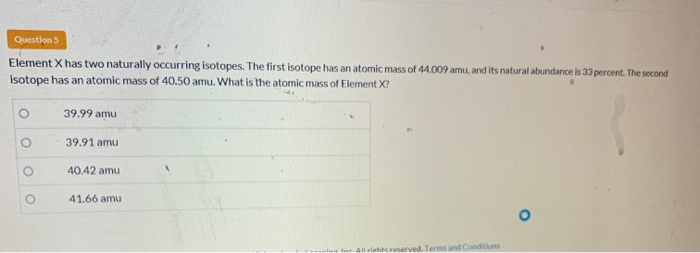
Element X Has Two Naturally Occurring Isotopes

Imagine you have a favorite candy bar, let’s call it the “Super Chocoholic.” It’s the best, right? You love its exact taste, its satisfying crunch, everything about it.
Now, what if I told you that some Super Chocoholic bars are just a tiny bit different? Not in a bad way, but like they have a secret ingredient that makes them… well, slightly heavier.
This is kind of like what happens with some of the fundamental building blocks of the universe, the things that make up everything from your coffee mug to the stars. Let’s call one of these building blocks Element X.
Element X is a pretty cool element, and it’s a key ingredient in so many things we interact with every day. Think of it as a star player on the elemental team, involved in all sorts of chemical reactions and holding things together.
But here's where the fun begins! Element X isn't just one thing. It’s actually like a family of two, two versions of itself that exist naturally. They are called isotopes.
Think of them like twins. They look very similar, they behave almost identically in most situations, but there’s a subtle difference that sets them apart. For Element X, this difference is in their weight.
Now, you might be thinking, "Weight? How big a difference are we talking about?" Well, it’s not like one is a feather and the other is a bowling ball. It's more like one twin has a slightly heavier backpack than the other.
These two isotopes of Element X are called X-10 and X-12. The numbers, 10 and 12, are a clue to their slight weight difference. They tell us how many tiny particles, called protons and neutrons, are zipping around in the center of each atom of Element X.
Most of the time, X-10 and X-12 are inseparable. They go through life together, bonding with other elements, forming molecules, and generally being the best of buddies.
However, their little weight difference can sometimes be a big deal, especially when scientists are trying to figure out how old something is or trace the path of materials through the environment. It’s like having a special tag on each twin that helps you tell them apart if you need to.
Imagine you're baking a cake, and your recipe calls for Element X. You grab a bag, and it's full of both X-10 and X-12, all mixed together. The cake will still turn out delicious because they are so similar in how they work.
But let’s say you're a detective trying to solve a mystery. You find a tiny speck of Element X at a crime scene. By carefully measuring the ratio of X-10 to X-12 in that speck, you might be able to figure out where it came from!
This is because different places, or different geological processes, might favor one isotope over the other. It’s like some parts of the world have more people who prefer carrying a slightly heavier backpack.
So, X-10 might be the more common twin, the one you see more often. It’s like the dependable, always-there sibling.
Then there's X-12, the slightly less common but equally important sibling. It’s like the one who might have a slightly more adventurous spirit, even if it’s just a little bit heavier.
The proportion of X-10 to X-12 in a sample of Element X is like a fingerprint. It’s unique and can tell us a lot about the sample’s history.
Scientists use this isotope fingerprinting for all sorts of amazing things. They can study ancient climates by looking at the isotopes in ice cores, or even track pollution by following the trail of specific isotopes in the air or water.
Think about it like this: if you found a specific type of glitter on someone’s clothing, and you know that glitter only comes from one particular art studio, you’ve got a strong clue! The isotopes of Element X act as that unique glitter.
It’s also quite heartwarming when you think about it. These two slightly different versions of Element X have been together since the dawn of time, sharing the same cosmic origins.
They’ve witnessed the formation of stars, the birth of planets, and the evolution of life. All while being intrinsically linked, two parts of a whole, albeit with a subtle difference.
And the fact that they exist in specific, predictable ratios in nature is a testament to the elegant laws that govern our universe. It’s a quiet symphony of particles playing out on a cosmic scale.
So, the next time you hear about isotopes, don't let the fancy word scare you. Just think of them as slightly different versions of the same thing, like twins with slightly different backpacks.
Element X, with its two natural isotopes, X-10 and X-12, is a perfect example of this fascinating phenomenon.

It reminds us that even in the seemingly simple building blocks of existence, there’s a depth and a story waiting to be discovered.
These isotopes are not just abstract scientific concepts; they are the silent storytellers of our planet’s past and the clues that help us understand the complex world around us.
And that, in its own subtle way, is pretty amazing, isn’t it?
The next time you marvel at a beautiful landscape or ponder the age of an ancient artifact, remember Element X and its two dutiful isotopes, silently contributing to the grand narrative.
They are a reminder that even the smallest differences can hold the biggest secrets, waiting to be unlocked by curious minds.
It’s like finding out your favorite song has a hidden track, or that a familiar path has a secret grove you’ve never noticed before.
So, here’s to Element X, and its two wonderfully distinct yet undeniably connected isotopes!
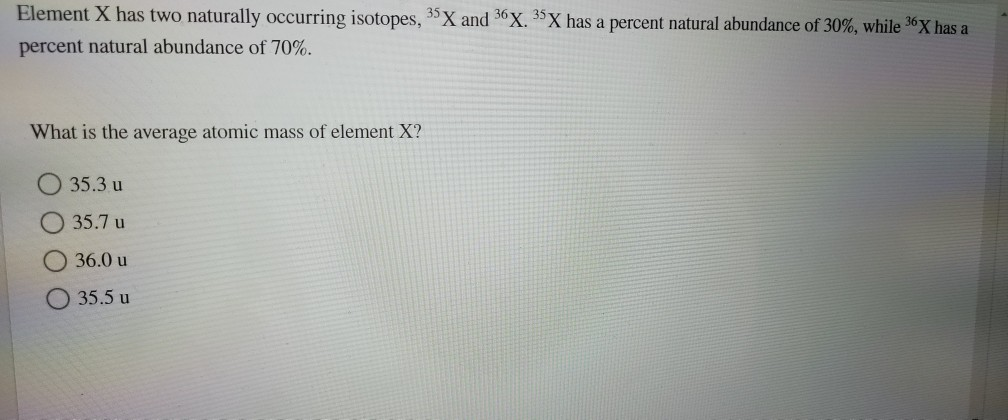
May their subtle differences continue to shed light on the mysteries of our universe for generations to come.
It’s a beautiful illustration of how even the most fundamental aspects of nature can offer a sense of wonder and a touch of the extraordinary.
The universe, much like a well-loved storybook, has its share of variations on a theme.
And these variations, like X-10 and X-12, are what make the book so rich and endlessly fascinating.
So, embrace the subtle differences, the slight variations, for they often hold the most profound and surprising insights.
Element X, in its isotopic duality, is a perfect example of this universal truth.
It's a gentle nudge to look closer, to appreciate the nuances, and to find joy in the unexpected variations that nature so generously provides.
