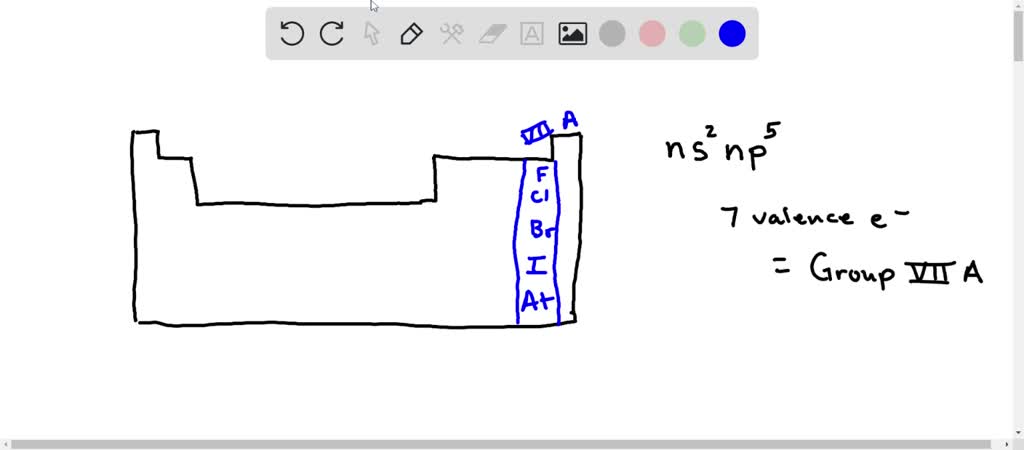
Elements With The Valence-shell Electron Configuration Ns2np5

Hey there, science curious folks! Ever stop to think about what makes the universe tick? It's all about these tiny, fundamental building blocks we call elements. And within those elements, there's a whole world of fascinating behavior happening right at the edges – in what scientists call the valence shell. Today, we're going to peek into a very special group of elements, those that have this particular setup in their outermost electron shell: ns²np⁵. Sounds a bit technical, right? But stick with me, because this is where some of the most exciting chemistry happens!
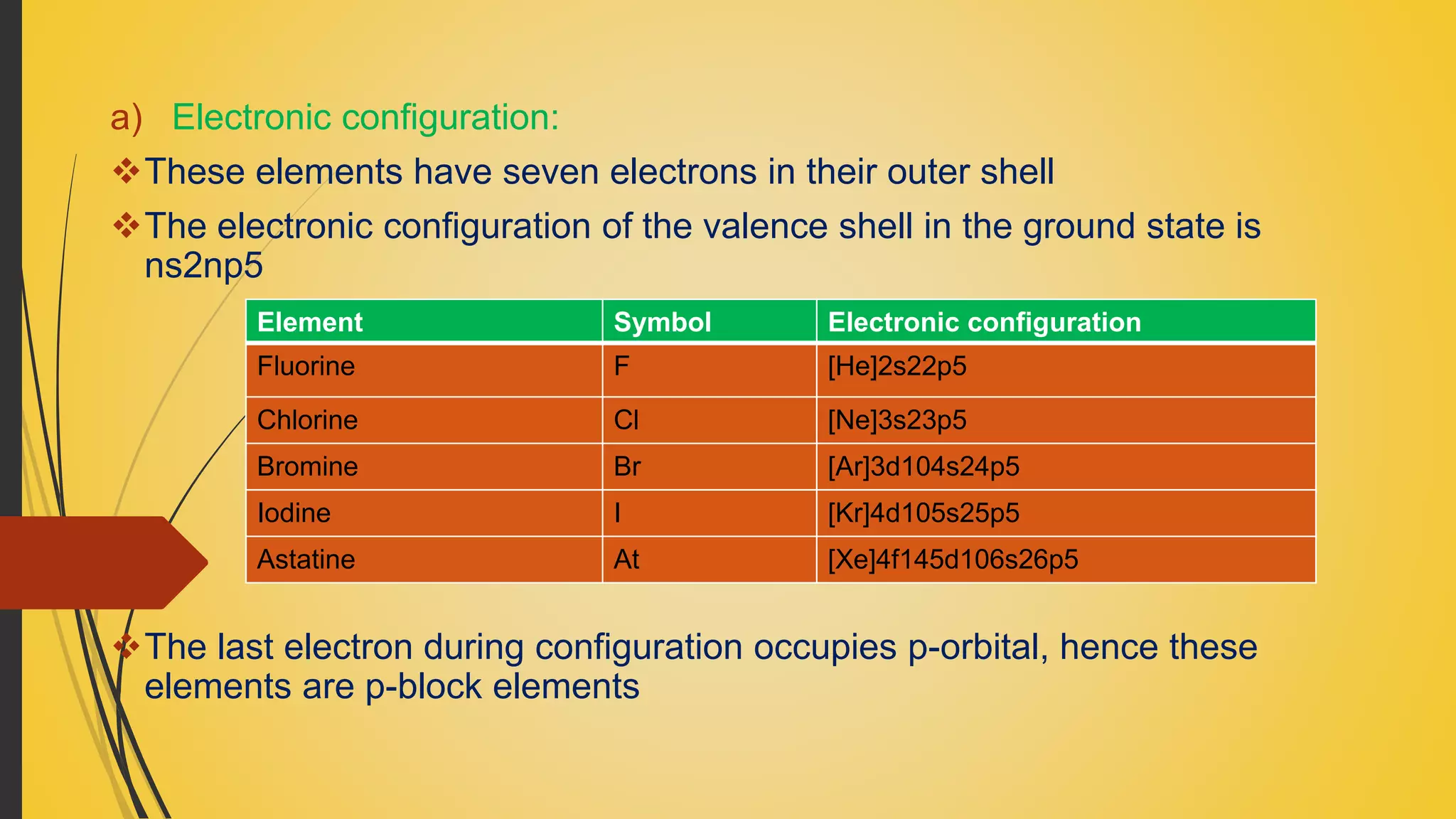
So, what does ns²np⁵ actually mean? Imagine an atom as a mini solar system. The nucleus is the sun, and the electrons are planets orbiting around it. These electrons are arranged in different "shells," like orbits at different distances. The valence shell is simply the outermost one, the one that gets to interact with other atoms. The 'n' just tells us which energy level or shell we're talking about (like the third orbit, or the fourth, etc.). The 's²' means there are two electrons in an 's' type orbital within that shell, and the 'p⁵' means there are five electrons in the 'p' type orbitals in the same shell. Put it all together, and you've got a grand total of seven electrons chilling in the outermost layer of these atoms.
Now, why is having seven valence electrons so special? Well, atoms are a bit like puzzle pieces. They love to be complete. And what does "complete" mean for a valence shell? Usually, it means having eight electrons. It's like they're aiming for a full house, a stable arrangement that makes them feel perfectly content. Think of it like this: you've got a party favor bag with eight slots, and these atoms have seven goodies already in there. They're just one item away from being perfectly packed!
The Hunger for One More
This "one electron short" situation is what makes the ns²np⁵ elements so incredibly reactive and interesting. They have a serious craving for that eighth electron. It's like being just a stone's throw from a delicious cookie jar, but not quite able to reach it. This intense desire drives them to do all sorts of things.
What do they do? Mostly, they go out and try to steal or borrow that missing electron from other atoms. They are the ultimate electron magnets! This is where things get really fun, because it leads to the formation of a huge variety of compounds. These aren't shy wallflowers; they're the life of the chemical party, actively seeking out partners to complete their electron shells.

