Ethers Epoxides And Sulfides Practice Problems
Alright, my fellow curious minds and kitchen chemists (even if your kitchen’s still waiting for its molecular gastronomy makeover), let’s dive headfirst into a world of tiny, mighty molecules that are frankly, a blast to wrangle! We’re talking about Ethers, those wonderfully versatile compounds that can smell like anything from roses to nail polish remover (and yes, sometimes both at once!). Then we’ve got the super-powered, ring-shaped wonders called Epoxides. Think of them as tiny, energetic donuts of pure chemical potential. And finally, the sulfur-infused siblings, the Sulfides. They might get a bad rap for their… distinctive aromas, but don't let that fool you; these guys are crucial players in the grand chemical orchestra!
Now, I know what you’re thinking. "Practice problems? Sounds like homework from a very enthusiastic, slightly mad scientist!" And you know what? You’re not entirely wrong, but in the best possible way! These aren't your grandpa’s dusty textbook problems; these are like delicious little brain teasers that help you get a feel for how these molecules behave. It’s like learning to juggle – you start with one ball, then two, and soon you’re a bona fide circus act of chemical understanding!
Let’s kick things off with our friendly neighborhood Ethers. Imagine you’re decorating your dream treehouse. Ethers are like the super-glue that holds different branches together. They're fantastic at linking up other bits of molecules, making them super useful in all sorts of things, from solvents (they’re the reason your paint comes off your hands, thankfully!) to even being used in perfumes. When you're looking at an ether practice problem, you're essentially figuring out how this "treehouse glue" might stick to other parts of the structure or how it might be nudged off to make room for something new. It’s all about understanding its connections!
Ethers: The Chemical Connectors!
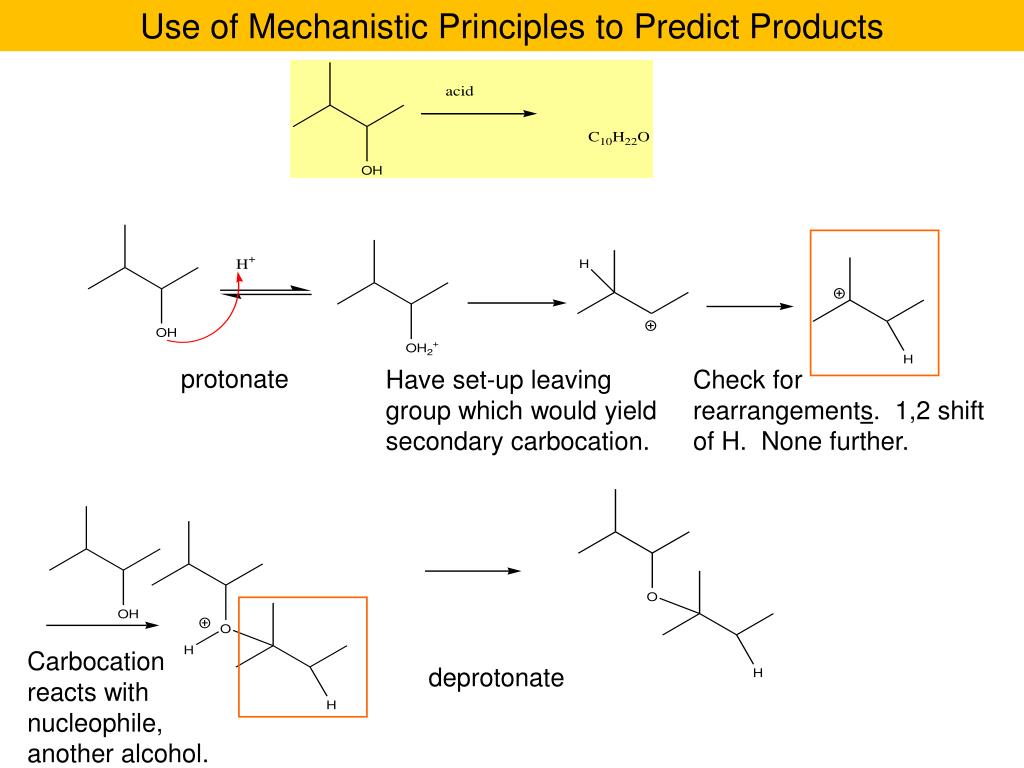
Think of a common ether like diethyl ether. It’s got an oxygen atom nestled between two ethyl groups (which are basically two carbon atoms with a bunch of hydrogens tagging along). In practice problems, you might be asked to predict what happens when this ether meets a strong acid. It’s like asking what happens when you try to unstick your favorite treehouse branch with a very, very determined squirrel. The squirrel (the acid) wants to break that bond, and the ether’s job is to… well, it can be broken, but it puts up a bit of a fight, often in predictable ways. You’re learning to see the subtle cues that tell you how this molecule will react. It’s like being a detective, spotting the tiny clues!
