in all sorts of things, from making fertilizers for our food to refining metals. It’s a workhorse chemical!
The "M" in "0.0075 M" is just a fancy science way of saying "molarity." Think of it like a recipe. If a recipe calls for 2 cups of flour, that's a specific amount. Molarity is a specific amount of our sulfuric acid dissolved in a liter of water. So, we're dealing with a very diluted solution of sulfuric acid. It's not like someone just poured a bucket of concentrated acid into your bathtub! This is more like adding a tiny drop of super-concentrated juice to a large glass of water to make a mildly flavored drink.
Let's Talk About the "pH" Part
Remember how we said pH is like a personality? For acids, a lower pH means they're more "acidic." The pH scale runs from 0 to 14. 7 is neutral (like pure water). Anything below 7 is acidic, and anything above 7 is alkaline (or basic). The lower you go on the scale, the stronger the acid. A pH of 1 is way more acidic than a pH of 6.
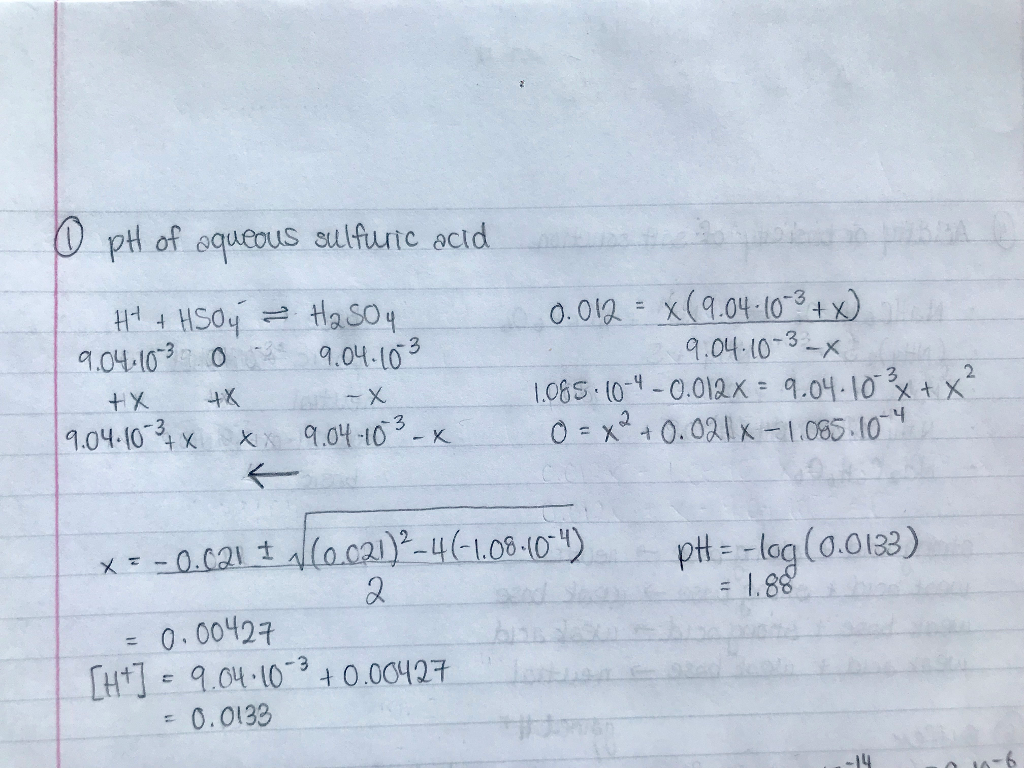
Sulfuric acid, H₂SO₄, is a bit of a special case among acids. It's what we call a "strong diprotic acid." Now, don't let those words scare you! "Diprotic" just means it can donate two hydrogen ions (H⁺) when it dissolves in water. Think of it like a superhero who has two special powers instead of just one. The first hydrogen ion comes off pretty easily, making it a strong acid. The second one is a bit more reluctant, but it still contributes to the overall acidity.
Because sulfuric acid has these two potential H⁺ ions, when we calculate its pH, we have to be a little clever. For our very diluted 0.0075 M solution, the first hydrogen ion dissociates (comes off) almost completely. This means that for every molecule of sulfuric acid, we get one H⁺ ion. So, at first glance, you might think the concentration of H⁺ ions is just 0.0075 M.
If that were the case, we could use a simple formula: pH = -log[H⁺]. So, pH = -log(0.0075). Let's plug that into our calculator. You'd get a pH of roughly 2.12. That’s pretty acidic, like strong vinegar!

But Wait, There's a Little More!
Here's where the "diprotic" nature of sulfuric acid gets interesting, especially when we're dealing with less dilute solutions. While the first hydrogen comes off readily, the second one doesn't always completely dissociate. It's like the second superhero power isn't always switched on. This means the concentration of H⁺ ions might be slightly higher than what we'd expect from just one H⁺ per molecule.
However, for our very, very dilute 0.0075 M solution, the concentration of the acid is so low that the second dissociation becomes much more significant. It's like in a huge crowd, even a quiet whisper can be heard. The water molecules are very good at helping that second hydrogen ion find a home. So, in this specific case, it's often a good approximation to assume both hydrogen ions from the sulfuric acid molecules are contributing to the H⁺ concentration.
This means we can effectively double the concentration of H⁺ ions we're considering for our pH calculation. So, instead of 0.0075 M, we're looking at something closer to 0.0075 M * 2 = 0.015 M of H⁺ ions.
Let's Do the Math (The Easy Way!)
Now, let's use that same handy formula: pH = -log[H⁺].
So, pH = -log(0.015).
Punching this into your calculator (or using an online pH calculator), you'll find that the pH of a 0.0075 M sulfuric acid solution is approximately 1.82.

See? Not so scary after all! We've gone from a confusing chemical formula to a number that tells us something about its personality. A pH of 1.82 is definitely on the acidic side of the scale. It's more acidic than lemon juice (which is around pH 2), but not as strong as stomach acid (which is around pH 1.5 to 3.5).
Why Does This Even Matter in the Real World?
Knowing this helps us understand things like:
- Acid Rain: Sometimes, pollution in the air can react with water to form acids, including sulfuric acid. This can make rain acidic, and even a small change in pH can harm plants, animals, and buildings. Understanding the pH helps scientists monitor and address this.
- Industrial Processes: Many industries rely on precise pH control. Whether they're making pharmaceuticals, cleaning metals, or treating wastewater, knowing the pH of solutions like sulfuric acid is crucial for safety and efficiency. Imagine trying to bake a cake without knowing how much baking soda to add – you wouldn't get the fluffy results you want!
- Environmental Monitoring: Rivers, lakes, and oceans have their own pH levels. Changes can indicate pollution or other environmental issues. A sudden drop in pH could be a red flag, like seeing a wilting plant that needs a drink.
- Everyday Products: Even the products you use at home have pH. Shampoos, soaps, and cleaning agents are formulated to be at specific pH levels to work effectively and be safe for you. While you won't be directly working with 0.0075 M sulfuric acid at home, understanding pH helps you appreciate the science behind everyday items.
So, the next time you hear about pH, remember it's not just for scientists. It's a fundamental concept that helps us understand and interact with the world around us, from the tiniest chemical reactions to the health of our planet. And that 0.0075 M sulfuric acid solution? It’s just one tiny piece of a much bigger, and fascinating, chemical puzzle! Keep exploring, and don't be afraid to ask questions – it's how we all learn and grow, one bubbly experiment (or easy-going article) at a time!