And guess what? This group of elements is so important that it has its own special name: the halogens. You might have heard of some of them before. We're talking about elements like fluorine, chlorine, bromine, iodine, and astatine. They're all part of the same family, and they all share this 'electron-hungry' trait.
Meet the Halogens: A Colorful Bunch
Let's take a quick tour of some of these guys. Fluorine, for instance. It's the undisputed heavyweight champion of electron-stealing. It's so electronegative – that's the fancy word for "electron-pulling power" – that it can snatch an electron from pretty much anything. It's like the superhero who always gets the job done, no matter what.
Then there's chlorine. You've definitely encountered chlorine, even if you didn't realize it. It's used in swimming pools to keep them clean (and that distinctive smell!). It's also a key ingredient in things like bleach and disinfectants. Chlorine is a powerful oxidizer, meaning it readily removes electrons from other substances, making them react.
Bromine is a bit more of a liquidy character. It's a reddish-brown substance that's also used in various industrial processes and has applications in things like flame retardants. It's a bit less intense than fluorine but still a very determined electron seeker.
Iodine is another fascinating one. It's a solid at room temperature, but when heated, it turns into a lovely purple vapor. You might have encountered it as a tincture of iodine, used as an antiseptic. It's also essential for our thyroid health – a tiny amount of iodine in our diet is crucial!
And then there's astatine. This one is much rarer and pretty radioactive, so we don't interact with it as much in our daily lives. It's still a halogen, though, and would theoretically be just as eager to grab that extra electron if it had the chance.
Why So Reactive? It's All About Stability!
So, why is this constant quest for an electron so important? Because achieving that full, stable outer shell makes atoms much more content and less likely to react further. It's like finding your perfect comfy spot on the couch – you're settled and happy. When a halogen gains that electron, it becomes an ion with a negative charge (because it now has one more electron than proton). This negatively charged ion is called a halide ion.
For example, when chlorine (Cl) with its ns²np⁵ configuration gains an electron, it becomes a chloride ion (Cl⁻), which has a stable ns²np⁶ electron configuration, just like the noble gases. These halide ions are the building blocks for many familiar compounds. Think about sodium chloride – that's just table salt! It's formed when sodium (which is eager to lose an electron) gives its electron to chlorine (which is eager to gain one). It's a perfect match, a chemical handshake that creates something essential for life.
The ns²np⁵ configuration is essentially a blueprint for high reactivity. These elements are so close to achieving a noble gas configuration, the "happily ever after" for electron arrangements, that they are constantly seeking out ways to get there. It makes them incredibly useful in chemistry, forming the basis of so many substances we rely on every day.

More Than Just Salt: Everyday Encounters
Beyond table salt, the reactivity of halogens means they're involved in countless processes. They form compounds with metals, creating salts that are used in everything from batteries to food preservation. They form compounds with non-metals too, creating molecules essential for life, like those found in organic chemistry. Many plastics, refrigerants, and even medicines contain halogen atoms.
It's quite amazing to think that this simple arrangement of electrons – ns²np⁵ – leads to such a diverse and impactful group of elements. They are the great connectors, the ones that readily form bonds and create the complex world of molecules around us. They are the essential ingredients in so many of the things we use, see, and even are!
So, the next time you sprinkle salt on your fries, or notice the clean smell of a swimming pool, or even just think about the air you breathe, remember the ns²np⁵ configuration. It's a little bit of chemical magic, a constant striving for completeness that makes our world so wonderfully diverse and interesting. Pretty cool, huh?
