Examine The Following Unbalanced Chemical Equation
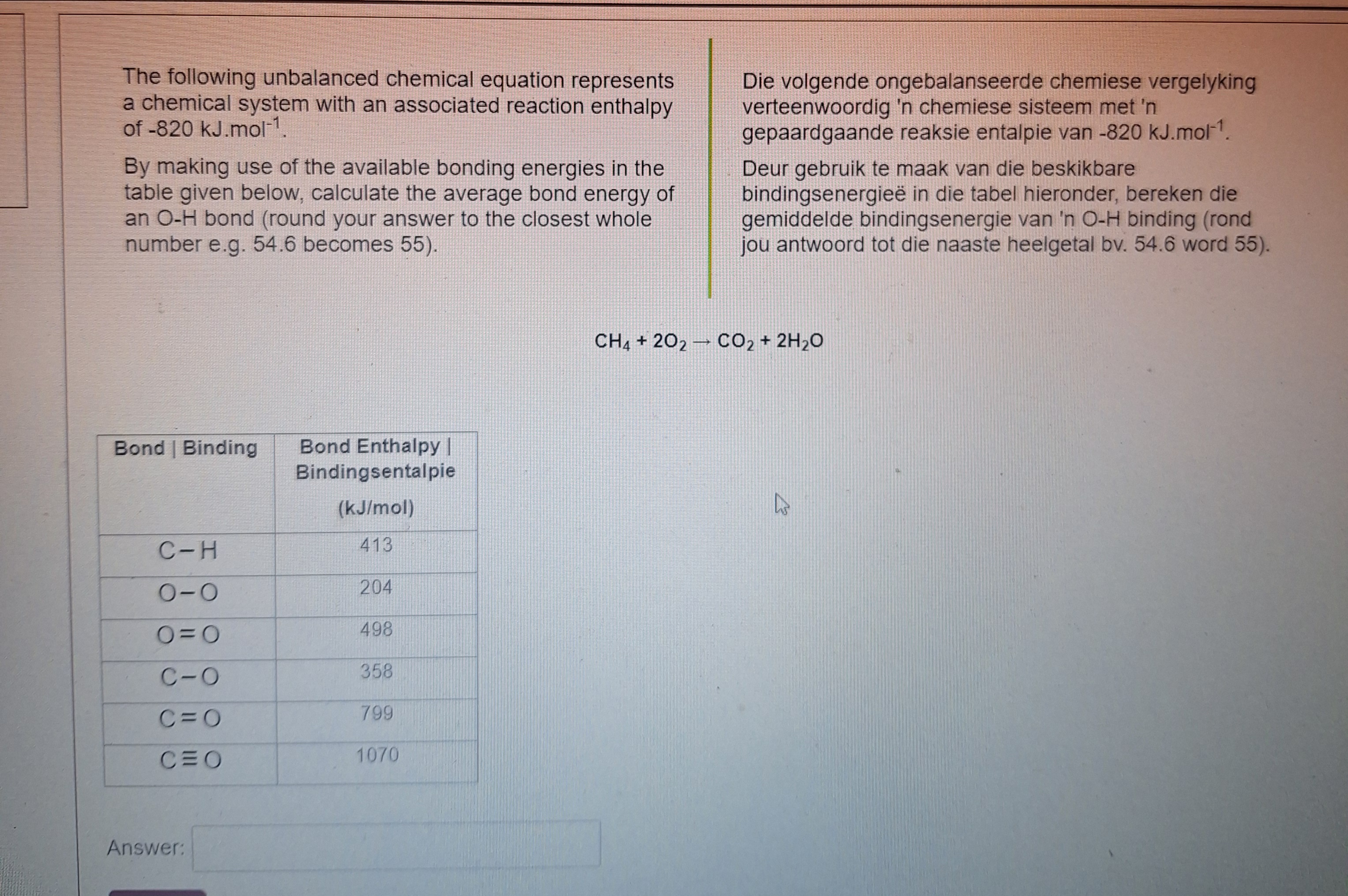
Hey there! So, have you ever just looked at a jumbled mess of letters and numbers and thought, "What in the world is going on here?" Like, it's a chemical equation, and it’s supposed to make sense, right? But sometimes, they just… don't. They’re all higgledy-piggledy. They're like a toddler's toy bin after a major earthquake. You know, the kind where a stuffed bear is trying to have a tea party with a screwdriver and a lone sock. Utter chaos! That's what we're diving into today. We’re talking about those unbalanced chemical equations. You know, the ones that make you scratch your head and wonder if the chemist had too much coffee. Or maybe not enough. It’s a real mystery sometimes, isn't it?
