Elemental Sulfur Can Exist As Molecules With The Formula S8
Get ready to have your mind blown, because we're about to dive into the utterly fascinating world of a humble element that's way cooler than you might think. We're talking about sulfur, that yellowish, sometimes stinky stuff that pops up in all sorts of surprising places. And guess what? This elemental superstar has a secret life, a secret family, if you will.
Now, you might picture sulfur as just a bunch of individual atoms, zipping around on their own, doing their own atomic thing. Think of it like a bunch of solo singers, each belting out their own tune. But sulfur has a much more social, much more organized side.
It turns out that when sulfur atoms get together, they don't just mingle; they form incredible, intricate structures. And one of their absolute favorite ways to hang out is in a perfectly formed little circle. Imagine a group of friends deciding to hold hands and form a ring, but with atoms instead of hands.
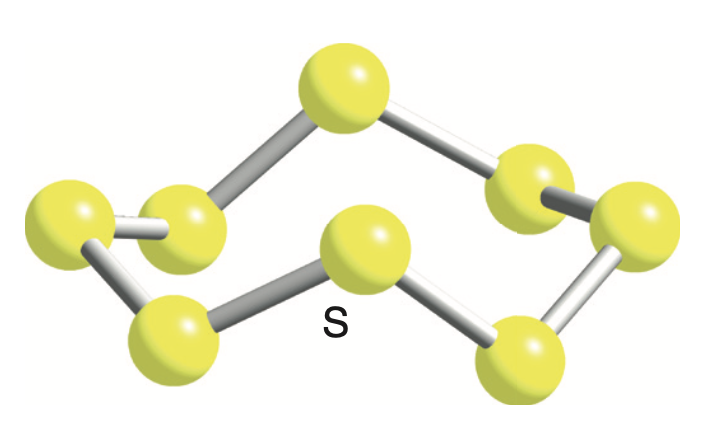
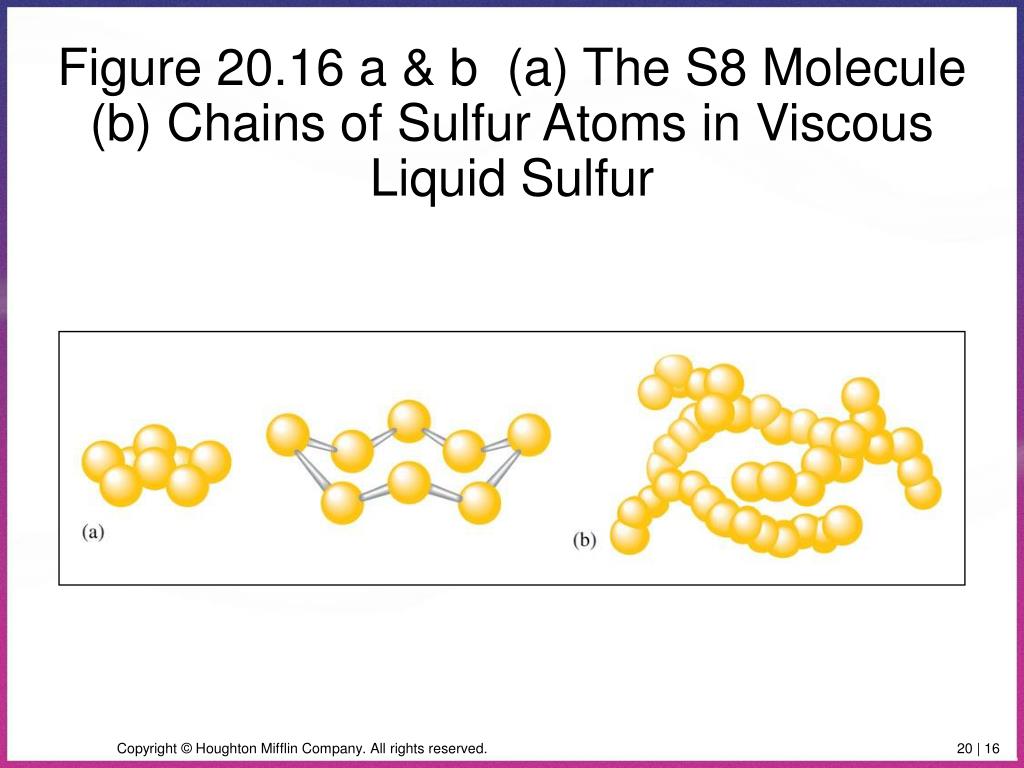
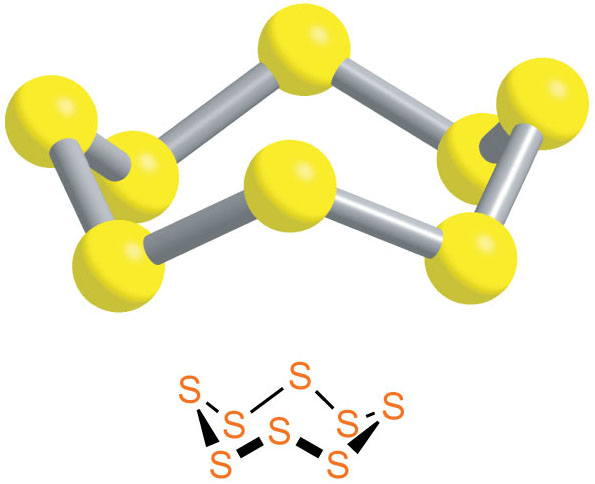
This special atomic ring has a very specific number of members: eight! Yep, you heard that right. Eight sulfur atoms, all holding hands in a beautiful, stable ring. This isn't just a casual get-together; this is a formal, established club, and its official name is S8.
