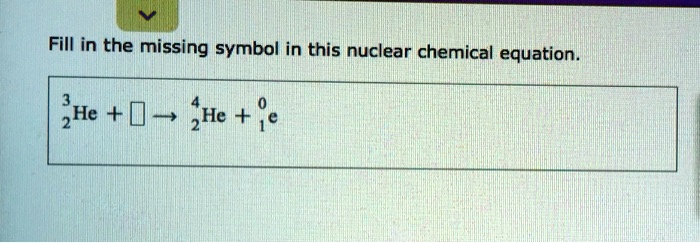
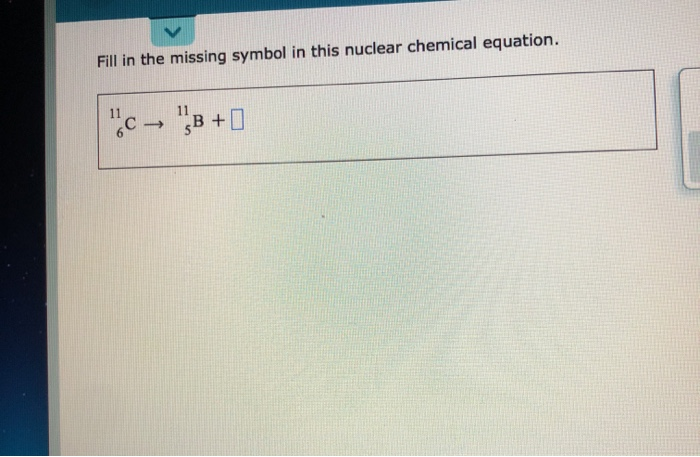
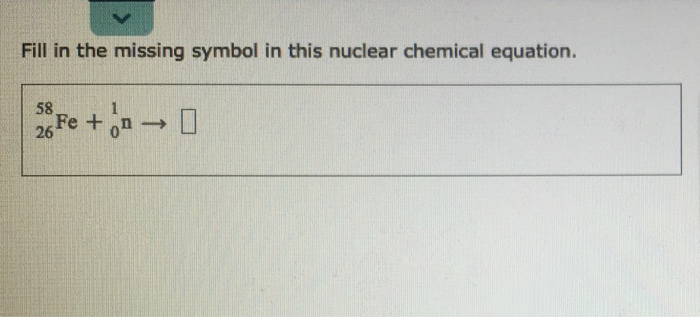
Fill In The Missing Symbol In This Nuclear Chemical Equation.
Hey there, science curious folks! Get ready for a little brain tickler that's way more exciting than your average Sudoku. We're about to dive into the wonderfully weird world of nuclear chemistry, and it’s not as scary as it sounds – promise!
Imagine you're putting together a super-secret recipe, but instead of flour and sugar, we're dealing with tiny, energetic particles. These particles have names like protons, neutrons, and electrons. They’re like the tiny Lego bricks of the universe, and sometimes, they go through some pretty dramatic transformations.
Today, we’re playing a game called "Fill In The Missing Symbol In This Nuclear Chemical Equation." Think of it like a cosmic scavenger hunt where the treasure is a missing piece of the atomic puzzle. It’s a puzzle that nature itself is constantly solving, and we get to be super-sleuths and figure out the missing ingredient.
So, what exactly is a nuclear chemical equation? Well, it's basically a way to show how atoms change and release energy. It’s like watching a tiny, invisible firework show happening inside atoms! These aren't your garden-variety chemical reactions, like baking a cake or rusting a nail. This is the heavyweight championship of atomic transformations.
In the world of nuclear chemistry, things get a bit wild. Atoms can split apart, join together, or even change from one element into another. It’s like a magician pulling a rabbit out of a hat, but instead of a rabbit, it's a whole new element popping into existence!
Our mission, should we choose to accept it (and you totally should because it's fun!), is to find the one missing symbol in a given equation. This symbol could be an alpha particle, a beta particle, a gamma ray, or even a different type of atom altogether. It's like a missing piece in a jigsaw puzzle, and once you find it, the whole picture makes perfect, atomic sense.