So, picture this: you’ve got a recipe, right? Let's say you're making cookies. You need, like, two cups of flour, one egg, and a sprinkle of fairy dust (okay, maybe not fairy dust, but you get the idea). Now, imagine you’re just looking at the ingredients list and it says "flour, egg, sugar." That's it. No amounts. No proportions. How are you supposed to bake anything? It's the same with these equations. They’re supposed to tell us what’s reacting and what’s being produced, and crucially, how much of each. And when they’re unbalanced, well, it's like trying to bake those cookies with just a vague notion of what goes in. Disaster waiting to happen, my friend. Total kitchen catastrophe.
Let’s get a little bit scientific here, but in a super chill way, okay? Think of a chemical reaction like a big party. You’ve got guests arriving (the reactants) and guests leaving (the products). And what’s the most basic rule of any good party? You can’t just create guests out of thin air, can you? And you can’t have guests just vanish into the ether. It just doesn't work. The number of people who show up has to equal the number of people who leave, right? Or at least, the total number of "partygoers" should stay the same, even if they’ve changed their outfits or are now dancing in a different corner of the room. That’s the core idea behind the Law of Conservation of Mass. It’s this fundamental principle that says matter can’t be created or destroyed. It can only be rearranged. Like LEGOs, you know? You can take apart a spaceship and build a car, but you still have the same number of LEGO bricks. Mind. Blown. Well, maybe not blown, but it’s pretty neat, right?
So, when we see an unbalanced equation, it’s basically violating this law. It’s like saying you put in two apples and somehow ended up with five apples, poof, just like that! Or, you started with three bananas and ended up with zero. Where did they go? Did they elope? Did they join a circus? These equations are giving us the cold shoulder on the whole conservation thing. And that, my dear coffee companion, is why they’re so… unbalanced. They're telling a story that just doesn't add up. It’s like watching a movie where the plot holes are bigger than your living room. You’re just sitting there, bewildered, wondering how they expect you to believe this nonsense.
Let’s look at a classic example. You know, the kind that makes you feel like you’re back in chemistry class, desperately trying to remember your periodic table. We’re going to take a peek at the formation of water. Everyone knows water, right? H2O. Super important stuff. We can’t live without it. It quenches our thirst, makes our tea taste good, and is basically the reason we’re all here. So, how do we make it? Well, usually, you’d react hydrogen gas (H2) with oxygen gas (O2). Sounds simple enough. Let’s write it down, shall we?