Think of it like a super-exclusive, molecular dance troupe. The S8 molecule is the star performer, always appearing in this perfect octet. It’s like finding out your favorite band always plays their hits in a specific, magical order.
So, next time you hear about elemental sulfur, don't just picture a lone wolf atom. Picture this amazing, eight-membered ring, a perfectly arranged S8 molecule, ready to do its elemental thing. It’s a testament to how even the simplest elements can have such complex and elegant behaviors.
You might be wondering, "Why eight?" That's the beauty of chemistry, isn't it? Nature has its own quirky rules and preferred configurations. The S8 ring is particularly stable and happy. It’s like finding the perfect fit for a puzzle piece – it just clicks into place.
It's almost as if these eight sulfur atoms got together and said, "You know what? We're going to form the most awesome, most stable ring ever! We're going to be the S8 club, and everyone else can just watch in awe." And honestly, who can blame them?

This isn't some rare, microscopic oddity. The S8 form is actually the most common and stable form of elemental sulfur under normal conditions. It's like finding out that the most popular kid in school isn't just popular, they're also the reigning champion of something incredibly cool.
When you see a chunk of yellow sulfur, the vast majority of those sulfur atoms are happily chilling out in these perfect S8 rings. It’s like looking at a crowd and realizing everyone is wearing the same stylish uniform. They’re all part of the same magnificent molecular fashion statement.
Think about it: eight atoms, perfectly bonded, forming a crown-like structure. It’s a little bit like a tiny, atomic Ferris wheel, but instead of people, it’s sulfur atoms, spinning around in perfect harmony. They’re not just floating around; they’re in a beautifully organized, covalently bonded embrace.
And this S8 structure isn’t just pretty to think about; it has real-world implications. The way these molecules arrange themselves influences how sulfur behaves, how it melts, and how it reacts. It’s like knowing the choreography of a dance group – it dictates how they move together as a whole.
So, if you ever encounter sulfur, whether it's the faint smell from a burnt match (though that's more complex!) or in a laboratory setting, remember the hidden family. Remember the eight amigos, the sensational S8. They are the rock stars of the sulfur world.

It’s quite the feat of atomic engineering, isn't it? Nature, with its infinite wisdom and sometimes mischievous sense of humor, decided that sulfur's happy place was in a perfectly formed, eight-membered ring. It’s a molecular masterpiece, crafted by the universe itself.
Imagine these little S8 rings tumbling around, bumping into each other, a constant ballet of molecular motion. They're not chaotic; they're just enjoying their existence as this incredibly stable and beautiful structure. It’s like watching a flock of birds fly in perfect formation, only much, much smaller and made of pure element.
And the bond within the S8 ring is quite strong. This isn't a flimsy arrangement; these sulfur atoms are seriously committed to their ring. It’s like a lifelong friendship, a bond that’s tough to break.
So, the next time you hear about elemental sulfur, conjure up the image of these eight brave sulfur atoms, linked together in a perfect, majestic ring. They are the true representation of sulfur in its most elemental, most iconic form: S8.
It’s a little piece of chemical magic, a molecular marvel that’s just out there, doing its thing. And the fact that it's so common and stable just makes it even more special. It’s like discovering that your favorite superhero is actually incredibly humble and down-to-earth.
So let's give a round of applause, or perhaps a tiny, atomic cheer, for the fantastic S8 molecule! It's a shining example of how elements can come together to form structures that are both functional and undeniably cool. They are the ultimate atomic influencers.
Remember this: sulfur is not just a single atom. It's a community, a family, and their most celebrated family photo is of eight members, forming a perfect ring. That’s the story of S8, and it’s a story worth remembering because it’s simply awesome.
It just goes to show that even the most basic building blocks of our universe can have fascinating, hidden lives. The world of chemistry is full of these delightful surprises, and the S8 molecule is one of its most charming characters.
So, embrace the sulfur! Embrace the S8! It's a little bit of elemental wonder that's always been there, waiting to be appreciated. And now you know its secret, its beautiful, eight-membered secret. It's proof that sometimes, the most stable and significant things come in perfectly formed rings.
Think of it as a tiny, natural halo for each sulfur atom, or perhaps a molecular friendship bracelet. The S8 structure is a testament to the elegance of chemical bonding. It's a tiny, powerful unit that shapes how we understand this element.
The next time you encounter sulfur, give a little nod to its preferred molecular form. It’s more than just an element; it’s a team, a perfectly orchestrated group of eight. The legendary S8, a true wonder of the atomic world.
And this perfect ring is not just a novelty; it’s essential for sulfur’s stability and reactivity. It's like the secret recipe that makes your favorite cookie so delicious. The S8 structure is sulfur's secret sauce.
So, let your enthusiasm for sulfur soar! It’s an element that’s not only useful but also incredibly elegant in its most common form. The S8 molecule is a tiny, beautiful testament to the wonders of chemistry. It's an eight-sided story of elemental perfection.
It’s a reminder that even the most common substances can hold extraordinary secrets. The humble sulfur, in its magnificent S8 form, is a prime example of nature’s artistic flair. They are the eight musketeers of the periodic table.
So, let's celebrate the S8! This molecular masterpiece is a fundamental part of our world, a shining example of elemental beauty and stability. It’s a tiny, powerful circle of friends, forever bonded.
