Enter The Ions Present In A Solution Of Na2co3
So, you’ve got a little puddle of water, right? Nothing fancy. Now, imagine you toss in a pinch of something called sodium carbonate. It’s like the fizzy, happy-go-lucky salt that loves to make things bubbly. When it hits that water, it doesn't just sit there being smug. Oh no, it gets all excited and breaks apart. And that, my friends, is where the fun begins.
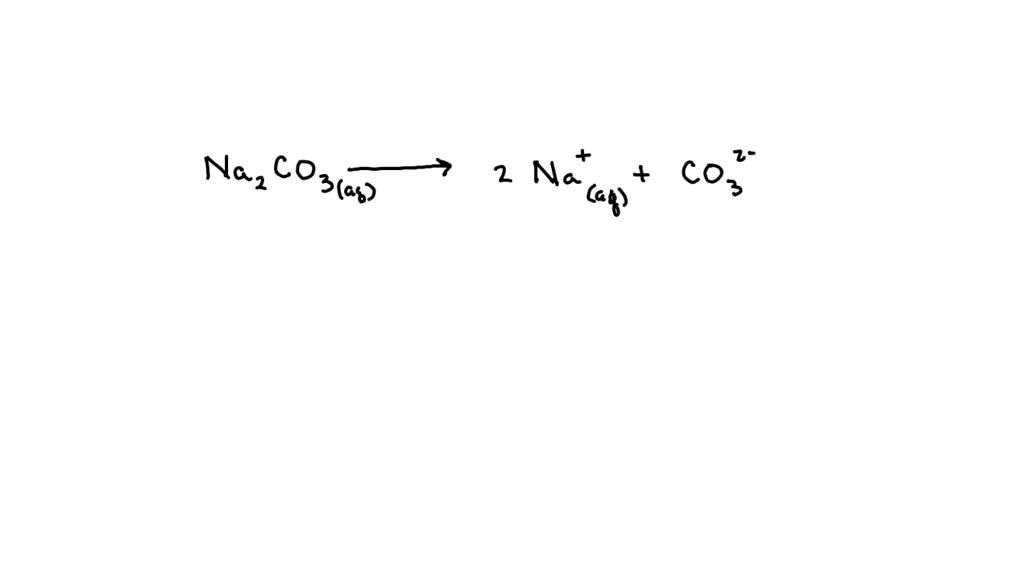
Think of it like a tiny party in your glass. When sodium carbonate (which we chemists affectionately call Na₂CO₃) goes for a swim, it leaves behind some very distinct party guests. These guests are called ions. They’re basically atoms that have either gained or lost a tiny bit of their electrical charge. It’s like they’ve put on or taken off a little sparkly vest, and now they’re ready to mingle.
The first guests to arrive, loud and proud, are the sodium ions. These guys are super chill. They’re like the friends who just show up, happy to be there, and don’t cause any fuss. In chemistry terms, they’re called Na⁺. See that little plus sign? That’s their sparkly vest. They’ve given away a tiny bit of negative charge, making them positively charged. They just float around, enjoying the ride. They’re the unsung heroes of the solution, the ones you don’t really notice but are always present.
But wait, there’s more! The sodium carbonate also brings along a much more… interesting guest. This one is a bit more complicated. It’s the carbonate ion. Now, this guy is a bit of a drama queen. It’s like the friend who arrives and immediately starts rearranging the furniture, but in a good way. Chemically, it’s written as CO₃²⁻. That little minus sign, and a double minus sign at that, tells you it’s been busy. It’s scooped up two extra negative charges. It’s basically overflowing with negative energy, but in a bubbly, effervescent sort of way. This is the ion that’s really responsible for the alkaline nature of the solution, making it a bit slippery to the touch, like a very mild soap. It’s the life of the party, the one who makes things happen.
Now, here’s where it gets really interesting, and perhaps a little unpopular opinion time. While Na⁺ and CO₃²⁻ are the stars of the show, the official break-down of Na₂CO₃ in water technically involves a bit more nuance. It’s not just these two. In water, especially pure water, there are always a few other characters hanging around. There are the hydrogen ions (H⁺) and the hydroxide ions (OH⁻). They’re the water molecules having a brief identity crisis, splitting up for a moment before getting back together.
Honestly, do we really need to talk about H⁺ and OH⁻ when we've got the main event, the Na⁺ and the fabulous CO₃²⁻, throwing a party? It feels a bit like mentioning the caterers when the bride and groom are about to walk down the aisle. They're there, sure, but are they the reason you're excited? Probably not.
So, while technically, a solution of sodium carbonate contains Na⁺, CO₃²⁻, a sprinkling of H⁺, and a handful of OH⁻, my heart, and I suspect yours too, belongs to the main players. It’s the sodium ions, with their unwavering positivity, and the dramatic, charge-carrying carbonate ions, who are the real reason this solution is so… itself. They’re the ones you think of, the ones you can visualize. The others are just background noise, like the awkward cousin at a wedding who you barely remember.
When you add Na₂CO₃ to water, you’re essentially inviting a dynamic duo and a slightly less interesting entourage to a pool party. The sodium ions are the cool, laid-back types who just float and nod along. They’re the dependable friends. Then you have the carbonate ions. These guys are the lifeblood of the operation. They’re the ones bringing the sparkle, the effervescence, and the slightly soapy feel. They’re the ones who make the solution alkaline, ready to tackle dirt or, you know, just make your science experiment a little more interesting.
And those other ions? The hydrogen and hydroxide? They’re like the people who wander into the party accidentally. They’re part of the water, and water is always doing its own thing. They’re present, but they’re not the reason the party is happening. The party is happening because of the sodium carbonate, and specifically, because of the sodium and carbonate ions it releases. It’s a simple equation, really. Salt goes in, ions come out, fun ensues. Or, at least, a chemically interesting situation ensues. And isn't that the best kind of fun?