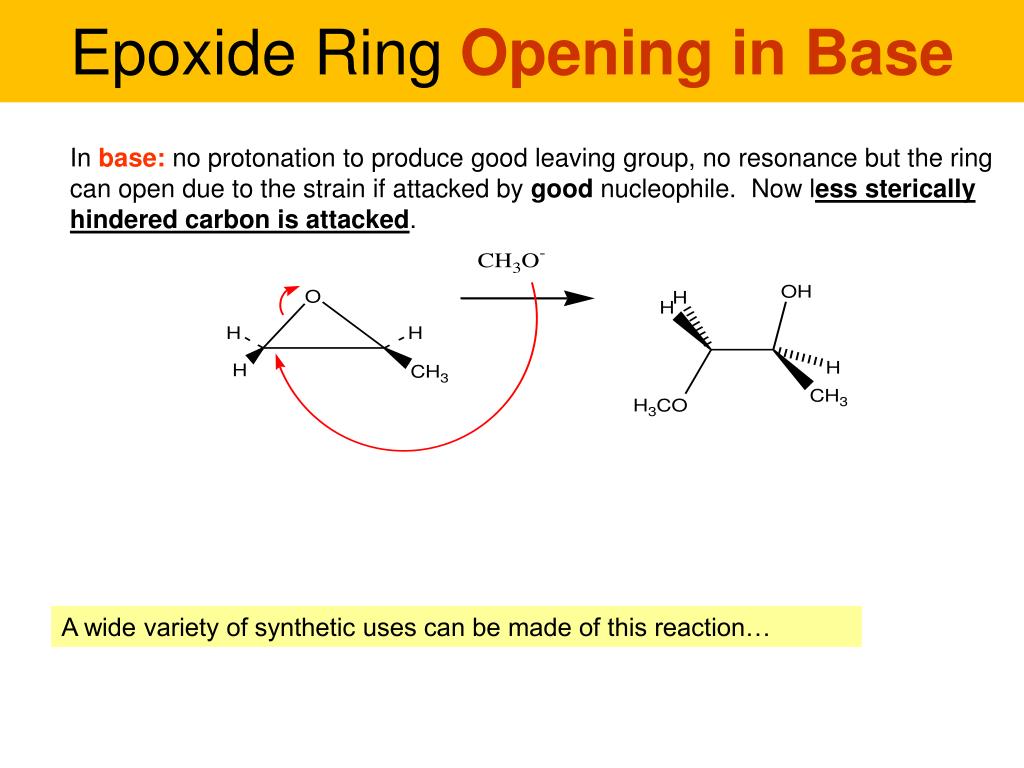
Then, BAM! Enter the Epoxides. These guys are like the hyperactive, three-membered rings of the molecule world. They’ve got an oxygen atom bonded to two carbon atoms, all in a tight, strained little triangle. This strain is their superpower! Because they’re so eager to relieve that tension, they're incredibly reactive. Imagine a tightly coiled spring just waiting to pop. That's an epoxide! In practice problems, you’ll often see them being attacked by all sorts of things – nucleophiles, which are basically molecules that are a bit greedy for electrons. The epoxide ring loves to open up and share its electrons, creating all sorts of new and exciting connections. It’s like a chemical game of “open sesame!” where the ring readily yields to the right key (the nucleophile).
Epoxides: The Ring of Reactivity!
Consider ethylene oxide, the simplest epoxide. It’s a tiny, three-atom ring. When a chemical “attacker” comes along, like a water molecule (H2O), the epoxide ring is basically saying, “Come on in! We’ve got room!” and it springs open, forming new bonds. Practice problems for epoxides often involve figuring out where the attacker will go and what the final product will look like. It’s like predicting the path of a domino after the first one falls – once the ring opens, the rest of the reaction follows a logical, albeit exciting, sequence.
