Express The Enthalpy Of The Following Reaction
Ever wondered about the secret energy lurking within chemical reactions? Well, get ready to have some fun because we're diving into the world of enthalpy! It might sound like a fancy science word, but think of it as the heat content of a chemical reaction. Understanding enthalpy is like unlocking a secret code to how and why things happen when substances mix and transform. It's incredibly useful, whether you're a budding scientist, a curious home cook, or just someone who likes to understand the world around them a little better. And the best part? It’s not as complicated as it seems!
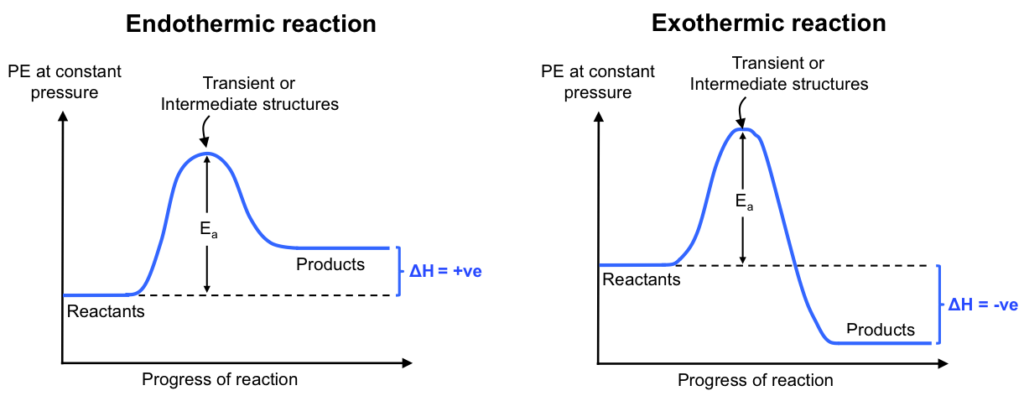
So, what exactly is enthalpy, and why should you care? In simple terms, it's the total heat energy of a system. When a chemical reaction happens, energy is either released or absorbed. Enthalpy change, often represented by the symbol ΔH (read as "delta H"), tells us exactly how much heat is involved. If ΔH is negative, the reaction releases heat (think of a campfire getting warmer) – we call these exothermic reactions. If ΔH is positive, the reaction absorbs heat (like an instant cold pack feeling cold) – these are endothermic reactions.
Why is this so cool? For beginners, it’s a fantastic way to grasp the fundamental concept that chemistry isn't just about mixing things; it’s about energy exchanges. For families, you can explore this by observing everyday phenomena. Think about cooking: when you bake a cake, heat is absorbed by the batter (endothermic process). When your stove burner heats up, it's releasing heat (exothermic). For hobbyists, perhaps you’re into making your own soaps or candles. Knowing about exothermic reactions can help you understand how they generate heat during the curing process, ensuring you handle them safely.
Let’s look at a simple example. When hydrogen gas (H₂) reacts with oxygen gas (O₂) to form water (H₂O), it releases a significant amount of energy. The enthalpy change for this reaction is approximately -483.6 kJ/mol. That negative sign tells us it's exothermic – it gives off heat! Imagine this happening on a grand scale; it’s the principle behind rocket fuel. On the flip side, when ice melts, it absorbs heat from its surroundings, which is why our drinks get colder. This is an endothermic process.

Getting started with understanding enthalpy is easy. You don't need a lab coat! Start by observing. Notice which everyday activities seem to give off heat and which ones feel cooler. Read up on common chemical reactions and look for their enthalpy changes – many resources are available online. You can even find fun DIY experiments that demonstrate these principles, like making a homemade volcano with baking soda and vinegar, and discussing the heat it might release or absorb.
Understanding enthalpy opens up a fascinating window into the energetic dance of molecules. It’s not just about numbers; it’s about appreciating the subtle and sometimes dramatic energy transformations that power our world. So, next time you feel warmth from a reaction or notice something getting cooler, remember the secret of enthalpy is at play, making science a truly warm subject!