Think of it like this: You’re making a cosmic sandwich. You’ve got your bread (one atom), your fillings (other particles), and you know what the final sandwich should look like. But oh no! A crucial slice of cheese (the missing symbol) has vanished!
Your job is to figure out what that missing slice of cheese is, based on the ingredients you have and the delicious sandwich you’re aiming for. It sounds simple, right? But in the quantum realm, those "simple" ingredients have some very specific rules they follow.

The fundamental rule we’re playing by is called the conservation of mass-energy. Don't let that fancy name scare you. It just means that in any nuclear reaction, the total "stuff" (mass) and the total "oomph" (energy) on one side of the equation must equal the total "stuff" and "oomph" on the other side. It’s like saying you can’t create something from nothing, or make something disappear into thin air, not even in the atomic world!
This is where the magic – or rather, the science – happens. We look at the numbers of protons and neutrons on each side. Protons are like the atomic ID number, defining what element an atom is. Neutrons are like its heavier, chill cousin.
In nuclear equations, we have these neat little numbers attached to our symbols. The top number is the mass number (protons + neutrons), and the bottom number is the atomic number (just the protons). It’s like a tag on each particle, telling us its weight and its identity.
Let’s say we have a situation like this (don't worry, we'll get to a real example in a sec!):
Atom A (Mass X, Atomic Y) ---> Atom B (Mass Z, Atomic W) + ???
We know Atom A and Atom B. We’re staring at the question mark, the ultimate cosmic mystery. Our mission is to make sure the numbers balance out.

So, for the mass numbers, we’d say: X = Z + Mass of ???. See? Simple subtraction!
And for the atomic numbers: Y = W + Atomic Number of ???. More simple subtraction!
Once we figure out the mass number and the atomic number of our missing piece, we can often identify exactly what it is. It might be something as common as a helium nucleus (which we call an alpha particle, written as ⁴₂He), or a speedy electron (a beta particle, written as ⁰₋₁e), or even just a packet of pure energy (a gamma ray, written as γ).
These particles have their own super-specific symbols and numbers. The alpha particle, for instance, has a mass number of 4 and an atomic number of 2. An electron has a mass number of 0 (because it’s so tiny!) and a "negative" atomic number of -1. This negative charge is super important and balances things out wonderfully!
Let’s try a real-life example, shall we? Imagine this equation:
²³⁸₉₂U ---> ²³⁴Th + ???
We have Uranium-238 (that's the ²³⁸₉₂U part – 238 is its mass, 92 is its atomic number) turning into Thorium-234 (the ²³⁴Th – 234 mass, 90 atomic number). See how the atomic number for Thorium is 90? That's a big clue!
Now, let’s balance the mass numbers. We start with 238 on the left. On the right, we have 234 from Thorium. What number do we need to add to 234 to get 238?
That’s right! It's 4! So, the mass number of our missing particle is 4.
Next, let’s balance the atomic numbers. We start with 92 on the left. On the right, we have 90 from Thorium. What number do we need to add to 90 to get 92?
You got it again! It's 2!
So, we’re looking for a particle with a mass number of 4 and an atomic number of 2. Does that sound familiar? It’s our good old friend, the alpha particle, which is essentially a helium nucleus! We write it as ⁴₂He.
And there you have it! We’ve cracked the cosmic code and found the missing symbol!
This little game of balancing numbers is fundamental to understanding how radioactive elements decay and how we can even use nuclear processes for things like medical imaging or generating power. It’s all about those precise, atomic-level bookkeeping skills.
The beauty of these nuclear equations is that they reveal the underlying order in what can seem like chaotic atomic transformations. It's like a hidden choreography of the universe playing out in real-time, and we're getting a peek behind the curtain.
So, next time you see a nuclear equation, don't get intimidated! Think of it as a fun puzzle, a cosmic scavenger hunt, or even a secret recipe where you just need to find that one missing, incredibly important ingredient. You’re not just looking at symbols; you’re uncovering the fundamental rules that govern the tiniest, most energetic parts of our world.
Keep playing, keep guessing, and keep that scientific curiosity alive! You’re doing great, and you’re officially a nuclear detective!
