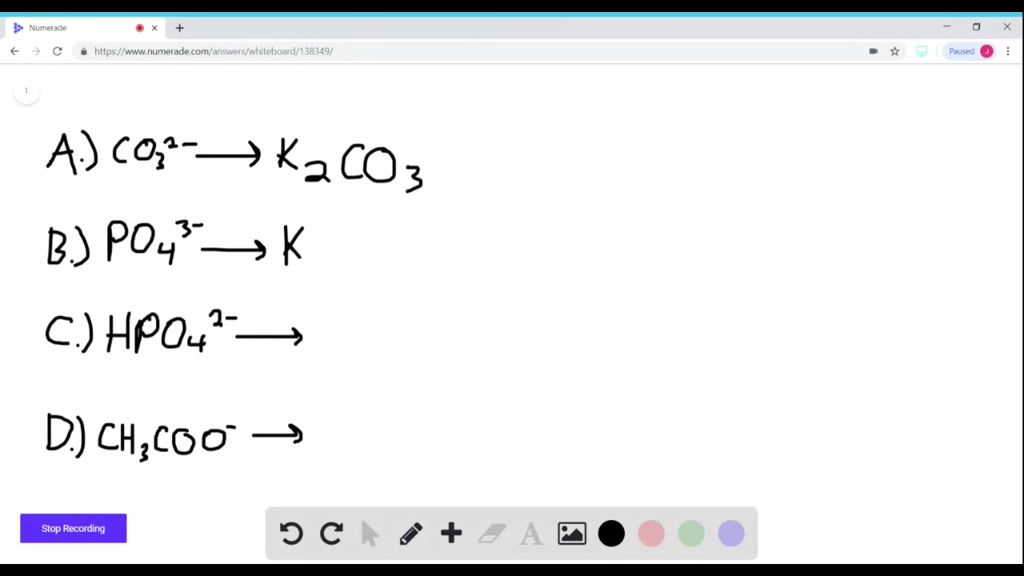
Enter The Compound That Forms Between Potassium And Carbonate
Hey there, curious minds! Ever stopped to think about the amazing things happening all around us, even in the most everyday stuff? Today, we're diving into something that sounds a little… well, like it belongs in a chemistry textbook, but trust me, it’s way cooler than you might think. We’re talking about the fascinating combo that forms when potassium and carbonate decide to get together. Yeah, I know, sounds a bit like a chemistry pickup line, right?
So, what exactly is this "compound" we're chatting about? Put simply, it's a chemical handshake between potassium (think of it as a friendly, eager metal) and carbonate (a group of atoms, kind of like a little molecular family). When they bond, they create a brand new substance with its own unique personality. We usually know it by its more common name: potassium carbonate, or sometimes, the old-school name, potash. Ever heard of that? It’s been around for ages!
Now, why should you care about potassium and carbonate doing their thing? Well, because this stuff is surprisingly useful and has a history as rich as a perfectly baked chocolate cake. It’s not just some obscure chemical; it’s a player in a lot of things we interact with, sometimes without even realizing it.
A Blast from the Past (and Present!)
Let’s rewind a bit. Potash, our friend potassium carbonate, isn’t some new invention. People have been using it for centuries! Think ancient civilizations, back when they were figuring out how to make soap, glass, and even fertilizers. They’d get it from the ashes of burnt wood, especially from hardwood trees. Imagine, using fireplace leftovers to create essential materials for daily life! Pretty ingenious, wouldn't you say?
This old-school method is kind of like how we might repurpose old cardboard boxes today to make something new. It’s all about finding value in what’s around you. The potassium compounds in the wood ashes, when mixed with water, would create a lye solution. This lye, containing potassium carbonate, was a game-changer for cleanliness and industry back then.
Even today, potassium carbonate is still a big deal. It’s got a role in making certain types of glass, like those fancy optical lenses or even the glass in your TV screen. It helps to make the glass clearer and more resistant to heat. So, next time you’re enjoying a crystal-clear view, you can give a little nod to potassium carbonate!
More Than Just Glass: The Versatile Nature of Potassium Carbonate
But it doesn't stop at glass, oh no. Potassium carbonate is like the Swiss Army knife of the chemical world. It’s also used in the food industry. Ever wonder how some baked goods get that lovely brown crust or how certain wines are clarified? Yep, potassium carbonate can be involved!
In baking, it can act as a leavening agent, helping things rise and become nice and fluffy. Think of it as a little helper that gives your cookies or cakes a gentle push upwards. And in winemaking, it can be used to help remove tartaric acid, which can sometimes make wine a bit too sour or cloudy. It’s like a tiny filter for your wine, making it smoother and more enjoyable. Pretty neat, huh?
