Elements With High Atomic Numbers Tend To Have _____.
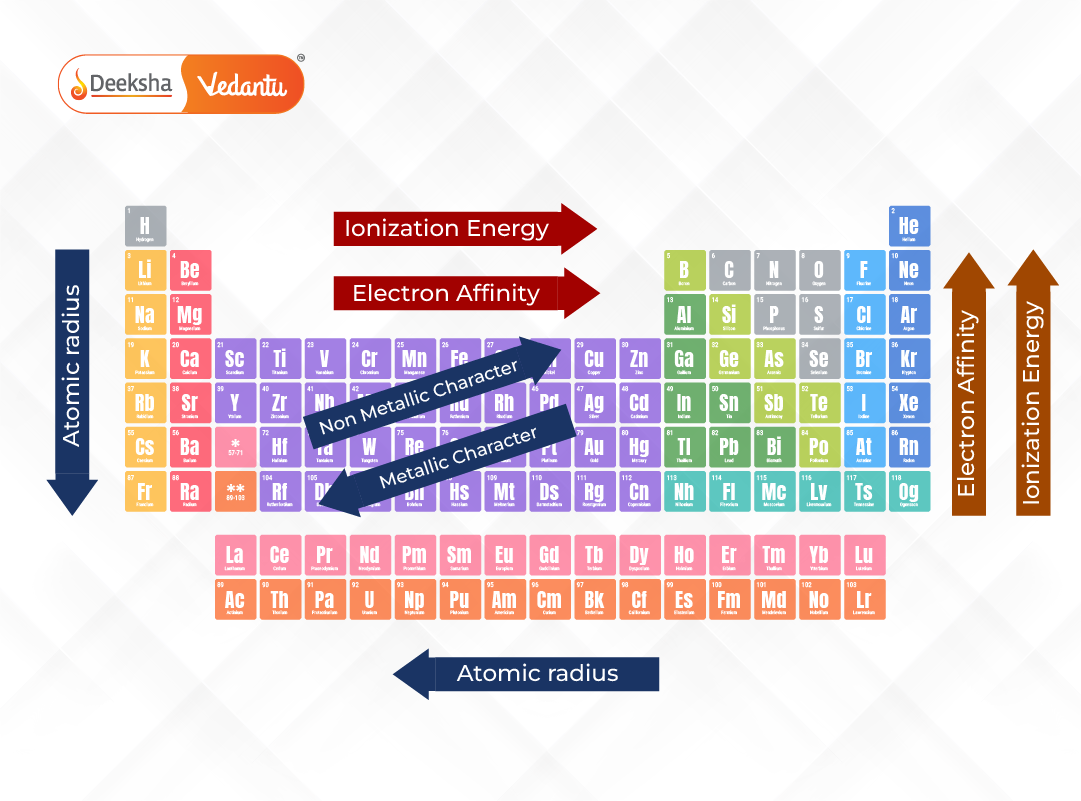
Hey there, science explorers! Ever wonder about the super-sized stars of the periodic table? You know, those elements that feel like they’ve been hitting the atomic gym a little too hard? Well, let me tell you, elements with high atomic numbers tend to have… well, they tend to have a whole lot of stuff packed inside them! Think of it like a giant, cosmic gumball machine. The higher the atomic number, the more chewy, flavorful gumballs are crammed in there!
Imagine trying to pack your entire life into a tiny backpack. It’s a squeeze, right? Now imagine trying to pack a whole city into that same backpack! That’s kind of what’s happening at the heart of these hefty elements. Their atomic nuclei are like bustling metropolises, with way more residents – protons and neutrons – hanging out than in their smaller, more nimble atomic neighbors.
So, what does all this extra "stuff" lead to? For starters, these elements are often heavyweights. Seriously, if you were to pick up a piece of gold versus a piece of helium (which is lighter than air, by the way!), you’d feel the difference instantly. Gold, with its impressive atomic number, feels substantial, solid, and frankly, a little bit luxurious. Helium? It’s practically a whisper in your hand.
These high-atomic-number elements are the stuff of legends and incredible inventions. Take uranium, for example. It’s got a whopping atomic number, and with all those particles crammed in its nucleus, it’s got a certain oomph to it. It’s like the element that ate its spinach and then some! This "oomph" is what makes it so useful in certain applications, where releasing that built-up energy can be a big deal. It’s the element that’s basically saying, "I’ve got more energy stored up than a toddler after a birthday party!"
And then there’s plutonium. Ooh, plutonium! Another one with a seriously high atomic number. It's like the secretive, powerful cousin in the element family. It’s got a lot going on inside, a real powerhouse. Because of its immense nucleus, it’s got a tendency to be a bit… well, unstable. Think of it like trying to balance a teeter-totter with a bowling ball on one end and a feather on the other. Eventually, something’s got to give, right? And for these elements, "giving" often means releasing some of that stored-up energy. It's the element that's constantly on the verge of a dramatic exit, but in a way that can be harnessed for incredible things.
But it’s not all about explosive energy. These massive elements are also often quite shiny and malleable. Have you ever seen a piece of platinum? It’s absolutely gorgeous, lustrous, and can be hammered into the thinnest sheets. It’s like the element that knows it looks good and isn't afraid to show it off! Its high atomic number means it’s got a lot of electrons zipping around, and those electrons are the rockstars of how elements interact and reflect light. They're the reason why these elements can look so dazzling and be so easily shaped into beautiful jewelry or critical components for technology.
Think about it this way: when you have a lot of tiny LEGO bricks, you can build all sorts of intricate and amazing structures. But when you have a few gigantic, super-dense bricks, you can build something incredibly robust and substantial. That’s the vibe of these high-atomic-number elements. They’re the building blocks for things that need to be strong, durable, and sometimes, downright awe-inspiring.

These elements are the titans of the atomic world, the ones that make you pause and say, "Wow, that’s a lot of nucleus!"
And get this, some of these high-atomic-number elements are so rare and so fascinating, they’re practically mythical. We’re talking about elements like flerovium or ununseptium. These are the VIPs of the periodic table, the ones you’d probably never encounter in your everyday life, but their existence is a testament to the incredible complexity that can arise from simply adding more and more particles to an atomic nucleus. They are the proof that nature, when given enough ingredients, can cook up some truly extraordinary substances. They’re like the limited edition, collector’s items of the universe!
So, next time you hear about an element with a really, really big number next to its name, just remember: it's probably dense, it might be powerful, and it’s almost certainly intriguing. These elements are the heavy hitters, the showstoppers, the ones that remind us of the sheer, unadulterated wonder of the universe and all the amazing things it’s capable of creating. They’re the elements that tend to have… well, they tend to have that certain je ne sais quoi, that extra something that makes them so incredibly special and useful!
