Elements That Have Properties Similar To Calcium

Hey there, science enthusiasts and curious minds! Ever wondered about those building blocks of the universe, the elements? We all know calcium, right? The stuff that makes our bones strong, keeps our hearts beating, and lets us crunch on popcorn without our teeth giving up the ghost. But what if I told you calcium isn't the only superstar in its league? Like a popular kid at school, calcium has some pretty cool pals who share a lot of its awesome traits. Let’s dive into the fascinating world of elements that are basically calcium's cousins, with a dash of their own unique sparkle!
So, what makes calcium so special? Well, it’s a member of a special club called the alkaline earth metals. Think of it like a VIP section in a really exclusive club. These guys are all located in Group 2 of the periodic table, just chilling next to the alkali metals (who are even more reactive, but we'll save that drama for another day). What’s the secret handshake for this club? They all have two electrons in their outermost shell, which they're just dying to get rid of. It's like they're holding a garage sale for electrons, and everyone’s invited!
This eagerness to shed those two electrons is the key to their similar properties. They’re all pretty reactive, meaning they like to jump into action and form chemical bonds with other elements. They’re also all metals, which means they’re good conductors of heat and electricity, and they have that shiny, metallic luster. Plus, many of them form compounds that are often found in minerals and rocks. So, when you're looking at a cool rock formation, you might just be looking at some of calcium's extended family!
First up on our elemental family reunion guest list is Beryllium (Be). Now, beryllium is the little guy of the alkaline earth metals, atomic number 4. It's often described as being less reactive than calcium, almost like the shy cousin at the party who hangs back a bit. But don't let that fool you; beryllium is a super important element, especially in high-tech stuff. Think airplanes, satellites, and even nuclear reactors! It's incredibly lightweight yet very strong, making it a superhero material. Its compounds can also be quite toxic, so it’s not something you want to be casually munching on. Definitely not popcorn material, that's for sure!
Beryllium’s compounds share some similarities with calcium’s. For example, beryllium oxide (BeO) is a refractory material, meaning it can withstand high temperatures. Calcium oxide (CaO), also known as quicklime, is famous for its use in construction and its ability to get really hot when mixed with water. So, while their specific applications differ, the underlying principle of forming stable, high-temperature compounds is a family trait.
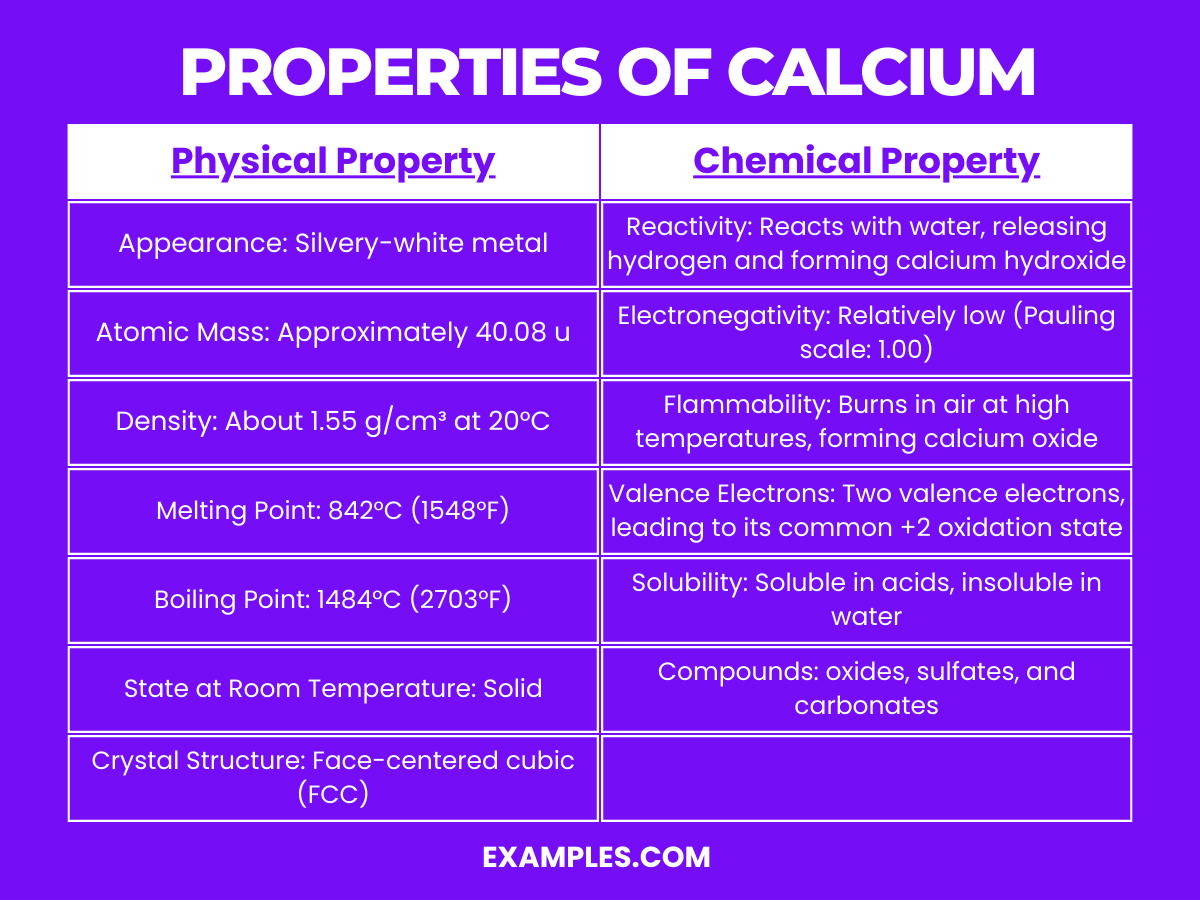
Next, we have Magnesium (Mg). Ah, magnesium! This is arguably calcium's closest buddy, and they’re often found together in nature. Magnesium is atomic number 12, just a few spots down from calcium. It’s also a vital element for life. You know those green leafy vegetables you're supposed to eat? They're packed with magnesium, which plays a crucial role in everything from muscle and nerve function to blood sugar control and blood pressure regulation. It's like the silent backbone of your health, working diligently behind the scenes.
Magnesium and calcium are so similar that they often compete for the same spots in biological systems. For instance, both are crucial for bone health, though calcium gets all the glory (typical!). Magnesium is also involved in muscle relaxation, while calcium is needed for muscle contraction. They work in a delicate dance, one tensing, the other releasing, to keep you moving. It's like a synchronized swimming routine for your muscles!
In terms of chemical properties, magnesium is also a reactive metal that readily forms ionic compounds. Think of magnesium oxide (MgO) and calcium oxide (CaO) – both are basic oxides and used in refractories and industrial processes. Magnesium burns with an incredibly bright white light, which is why it's used in fireworks and flares. Calcium, while not as flashy, also burns, but usually with a reddish-orange hue. Different strokes for different folks, I guess!

