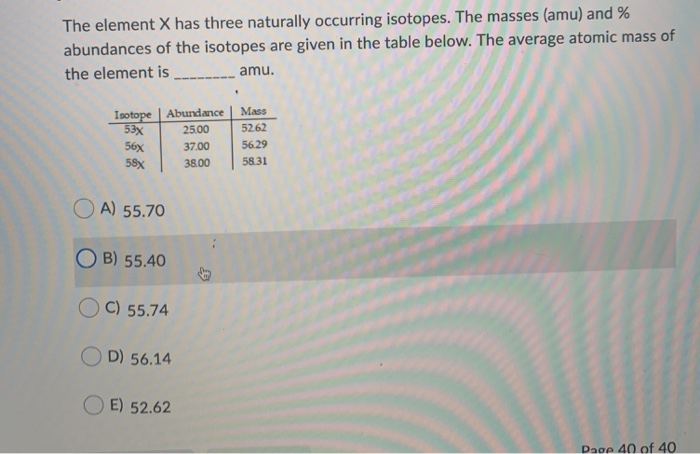
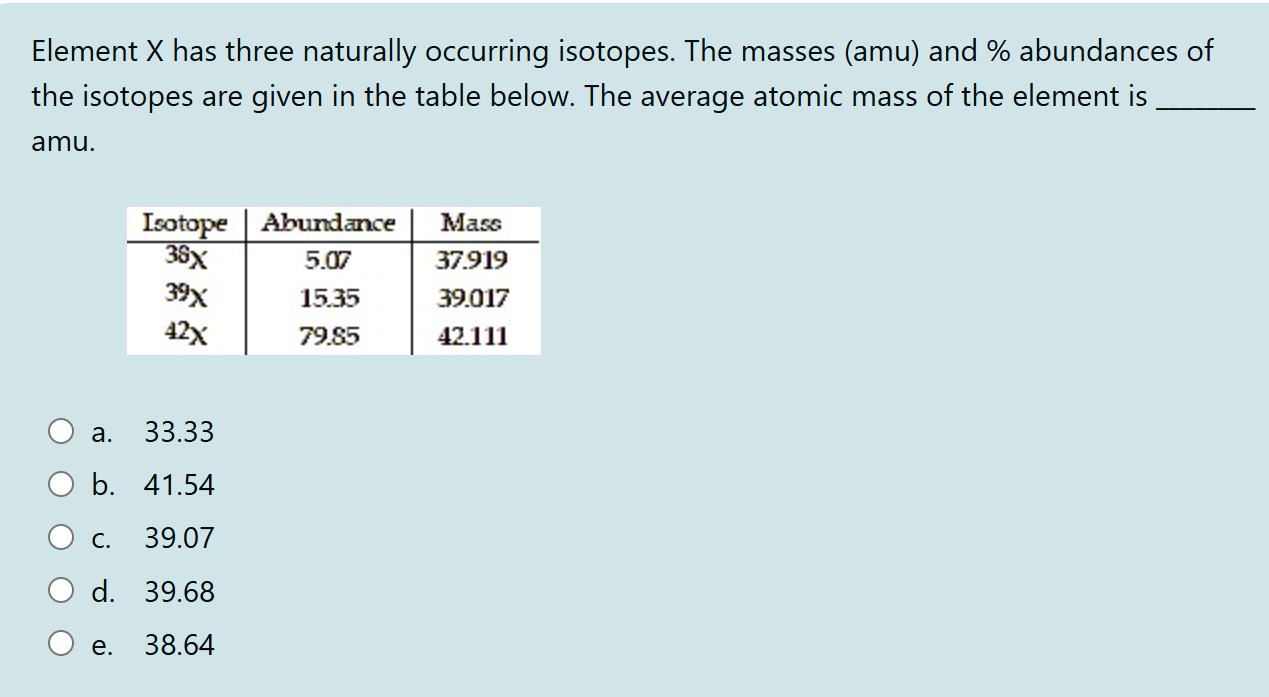
Element X Has Three Naturally Occurring Isotopes