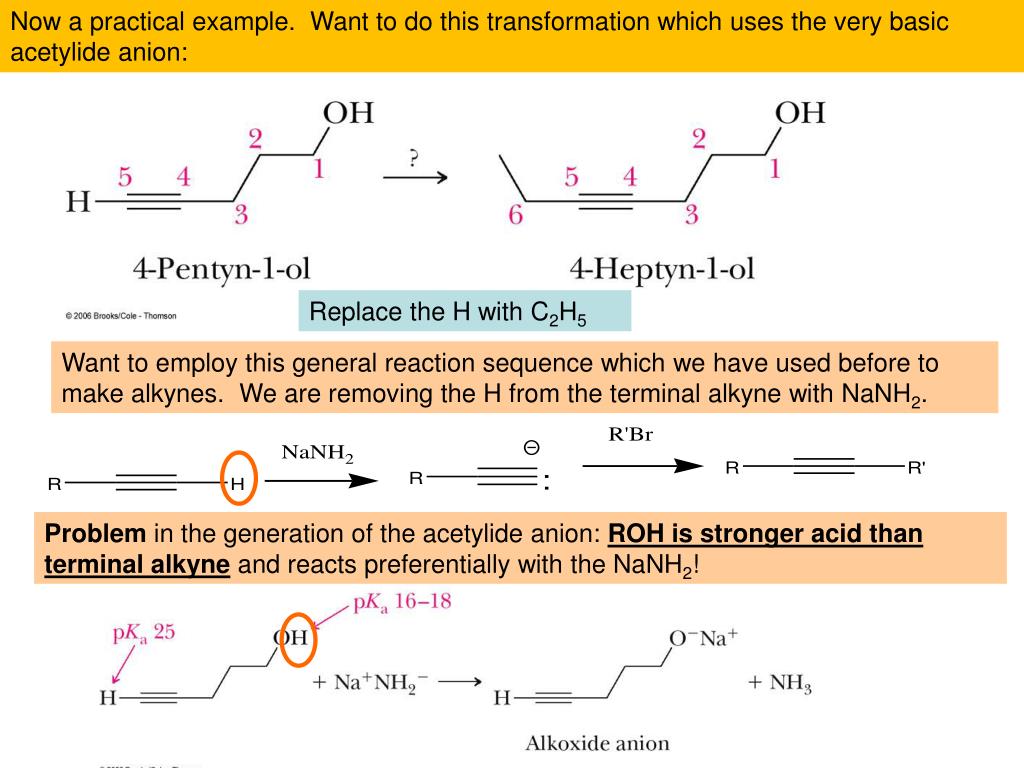
And now, for our final act, the Sulfides! These are the sulfur-analogues of ethers. Instead of an oxygen atom linking two carbon groups, it’s a sulfur atom. So, if ethers are the oxygen-hearted twins, sulfides are their sulfur-hearted cousins. They can smell a bit… pungent (think of a skunk’s signature scent, but sometimes even more… memorable). But don't be fooled by the aroma! Sulfides are vital in biological processes and are used in everything from pharmaceuticals to materials science. In practice problems, you’ll see them acting in ways similar to ethers, but the sulfur atom has its own unique personality. It can be oxidized more easily than oxygen, leading to some fascinating transformations!
Sulfides: The Pungent but Powerful Players!
Think of dimethyl sulfide. It’s like diethyl ether but with sulfur instead of oxygen. When it comes to practice problems, you might be asked about its oxidation. This means adding oxygen to it, which can turn it into a sulfoxide (with a sulfur-oxygen double bond) or even a sulfone (with two oxygen atoms attached to the sulfur). It’s like taking a simple molecule and giving it a chemical facelift, often with very different properties afterward. You're learning to predict the consequences of adding these little oxygen tags, turning a smelly compound into something potentially much more useful and complex!
The beauty of working through these practice problems is that you start to see the patterns. You begin to anticipate how these molecules will dance and react. It’s not about memorizing endless facts; it’s about building an intuition for chemical behavior. You'll tackle problems where you're given a starting material and asked to predict the products, or you'll be given the products and asked to figure out the starting materials and the steps in between. It’s like solving a chemical mystery, and you, my friend, are the star detective!
So, grab your virtual lab coat, embrace the playful challenge, and dive into those ether, epoxide, and sulfide practice problems. You'll be amazed at how quickly you start to feel like a seasoned chemist, navigating the exciting world of molecular transformations with a smile and a dash of scientific flair. Happy problem-solving!
