Experiment 3 Osmosis Direction And Concentration Gradients
Ever feel like you're just… deflating? Like all your hopes and dreams are slowly trickling out? Well, maybe it's not about your life coach, but about your cells. Seriously! Today, we’re diving into the surprisingly dramatic world of osmosis. It’s like a microscopic soap opera happening inside you, right now.
Think of your cells as tiny, pampered apartments. They have walls, which we scientists affectionately call membranes. These membranes are picky. They let some things in and out, but not everything. It’s like a bouncer at a fancy club, but for water.
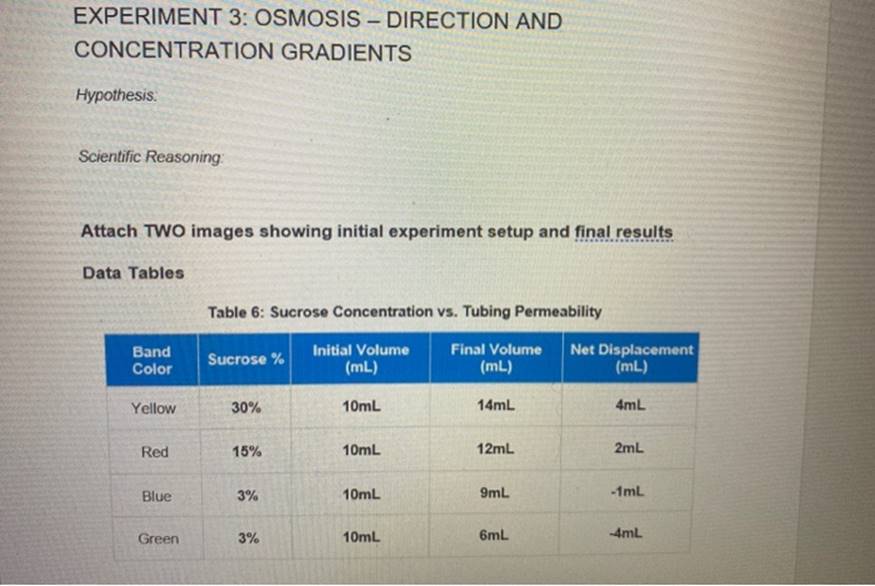
And where does this water go? It follows the concentration gradient. This is basically a fancy way of saying water is a bit of a follower. It likes to hang out where the party is, or in this case, where there are more solutes (stuff dissolved in water).
Imagine you have a glass of water and you drop a sugar cube in it. The sugar is the solute. It’s not evenly spread at first, right? There’s a lot of sugar right where the cube was, and hardly any elsewhere. That's a concentration gradient in action.
Now, our cell membranes are special. They're selectively permeable. This means they're good at letting water pass through, but they're much more hesitant about letting those solute party-goers through. It's like the bouncer saying, "Water? Yeah, you can chill. Sugar? Nah, you gotta wait outside."
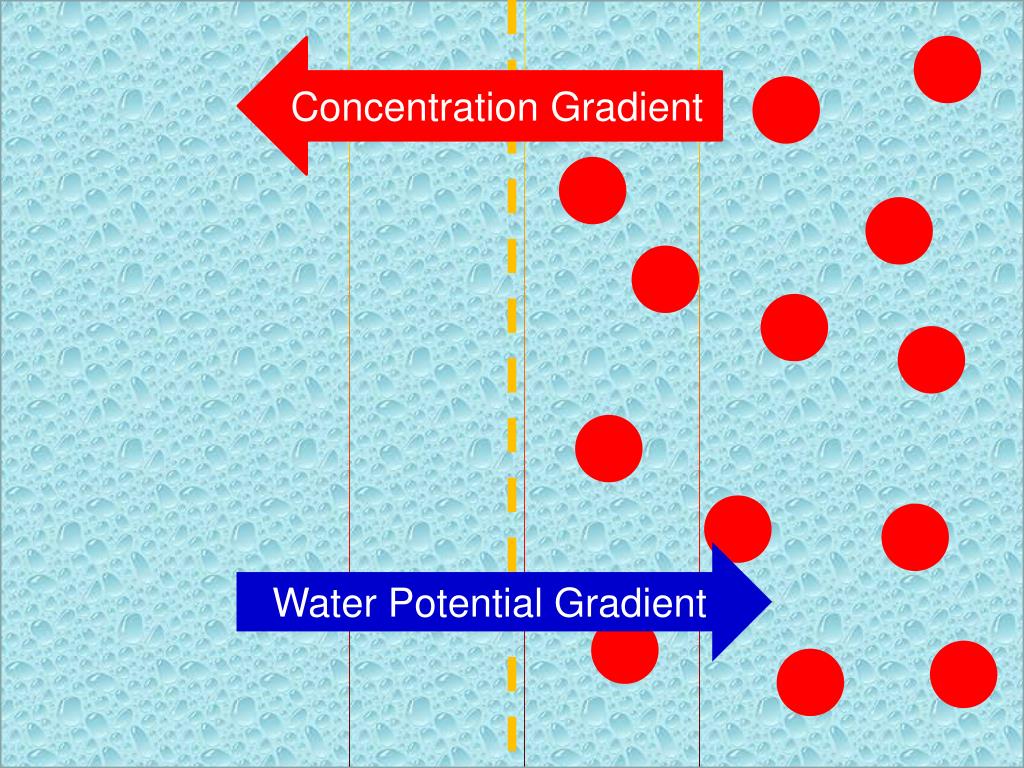
So, if you have a cell sitting in a watery environment where there’s more stuff dissolved outside the cell than inside, guess what happens? The water from inside the cell is going to be tempted by the "party" outside. It's going to start migrating, like tiny water refugees seeking a more concentrated haven.
