Enter A Balanced Equation For The Dissociation Of Ki
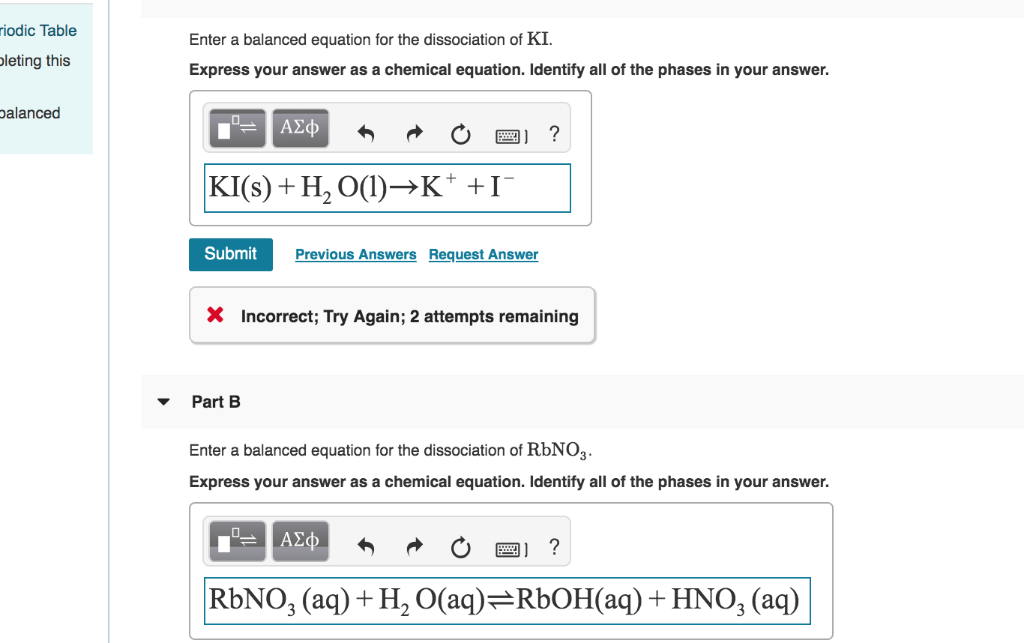
Ever wondered what happens when a simple salt, like Potassium Iodide (you might know it as KI), decides to take a little dip in some water? It’s not just a boring splash! Oh no, it’s a full-blown chemical party, and we're here to spill the beans on why this particular party is so darn entertaining. Forget fireworks and confetti; we're talking about atoms doing a happy dance!
So, what's the big deal with KI? Well, it's a classic. Think of it as the reliable friend who always shows up to the party. When you toss KI into water, something pretty cool happens. It doesn't just sit there looking smug. Nope, it decides to break apart. And when we say break apart, we mean it! It’s like a shy couple suddenly deciding to mingle with the crowd. This breaking apart is what scientists call dissociation. Sounds fancy, right? But really, it’s just the salt being a bit of a drama queen and splitting into its individual parts.
Now, for the main event: the equation! This is where we get to see the magic unfold on paper. Imagine KI as a neatly packaged little bundle. When it hits the water, the water molecules act like tiny, enthusiastic matchmakers. They gently nudge and pull at the bundle until it pops open. What pops out? Two totally different, but equally important, characters: a Potassium ion and an Iodide ion.
The equation that describes this grand separation is super straightforward, but don't let its simplicity fool you. It’s got a certain elegance, a quiet confidence that makes it special. It looks like this:
KI(s) → K+(aq) + I–(aq)
See that? It’s like a mini-story unfolding. On the left, you’ve got your starting point, the solid KI. The little ‘(s)’ tells us it's in solid form, like a little crystal. Then, the arrow (→) is the star of the show – it’s the symbol for change, for transformation. It’s saying, "And then, this happened!"

And what happened? On the right side, we have the stars of the dissociated party. We have K+(aq). That ‘K’ is the symbol for Potassium, and the ‘+’ sign tells us it's now carrying a little positive charge. Think of it as having just told a really good joke and feeling all puffed up. The ‘(aq)’ is like a little badge that says it's now dissolved and happily swimming around in water. It's in its aqueous phase, ready to mingle!
Then, we have I–(aq). This ‘I’ is for Iodide, and the ‘–’ sign means it’s picked up a negative charge. It’s like it’s just received a really nice compliment and is feeling a bit more grounded. And again, the ‘(aq)’ means it's also floating around in the water, all charged up and ready for whatever comes next.

Why is this so entertaining? Because it’s a visual representation of something invisible happening! We can’t actually see the KI breaking apart into charged particles. But this equation? It's like a secret decoder ring that lets us peek into the microscopic world. It’s a perfectly balanced whisper about how the universe works at its most fundamental level. It’s a tiny, yet mighty, testament to the order and predictability within chemistry.
What makes it truly special is its universality. This isn’t some rare, exotic chemical reaction. This is something that happens every day, in laboratories and, well, potentially even in the water you might be drinking (though probably in much smaller, less exciting amounts!). It’s a fundamental building block of understanding how ionic compounds behave when they meet water. It’s the foundational step for so many other cool chemical adventures.

Think of it as the opening scene of a great movie. You've got your main characters introduced, their initial state defined, and the inciting incident (the water!) kicking things off. The balanced equation is that perfect, concise summary that sets the stage for everything that follows. It's clean, it's logical, and it's surprisingly satisfying to see everything line up so neatly.
And the best part? You don’t need a super-powered microscope or a PhD in quantum physics to appreciate it. You just need a little curiosity and a willingness to see the extraordinary in the ordinary. This simple equation for the dissociation of KI is a friendly invitation to the world of chemistry. It’s a little wink from the molecular universe saying, "Hey, there's more going on here than meets the eye, and it’s actually pretty neat!" So next time you hear about Potassium Iodide, remember its little dissociation party. It’s a silent, elegant, and totally entertaining performance that happens all the time.
It’s like a chemical magic trick. You start with one thing, and poof! You get two (or more!) new things, each with its own personality and role to play. And it’s all perfectly accounted for. No disappearing acts, no sleight of hand – just pure, unadulterated chemical transformation. It’s the kind of wonder that makes you want to learn more, to see what other amazing transformations are waiting to be discovered. So go ahead, dive into the world of chemical equations. You might be surprised at how much fun you have!