H₂ + O₂ → H₂O
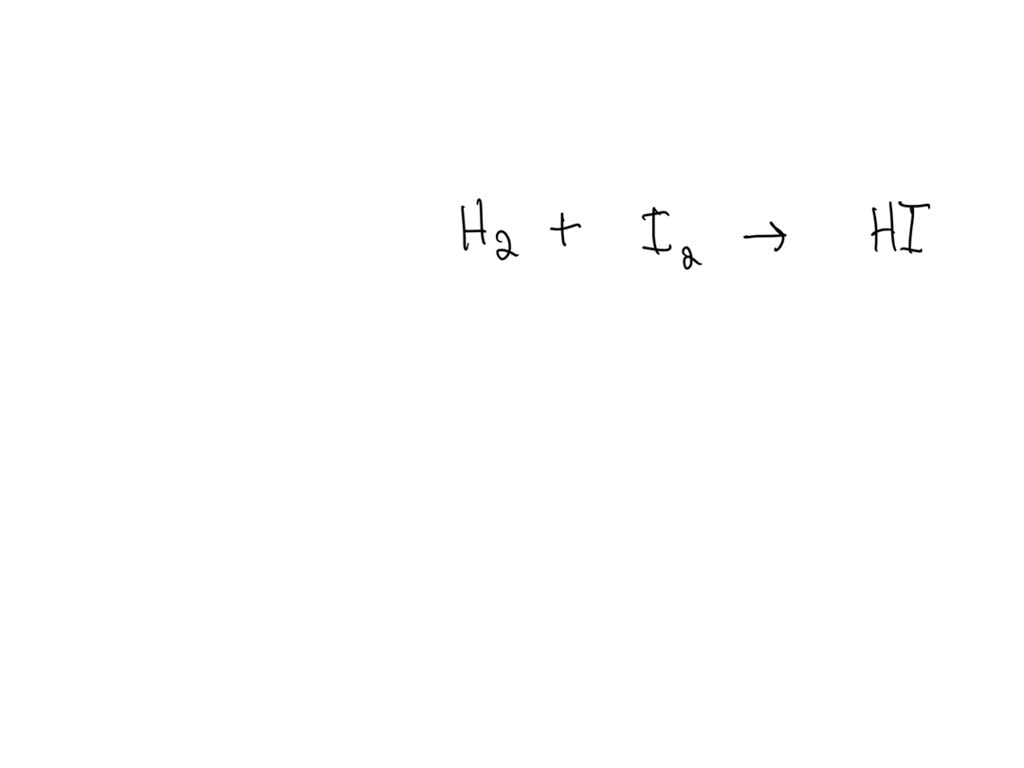
Now, take a deep breath. Look at it. Does it feel… right? Let’s count the atoms on each side. On the left, we’ve got two hydrogen atoms (that little ‘2’ next to the H, remember?). And we’ve got two oxygen atoms (the ‘2’ next to the O). So, 2 hydrogens, 2 oxygens. Easy peasy. Now, let’s hop over to the right side, where our water is chilling. We’ve got two hydrogen atoms (the ‘2’ next to the H). So far, so good. Our hydrogen count is balanced. But then… we’ve only got one oxygen atom. Just one. Where did the other one go? Did it get shy and hide behind the hydrogen? Did it decide it needed some alone time and phased out of existence? This, my friends, is the essence of an unbalanced equation. It’s a chemical cliffhanger, and not in a good way.
It’s like you’re making a sandwich. You’ve got two slices of bread, and you’re going to put some cheese between them. So, two bread slices + cheese = sandwich. Makes sense. But what if the equation looked like this: Bread + Cheese → Sandwich? You’d be like, "Wait a minute. How many slices of bread are we talking about here? Is it one? Two? Am I supposed to guess? Because if I only use one slice, this is going to be a very sad, open-faced disappointment." That’s the same feeling you get with that water equation. It’s missing crucial information, or rather, it’s presenting information that just doesn't align. The atoms aren't playing by the rules!
The problem isn't that we can't form water from hydrogen and oxygen. We absolutely can! That's how stars do it, and they're pretty good at making things, wouldn't you say? The problem is that the equation, as written, is lying to us about the quantities. It's like a fortune teller giving you a vague prediction. "You will meet a tall, dark stranger." Okay, great. Is he bringing pizza? Is he going to steal my wallet? Is he a vampire? So much room for interpretation! Unbalanced equations are like that. They tell us the ingredients, but not the recipe. And without the recipe, well, we’re just winging it. And in chemistry, winging it can lead to… let’s just say, interesting results. Not always the desired, life-sustaining water results.