This migration of water across the membrane is what we call osmosis. It's a passive process, meaning the cell doesn't have to lift a finger (or a tiny organelle) to make it happen. Water just does its thing, driven by this invisible pull of concentration.
Now, let's talk about those cell apartments. If water leaves the cell to join the more concentrated party outside, the cell starts to shrink. It gets all shriveled up, like a raisin that forgot to moisturize. Not a good look for any self-respecting cell.
This is called crenation. It sounds fancy, but it's basically your cell having a really bad hair day, or rather, a really bad cell day. It’s lost its plumpness, its youthful bounce. It’s seen better days, and probably tasted better water.
On the flip side, what if the water party is inside the cell, and the outside is just plain boring and dilute? Then, water from the outside is going to rush in. It's like a water stampede heading for the cell's internal fiesta.
And when water floods into the cell, it starts to swell. It gets bigger and bigger, like a balloon being overfilled. It’s all excited, full of water and ready to burst with… well, water.
This can also be a problem. If a cell takes on too much water, it can actually burst. Poof! Gone. It's the dramatic exit from the microscopic stage. We call this hemolysis when it happens to red blood cells. It’s like a tiny water bomb going off internally.
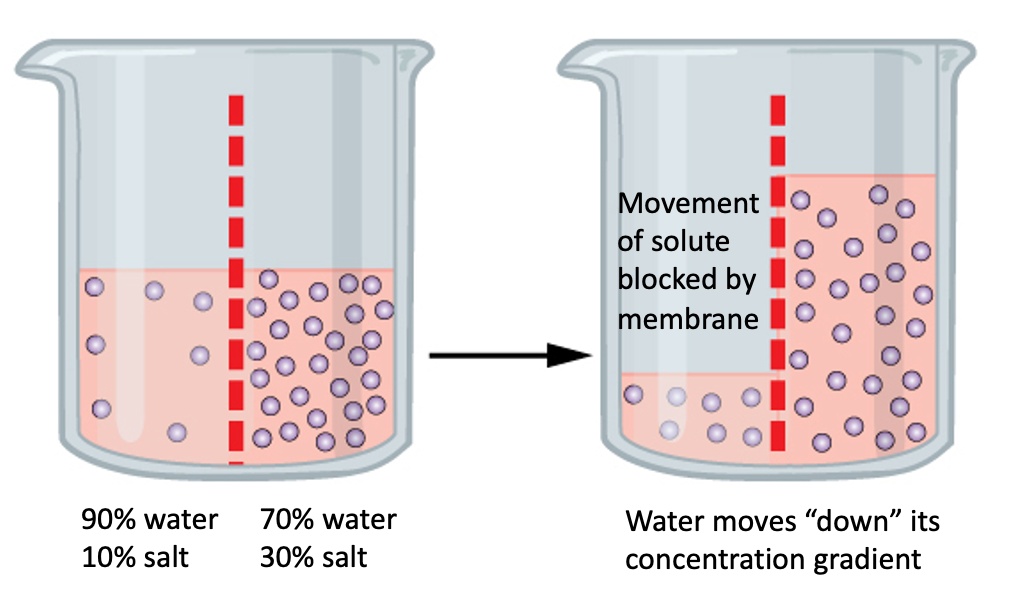
So, the direction of osmosis is all about where the concentration of solutes is higher. Water flows from an area of low solute concentration to an area of high solute concentration. It’s the universe’s way of saying, "Let's balance things out, shall we?"
Think of it like a crowded room versus an empty one. If you have a bucket of water and you pour it into a tiny, crowded closet, the water is going to spread out. But if you pour that water into a vast, empty gymnasium, it’s going to take a long time to get everywhere.
Our cells, however, are much more immediate. They don't have all day to wait for things to equalize. They're constantly trying to maintain a delicate balance, a state we scientists call homeostasis. It’s like keeping your apartment at the perfect temperature and humidity all the time.
The terms hypotonic, isotonic, and hypertonic are where things get really interesting, and potentially a little confusing. But let's break them down, like a slightly overzealous science teacher.