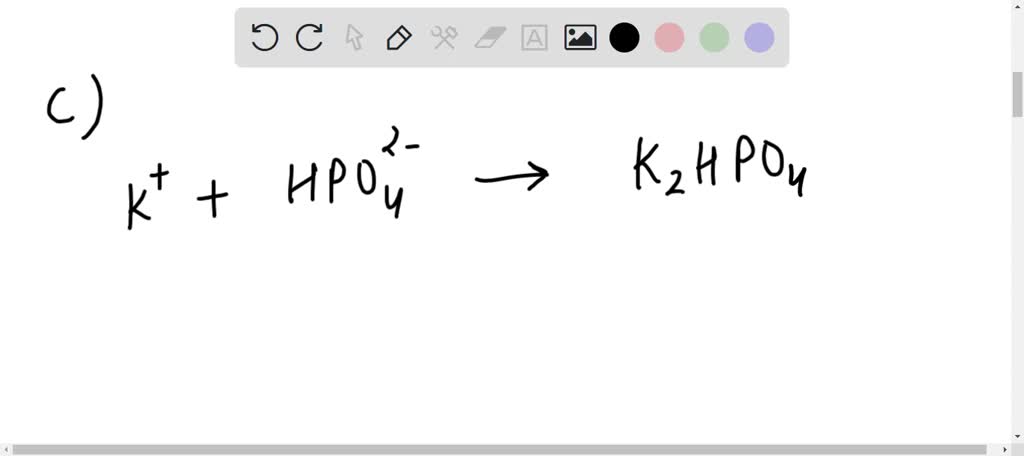
And let’s not forget about its role in agriculture. Remember that historical connection to fertilizer? It's still relevant! Potassium is a vital nutrient for plants, helping them grow strong and healthy. Potassium carbonate can be a source of this essential nutrient, ensuring our crops have what they need to flourish. It's like giving the plants a power-up drink!
What Makes It Special? The Chemistry Bit (Don’t Worry, It’s Easy!)
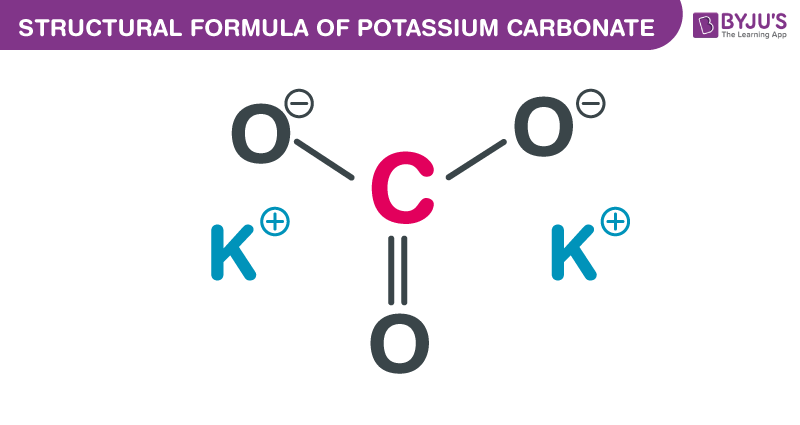
So, what’s the secret sauce behind potassium carbonate’s usefulness? It all comes down to its chemical properties. Potassium (K) is an alkali metal, meaning it’s very reactive and loves to form bonds. Carbonate (CO₃²⁻) is an anion, a negatively charged group of atoms. When these two meet, they form an ionic bond, creating a stable compound.

Potassium carbonate is typically a white, crystalline solid. It’s highly soluble in water, meaning it dissolves really easily, which is why it’s so handy for making solutions for various applications. Think about dissolving sugar in your tea – potassium carbonate does a similar trick, but for more industrial and agricultural purposes.
One of its most interesting characteristics is its alkalinity. When dissolved in water, it creates an alkaline solution. This alkalinity is what makes it useful for things like neutralizing acids, as we saw with the wine clarification. It’s like a gentle chemical balancer, helping to bring things into harmony.
Imagine you have a slightly too-sour lemonade. Adding a tiny bit of something alkaline could balance out the tartness. Potassium carbonate can do that, but on a much grander scale in different processes.
The Future is Potash-Powered?
As we continue to explore new materials and refine old processes, compounds like potassium carbonate remain essential. It’s a testament to how fundamental chemical principles can lead to practical applications that impact our lives in countless ways.
From the ancient world to modern high-tech manufacturing and sustainable agriculture, potassium carbonate has carved out its niche. It’s a reminder that even seemingly simple combinations of elements can unlock a world of possibilities. So, the next time you encounter glass, a tasty baked good, or even a healthy plant, take a moment to appreciate the silent, hardworking chemistry that might be at play!
It’s the everyday magic of molecules, you know? And the bond between potassium and carbonate is definitely one of those cool, quiet heroes. What other everyday chemical combinations do you think are secretly awesome? Let’s keep that curiosity buzzing!
