Examine The Following Unbalanced Chemical Equation Co2 + C

Hey there, science curious cats and curious cats of all stripes! Ever glanced at a chemical equation and thought, "Whoa, what’s going on there?" Today, we’re diving into a tiny, but totally fascinating, little mystery. It’s just two things, really. We're talking about CO₂ and C. Yep, that’s it. Super simple, right? But oh boy, is there more to it than meets the eye.
So, you see this equation: CO₂ + C. Sounds innocent enough. Like two buddies hanging out. But here’s the kicker: it’s unbalanced. What does that even mean? Think of it like a recipe. You need a certain amount of flour to make a cake. If you just throw some flour in a bowl and call it a cake, well, that’s not gonna work. Chemistry is kinda like that. Everything needs to be in balance.
In chemistry, we’ve got this super important rule called the Law of Conservation of Mass. Basically, it says you can’t just make stuff out of thin air. The atoms you start with have to end up somewhere. They might rearrange themselves, do a little atomic tango, but they’re all still there. It’s like a cosmic game of LEGOs. You can build a spaceship or a castle, but you’re always using the same bricks.
Now, back to our stars of the show: CO₂ and C. What are they, you ask? CO₂ is that stuff we breathe out. Yeah, it's carbon dioxide. It's what makes your fizzy drinks fizzy. It’s also a greenhouse gas, which is why we talk about it a lot, but let’s save the serious stuff for another day. For now, let's just appreciate its bubbly personality.
And then there’s C. That’s just plain old carbon. It's like the LEGO brick of the universe! It's in diamonds, it's in pencil lead (that's graphite, a form of carbon), it's even in us! Carbon is a real party animal in the chemistry world, always connecting with other atoms. It’s the backbone of so many cool molecules. Think of all the amazing things carbon can do. Pretty neat, huh?

So, when we put CO₂ and C together, what are we trying to do? We're basically saying, "Hey, carbon, let’s have a little chat with carbon dioxide." But our equation, CO₂ + C, is like saying, "I have a bag of apples and a single orange." It doesn't tell you what you get at the end of the conversation. It’s incomplete. It’s like a story with no ending.
To make it a complete story, we need to figure out what happens when these two elements decide to mingle. What kind of chemical magic do they cook up? This is where it gets really fun, because there are a few possibilities, and they depend on how we encourage them to react. Think of it like setting the mood for a date. Do you want a romantic dinner, or a wild adventure?
One of the most common things that happens when carbon gets together with carbon dioxide, especially when things get a bit toasty (we're talking high temperatures here, folks!), is that they can form… more carbon monoxide! Yep, you heard that right. CO₂ and C can team up to make CO. It’s like carbon saying, "You know what? I think I'll take one of your oxygen atoms, CO₂, and we can go our separate ways. You can keep the other one."

So, the balanced equation for that would be: CO₂ + C → 2CO. See how we now have two carbons and two oxygens on both sides? That’s balance! We started with one carbon in CO₂ and one lonely C, so that's two carbons total. And we started with two oxygens in CO₂. On the other side, we have two CO molecules, each with one carbon and one oxygen. So, two carbons and two oxygens. Boom! Balanced.
This reaction is actually super important in the real world. It’s how we make things like producer gas, which is a mix of carbon monoxide and hydrogen. This gas can be used as a fuel. So, our seemingly simple unbalanced equation is actually the starting point for some pretty cool industrial processes. Who knew that a little bit of imbalance could lead to so much utility?
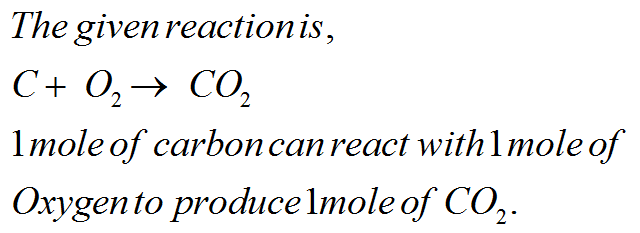
But wait, there's more! Carbon and carbon dioxide can get a little more adventurous. Under different conditions, especially with a bit more heat and perhaps some other catalysts (think of catalysts as the matchmakers of the chemical world, helping reactions happen), they can form solid carbon. This might sound a bit weird, like carbon making more carbon. But it's about rearranging things.
Imagine this: the carbon dioxide molecule is a bit unstable. The carbon atom from the solid carbon is like, "Hey, CO₂, I can offer you a more stable arrangement." And poof! Under the right circumstances, you can end up with more solid carbon and a different gaseous product. This is often seen in things like the Boudouard reaction, which is a real mouthful, but it’s essentially this carbon and CO₂ dance.
The unbalanced equation, CO₂ + C, is like a cliffhanger. It leaves you wanting to know what happens next. It sparks that curiosity. It’s a puzzle waiting to be solved. And the solutions? They're not just abstract chemical formulas. They have real-world implications, from how we generate energy to how certain materials are formed.

Why is it fun to talk about? Because it’s a tiny glimpse into the amazing, invisible world of chemistry. It shows us that even the simplest-looking things can have hidden depths and fascinating stories. It’s like finding a secret code in plain sight. And once you start to understand the code, you see it everywhere.
The fact that CO₂, something we exhale, and C, the fundamental building block of so much, can interact in these ways is pretty mind-blowing. It highlights the constant dance of atoms and molecules happening all around us, all the time. It’s a reminder that the universe is a dynamic, ever-changing place.
So, the next time you see an unbalanced equation, don’t just shrug it off. Think of it as an invitation. An invitation to explore. To discover. To understand the beautiful, intricate ballet of chemistry. CO₂ + C might look simple, but it’s a gateway to a whole world of reactions, energy, and everyday science. It’s proof that even in the smallest packages, there’s a universe of wonder waiting to be uncovered. And isn't that just the coolest thing?