Moving along the periodic table, we encounter Strontium (Sr), atomic number 38. Strontium is a bit more of a distant relative, but it still shares some familiar traits. You might have heard of strontium in relation to toothpaste – strontium chloride is sometimes added to toothpastes to help with tooth sensitivity. It works by blocking the tiny tubules in your teeth that lead to the nerve endings. Clever, right?
Strontium compounds can also exhibit bright colors. Strontium nitrate, for example, is responsible for the vibrant red color in fireworks. Calcium, when burned, gives off a reddish-orange, so there's a color connection, even if the shades are different. Strontium also readily forms salts, just like calcium. While not as essential for human life as calcium or magnesium, strontium is found in some minerals and can be incorporated into bones, though in much smaller quantities.

Then there's Barium (Ba), atomic number 56. Barium is a bit of a heavier hitter in the alkaline earth metals. It’s known for its use in medical imaging, specifically a barium sulfate "milkshake" that patients drink before an X-ray of their digestive system. Barium sulfate is insoluble in water, so it passes through your system without being absorbed, coating your insides and making them visible on the X-ray. Pretty neat trick, though probably not the most delicious drink!
Barium compounds are also quite reactive. Barium oxide (BaO), like calcium oxide, is a basic oxide. Barium sulfate (BaSO4) is the less toxic, insoluble form that we use for medical imaging, while other soluble barium salts can be quite toxic. This variability in toxicity is a bit of a quirk, but the tendency to form solid, often insoluble compounds is a common thread with calcium. Barium's flame test color is a brilliant green, a bit further from calcium's red, but still a colorful character!
Finally, we have the big daddy of the alkaline earth metals, Radium (Ra), atomic number 88. Radium is, shall we say, the eccentric uncle of the group. It’s radioactive, which means it’s constantly emitting energy and transforming into other elements. This was once seen as magical, with radium being used in everything from luminous paint to "health tonics" back in the day. Of course, we now know that radioactivity is pretty dangerous, so radium's days of being a popular addition to your breakfast cereal are long gone. Phew!

Even with its radioactive nature, radium shares some fundamental properties with calcium. Because it has the same electron configuration in its outermost shell, it behaves chemically very similarly. For example, radium forms radium chloride (RaCl2), which is analogous to calcium chloride (CaCl2). Both are soluble salts. However, due to its radioactivity, radium is incredibly rare and its compounds are handled with extreme caution. It's the element equivalent of a rockstar who's a little too wild.
So, there you have it! From the lightweight champion beryllium to the radioactive enigma radium, calcium has a whole family of elements that share its core characteristics. They’re all in the same group for a reason – that delightful desire to shed two electrons, making them reactive, metallic, and capable of forming a wide array of compounds. They’re the backbone of many minerals, play crucial roles in biological processes, and even add a splash of color to our world (hello, fireworks!).
It’s amazing to think how these fundamental building blocks, with their subtle differences and striking similarities, contribute to the incredible diversity and complexity of the universe around us. From the strength in your bones to the sparkle in a gemstone, these elements are working their magic. So next time you hear about calcium, give a nod to its amazing family. They’re all part of the same grand, elemental party, making our world a little stronger, a little brighter, and a whole lot more interesting. Keep exploring, keep wondering, and keep finding the wonder in the everyday! You've got this!