A hypotonic solution is like a very dilute drink. There's not much dissolved in it compared to the inside of your cell. So, water is going to rush into the cell. Prepare for swelling!
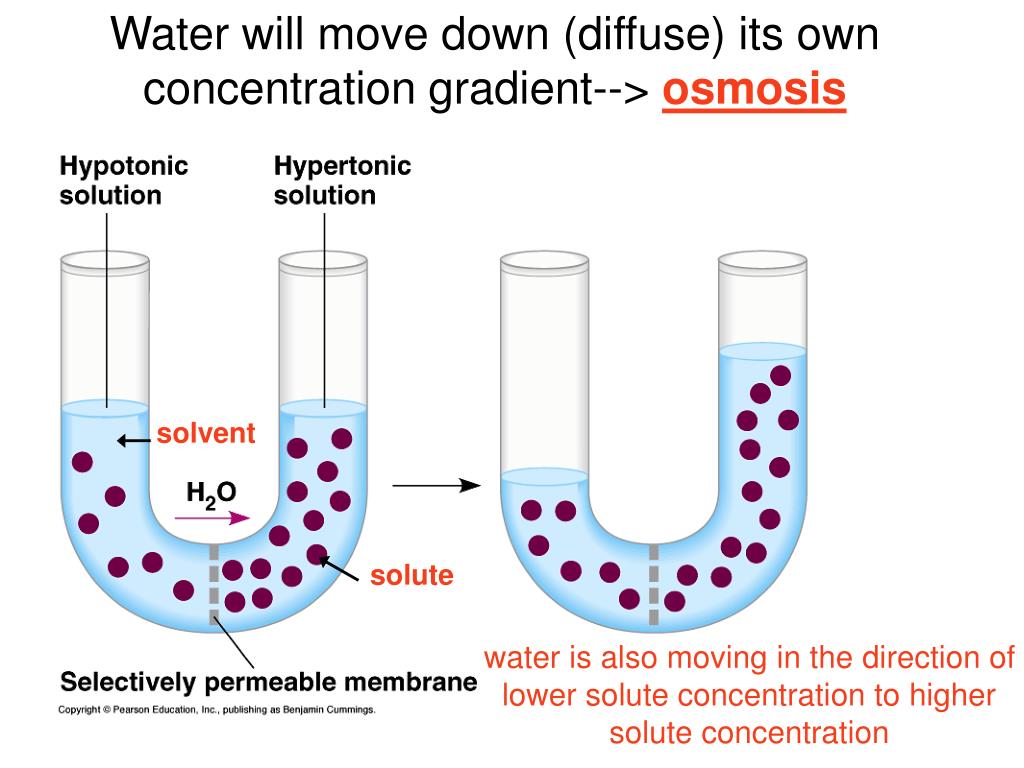
An isotonic solution is the Goldilocks of solutions. It's just right. The concentration of solutes outside the cell is the same as inside. So, water is pretty much chilling, moving in and out at an equal pace. No major drama here.
A hypertonic solution is the opposite of hypotonic. It's like a super-concentrated, syrupy mess. There’s way more stuff dissolved in it than inside your cell. So, water is going to bail from the cell and head for the sugary party outside. Prepare for shriveling!
It's kind of like choosing what to drink. If you're dehydrated, a glass of pure water (hypotonic) will hydrate you. If you drink something super sugary like soda (hypertonic), your body might even pull water from your cells to dilute it. And if you're just thirsty, maybe some sports drink (isotonic) is the way to go.
So, the next time you feel a bit sluggish or bloated, don't blame your diet entirely. It might be your cells having a water-related existential crisis. They're constantly navigating these concentration gradients, trying to stay plump and happy.
It’s a beautiful, microscopic dance of water molecules, all following the invisible signs that say, "More solutes this way!" It's a fundamental process, vital for life, and frankly, a little bit mind-blowing when you think about it.

And hey, if your cells are ever in a particularly stressful environment, just remember they’re doing their best. They’re the unsung heroes of your internal hydration system, all thanks to the magic of osmosis and the relentless pull of the concentration gradient.
So, here’s to our cells, our tiny water-loving apartments, and their constant, silent negotiation with the world around them. May your membranes be selectively permeable, and your gradients always in balance! Or, you know, at least interesting enough to write about.
It's the ultimate act of cellular self-care: understanding where the water wants to go.
We're all just tiny, complex systems of water moving around, trying to find our equilibrium. It's a lot like trying to find a parking spot on a Saturday afternoon, but with more… membranes.
And if you ever feel like your cells are having a party you weren't invited to, it's probably just the solutes doing their thing. Water will follow, eventually. It always does.
So, next time you drink water, give a little nod to the amazing osmosis happening within you. It’s a subtle, constant force that keeps us all going. And isn't that just… wonderfully microscopic?