Hey there! Grab your coffee, settle in. We're gonna chat about something super cool today – like, really cool. You know how sometimes you’ve got, like, different versions of the same thing? Think about your favorite song – you’ve got the original, maybe a remix, and a live acoustic version. They’re all the same song, right? But… different. Well, guess what? Atoms can totally do that too! It’s called having
So, what even is an isotope, you ask? Good question! It’s like this: you’ve got your basic atom. It’s got protons (those are positive little guys, chugging along) and electrons (negative, zipping around). The number of protons? That’s like the atom’s name tag. It never changes for a specific element. If it’s got six protons, it’s Carbon. Always. No ifs, ands, or buts. But then you have neutrons. These are the chill, neutral dudes hanging out in the nucleus. And they can vary. That’s where the isotope magic happens!
So, for Element X, we’ve got the standard X atom, let’s call it X-12 for now (we’ll get to the numbers in a sec). It’s got its usual number of protons. Then, BAM! We meet X-13. Same number of protons, but one extra neutron. It’s like the same person, but they’ve just had a really good snack. Slightly heavier, but still fundamentally them. And then, just to keep things interesting, there’s X-14. This one’s got two extra neutrons. Imagine our snack-loving friend from before, but they went back for seconds (and maybe thirds). Still our friend, just… a bit more substantial.
Why does this even matter, though? You might be wondering, “Is this just some nerdy science stuff for textbooks?” Nope! Isotopes are actually super important. They’re like secret agents of science, helping us figure out all sorts of things. Think about it like this: if you have three identical-looking rocks, but one is slightly heavier than the others, you can use that weight difference to tell them apart, right? Well, isotopes are kinda like that for elements. Their slight mass differences can be detected, and that’s a huge deal.
Let’s get a little more technical, but don’t worry, we’re keeping it light! The number after the element name – like that 12, 13, and 14 we were talking about? That’s the
Now, here’s the kicker: these different isotopes of Element X don’t really play by totally different rules chemically. Because, remember, the protons are what dictate how an atom behaves, how it bonds with other atoms. So, X-12, X-13, and X-14 will all generally do the same chemical dance. They’ll bond with oxygen to make oxides, with hydrogen to make hydrides, the whole shebang. It’s only their weight that’s the big difference. Imagine a set of identical twins. They look alike, they act alike, they probably even finish each other’s sentences. But one of them might be a little taller. That's kind of like our isotopes!
But why would nature bother making these different versions? It’s not like the universe is sitting around saying, “Let’s just add a neutron here for fun!” There are actually really good reasons. For Element X, these three isotopes are found in specific proportions in nature. It’s not like 99% is X-12 and then you find a tiny speck of X-14. No, no. They’re usually found in pretty significant amounts. This is super important because when we talk about the
So, the atomic mass on the periodic table for Element X is calculated by taking the mass of each isotope and multiplying it by its natural abundance (how common it is). Then you add all those up. It’s like saying, “Okay, X-12 makes up this much percentage of Element X, and it weighs this much. X-13 makes up this much, and it weighs this much…” and so on. And then you average it all out. That’s why the atomic mass of many elements isn’t a nice, neat whole number. It’s a testament to the fact that nature is usually dealing with a mix of isotopes! Pretty neat, huh?
Now, let’s imagine Element X is something like, say, Carbon. We know Carbon has isotopes like Carbon-12, Carbon-13, and even Carbon-14. Carbon-12 is super common, the backbone of life. Carbon-13 is also pretty common and used in research. And then there’s Carbon-14 – the famous one! It’s radioactive, meaning it decays over time. This is what scientists use for

So, for Element X, even though we don’t know its name yet (it’s a bit of a mystery element for our chat!), the fact that it has three naturally occurring isotopes tells us a lot already. It means that when you find Element X in nature, you’re not just finding one single type of atom. You’re finding a blend. And that blend, the specific proportions of X-12, X-13, and X-14, is unique to Element X. It’s like its fingerprint. Scientists can use these proportions to identify it, to study its origins, and even to track it in different processes.
Think about medical applications. Sometimes, certain isotopes are used in imaging or treatment. If Element X had an isotope that was particularly good for, let’s say, tracking blood flow, then knowing about all three of its naturally occurring isotopes could be crucial for developing the best medical tools. Maybe one isotope is too short-lived, or another is too heavy to be easily administered. The knowledge of all three allows for careful selection and utilization.
Even in geology, isotopes play a huge role. They can tell us about the formation of rocks, the history of the Earth’s climate, and even the processes happening deep within our planet. If Element X is found in certain minerals, the ratio of its isotopes could tell geologists when that mineral formed or under what conditions. It’s like reading a history book written in atomic particles! Seriously mind-blowing when you stop and think about it.

And what about environmental science? Tracking pollutants, understanding how substances move through ecosystems – isotopes can be incredibly valuable. If Element X is a component of a pollutant, scientists might use its isotopes to trace its journey from the source to its ultimate destination. This helps in understanding the impact of pollution and developing strategies to mitigate it. It’s all about following the clues, and isotopes are some of the best clues nature provides.
The existence of three isotopes for Element X also hints at the complex nuclear processes that led to its formation. In the vast cosmic furnaces of stars, elements are forged through nuclear fusion and other reactions. The relative stability and production rates of different isotopes are influenced by these astrophysical events. So, the specific mix of isotopes we find for Element X on Earth could even tell us something about where and how Element X was originally created in the universe. We’re talking stellar nurseries and supernova remnants here – the ultimate origin story!
It’s like having a family tree for an element. The parent elements, the conditions under which it was formed, the journey it took to get to Earth – isotopes can provide hints about all of this. It’s a constant unraveling of cosmic mysteries, one atom at a time. And Element X, with its three natural isotopes, is just another fascinating piece of that grand puzzle.
So, next time you’re looking at the periodic table, or just thinking about the world around you, remember that everything is made of atoms, and those atoms can have different flavors, so to speak. They can be isotopes! And for Element X, having three naturally occurring ones means it’s got a rich story to tell. It’s a little bit of diversity in the atomic world, and that diversity is what makes science so endlessly fascinating. It’s the little variations that often lead to the biggest discoveries. Who knew a few extra neutrons could unlock so many secrets?
It’s kind of like having a secret decoder ring for the universe. Each isotope is a slightly different key, and by understanding how these keys work and in what combinations they appear, we can unlock doors to understanding the past, the present, and even the future. Pretty powerful stuff for something so tiny, right? Element X is out there, quietly existing with its three isotopic personalities, contributing to the grand tapestry of matter. And we get to learn about it!
So, there you have it. Element X and its three natural isotopes. Not so scary, right? Just different versions of the same fundamental thing, each with its own slightly unique characteristic that, when looked at closely, can reveal a whole universe of information. It’s a reminder that even the smallest building blocks of our world have complexity and variety. And honestly? That’s what makes science so wonderfully, endlessly intriguing. Keep asking questions, keep sipping that coffee, and keep exploring the amazing world of chemistry. You never know what fascinating isotope stories you might uncover next!