So, why are these things even around? Are chemists just messing with us? Probably not. Most of the time, these unbalanced equations are just the starting point. Someone might have observed that hydrogen and oxygen react to form water, and they jotted it down quickly. They're like the rough sketch before the masterpiece. Or the initial brainstorm before the brilliant idea. They're not the final product. They're the messy middle. They're the "let's figure this out" stage. And thankfully, there's a way to fix them. It's called balancing the equation. It's like giving that recipe the proper measurements. It's adding the details that make everything click into place. It's the magic wand that turns a jumbled mess into a coherent, scientifically sound statement.
Balancing is all about making sure that the number of each type of atom is the same on both sides of the arrow. Remember our Law of Conservation of Mass? This is where it gets its workout! We’re not adding or removing atoms; we’re just adjusting the coefficients. Coefficients are those numbers you put in front of the chemical formulas. They’re like the multipliers for the whole molecule. So, if you have 2H₂O, it means you have two molecules of water, which translates to four hydrogen atoms and two oxygen atoms. See how that works? It’s not about changing the chemical itself (you can’t just turn one oxygen atom into two hydrogen atoms by magic, sadly), it’s about changing the number of times that chemical unit shows up.
Let's go back to our water example: H₂ + O₂ → H₂O. We saw we had 2 hydrogens and 2 oxygens on the left, and 2 hydrogens and 1 oxygen on the right. The oxygen is the troublemaker here. We have two oxygens on the left, and only one on the right. To fix this, we need to put a coefficient in front of the H₂O on the right side. What number, when multiplied by the one oxygen atom in H₂O, will give us two oxygen atoms? You guessed it: a 2! So, we get:

H₂ + O₂ → 2H₂O
Now, let’s re-count. On the left: 2 hydrogens, 2 oxygens. On the right: 2 * (2 hydrogens) = 4 hydrogens, and 2 * (1 oxygen) = 2 oxygens. Uh oh. Our oxygen count is happy now – both sides have two. But our hydrogen count? We've gone from 2 on the left to 4 on the right. We’ve created a hydrogen surplus! It’s like trying to fix one problem and accidentally creating another. Chemistry can be a real drama queen sometimes, can't it? It’s a constant balancing act, a chemical tightrope walk.
