Enter The Formula For The Compound Lead Ii Phosphate

Alright, settle in, grab your imaginary latte, and let me tell you a tale. It’s a story about… well, it’s about a compound. But not just any compound, oh no! We’re talking about the thrilling, the mysterious, the downright glamorous world of… Lead(II) Phosphate!
Now, before you yawn and start scrolling through cat videos, hear me out. This isn't your grandpa's dusty chemistry textbook. We're diving in with the enthusiasm of someone who just discovered they accidentally bought a winning lottery ticket. And the prize? The formula for this magnificent beast!
So, picture this: it's a dark and stormy night… okay, maybe it's a sunny Tuesday afternoon, but that doesn't quite have the same dramatic flair, does it? Our protagonist is Lead, specifically Lead in its +2 oxidation state. Think of Lead as a bit of a suave character, always showing up with a "+2" swagger. He’s not the volatile, hyperactive "+4" type, no sir. Lead(II) is more like the cool, collected dude at the party, everyone’s second cousin twice removed who surprisingly knows everyone.
And who does our dashing Lead(II) decide to team up with? None other than the Phosphate ion! Now, Phosphate is a bit of a gangly, multi-tasking character. It’s got this special little arrangement of one phosphorus atom surrounded by four oxygen atoms. Imagine a shy phosphorus kid trying to hide behind a bunch of boisterous oxygen friends. These oxygen friends are so energetic, they’ve collectively decided to give the whole group a negative charge. Specifically, a -3 charge. That's like a really, really big hug of negativity. So, you’ve got Phosphate, the PO43- ion, looking all… charged up.
Now, Lead(II), with his dignified +2, is looking at Phosphate, with its intimidating -3. This is where things get interesting. They're like two magnets, but instead of just sticking together, they have to negotiate their attraction. You can’t just slap a +2 and a -3 together and expect them to play nice forever. It's like trying to put a square peg in a round hole, but with electrical charges.
![[ANSWERED] the balanced ch equation that escri dissociation of lead II](https://media.kunduz.com/media/sug-question-candidate/20220429040612800424-4535300.jpg?h=512)
The universe, bless its orderly heart, likes things to be neutral. No net charge allowed! It’s like the bouncer at the club of chemistry, saying, “Alright, folks, let’s balance this out before we have a chemical riot on our hands!” So, our Lead(II) ions and our Phosphate ions have to figure out how many of them are needed to cancel each other out.
Think of it like a group project. Lead(II) is bringing +2 contributions, and Phosphate is bringing -3 contributions. How do we make the total score zero? Well, if we have one Lead(II) (+2) and one Phosphate (-3), we’re still at -1. Not quite there. If we have two Lead(II)s (+4 total) and one Phosphate (-3), we’re at +1. Still not zero. We're getting closer, but it's like trying to win a game of rock-paper-scissors where everyone keeps tying.
Here’s the magic number! We need enough positive charge to completely neutralize the negative charge, and vice versa. So, we need three of our charming Lead(II) ions. That’s 3 x (+2) = +6 total positive charge. Now, on the other side, we need two of our energetic Phosphate ions. That’s 2 x (-3) = -6 total negative charge.
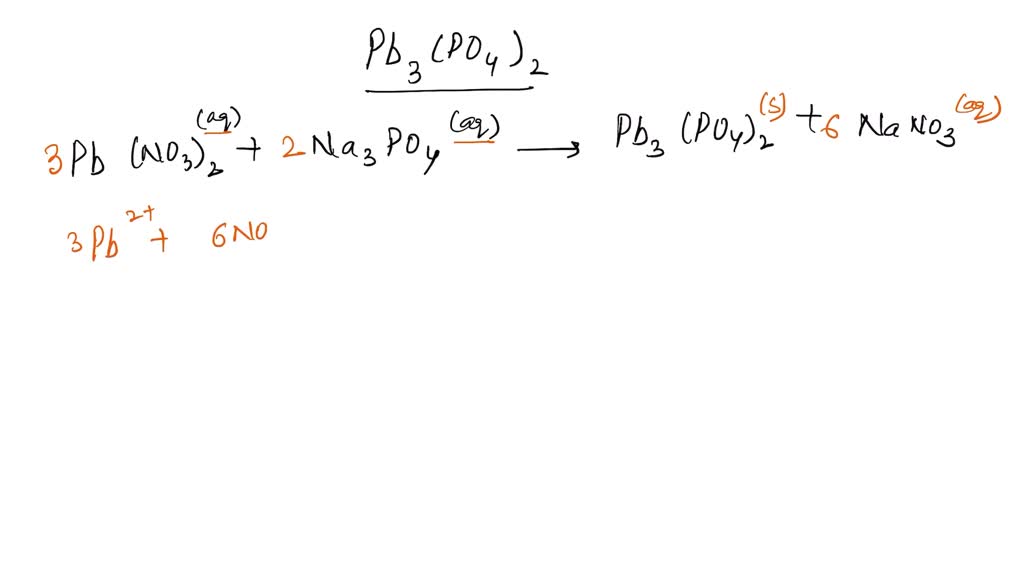
See that? +6 and -6! They’re a perfect match! They’ve canceled each other out, achieving the ultimate chemical goal: neutrality. It’s the chemical equivalent of finding your soulmate, but with more electrons.
So, how do we write this down? In the glorious language of chemistry, we use subscripts to show how many of each "character" are in our little chemical drama. We have three Lead (Pb) atoms, so we write a little ‘3’ after Pb. And we have two Phosphate groups, so we need to be careful. The Phosphate group (PO4) acts as a single unit. When we need more than one of these units, we have to put parentheses around it and then put the subscript outside.

So, our formula becomes: Pb3(PO4)2.
Ta-da! There it is! The grand, the magnificent, the… (drumroll please) …formula for Lead(II) Phosphate! It’s like finding the secret handshake to a super-exclusive club.
And what do these guys look like when they get together? Well, Lead(II) Phosphate is typically a white, crystalline solid. Think of it as tiny, sparkly, somewhat toxic confetti. It’s not exactly something you’d sprinkle on your birthday cake, unless you’re aiming for a very memorable and possibly life-altering celebration.

A surprising fact? Lead compounds, including Lead(II) Phosphate, have historically been used in things like paints and pigments because of their whiteness and opacity. Imagine a world where your walls were painted with this stuff! Thankfully, we’ve gotten a tad smarter about lead nowadays. We learned that while it might make your house look pretty, it’s also a bit of a party pooper for your brain cells.
So, the next time you hear about Lead(II) Phosphate, you can confidently say, "Ah yes, that's the one with the Pb3(PO4)2 formula! Three Leads doing a little electrostatic dance with two Phosphates, all to achieve that beautiful, neutral state. It’s chemistry, baby! And sometimes, it’s even a little bit funny."
Now, if you’ll excuse me, I think my imaginary latte has gone cold. Time for another adventure! Perhaps the secret formula for… something equally exciting. Maybe a compound that tastes like pizza. A chemist can dream, right?