So, what do we do? We need to make sure our hydrogen count is balanced too. We have 4 hydrogens on the right. To get 4 hydrogens on the left, we need to put a coefficient in front of the H₂. What number, when multiplied by the two hydrogen atoms in H₂, gives us four? That would be a 2! So, our equation now looks like this:
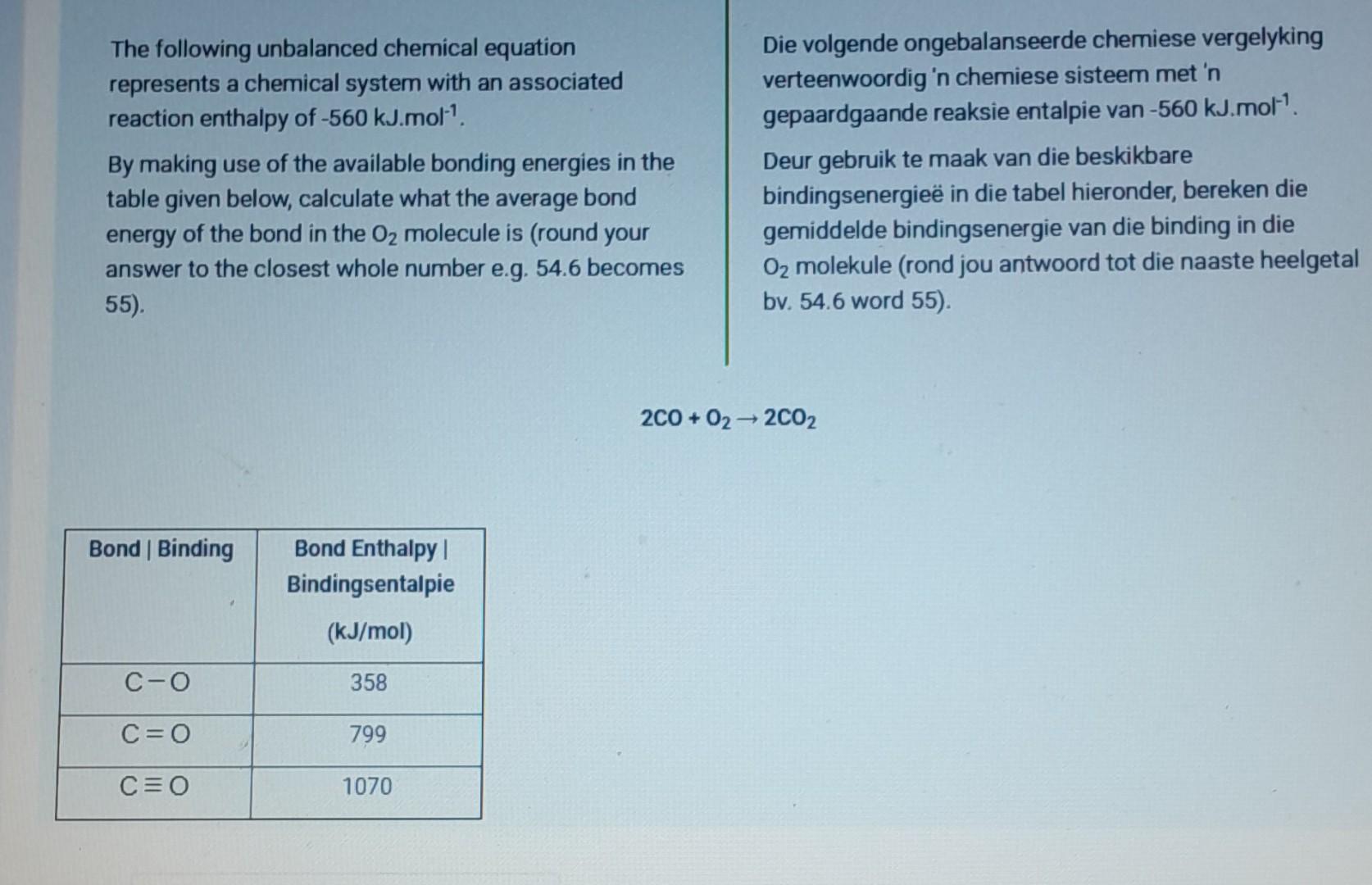
2H₂ + O₂ → 2H₂O
Let’s do a final count, shall we? Left side: 2 * (2 hydrogens) = 4 hydrogens. And 2 oxygens. Right side: 2 * (2 hydrogens) = 4 hydrogens. And 2 * (1 oxygen) = 2 oxygens. Ta-da! Every atom is accounted for. The hydrogens match, the oxygens match. The equation is finally telling the truth, the whole truth, and nothing but the truth about how much hydrogen and oxygen are needed to make water. It’s a beautiful thing, really. It’s like solving a really satisfying puzzle. You know, the ones where you’re left with a sense of accomplishment and a perfectly formed picture?
And that's the whole point of these unbalanced equations, I think. They’re the raw material. They're the starting point for understanding. They remind us that in the universe, nothing just magically appears or disappears. Everything has a reason, a cause, and a consequence. It's a fundamental truth, whether you're talking about atoms or your grocery budget. You can't just spend money you don't have, and you can't create energy out of nowhere. It's all about balance. It's all about conservation. It's about understanding the intricate dance of the universe, one atom at a time.
So, the next time you see an unbalanced chemical equation, don't just shrug your shoulders and move on. Give it a little nod. Recognize it for what it is: a question waiting to be answered. A puzzle waiting to be solved. A story waiting to be told accurately. It’s an invitation to explore the beautiful order that lies beneath the apparent chaos. It’s a reminder that even in the smallest building blocks of our world, there’s a profound sense of balance and purpose. And that, my friend, is worth a second cup of coffee, wouldn't you say? Cheers!
