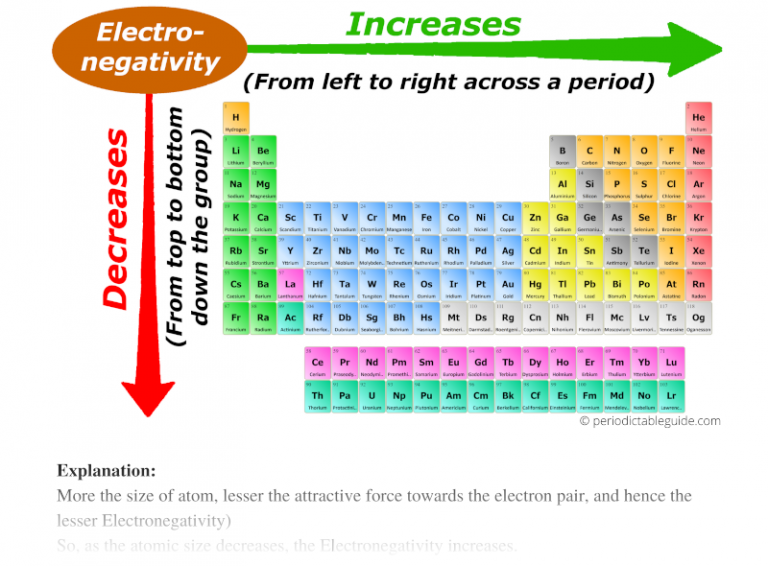
Electronegativity Increases As The Size Of The Atom Increases. Truefalse
Have you ever noticed how some things just naturally want to be close to each other? Think about magnets, or how your favorite socks always seem to find their way to each other in the laundry. Well, the tiny building blocks of everything around us, called atoms, have their own version of this clingy behavior.
These atoms are like little families, with a central part called the nucleus and little members called electrons whizzing around. The nucleus is kind of like the parent, holding onto the electrons with a special kind of invisible force.
Now, imagine an atom is like a tiny playground. The nucleus is at the very center, and the electrons are playing on different swings. The further away a swing is from the center, the less strongly it's held, right?
This is where things get a little topsy-turvy, and honestly, a bit hilarious. You might think, "Bigger atom, more space, electrons should be able to roam free!" But that's not quite how it works in the atomic world.
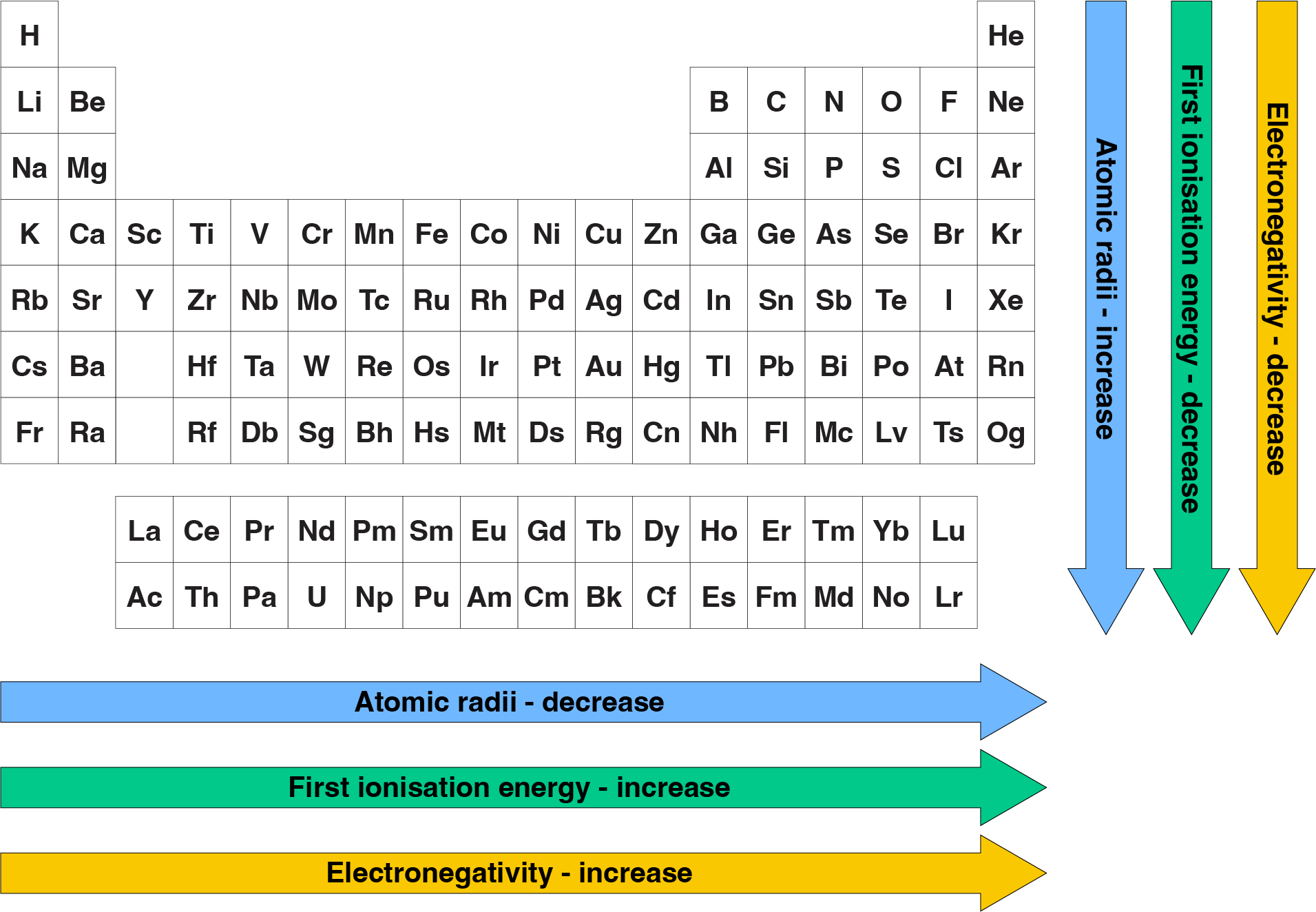
Let's talk about electronegativity. It sounds like a fancy word, but it's just a measure of how much an atom wants to grab onto electrons. Think of it as an atom's "electron greediness."
So, the big question for today is: Does electronegativity increase as the size of the atom increases? Is it a True or a False situation?
Prepare yourself for a surprising twist worthy of a superhero movie! The statement, "Electronegativity Increases As The Size Of The Atom Increases," is actually... False!
Wait, what? Didn't we just say bigger means more space and less pull? Yes, and that's exactly where the confusion comes in. It’s like expecting a giant's hug to be the strongest, but sometimes the most intense squeeze comes from someone much smaller.
Let’s dive into why this happens, and it’s quite a heartwarming story if you think about it. Imagine an atom is like a cozy little house. The nucleus is the fireplace, and the electrons are like family members who sometimes like to visit nearby friends.
In a small atom, the electrons are really close to the nucleus, the fireplace. They feel its warmth and pull very strongly. It's like being snuggled up right next to the fire on a cold night – very cozy and very hard to wander off.
Now, picture a really big atom. It's like a mansion with many, many rooms. The nucleus is still the fireplace, but it's now so far away from some of the outer rooms.
The electrons in these far-off rooms can't feel the warmth of the nucleus quite as intensely. They have more freedom to, well, explore. They might be tempted to wander into the garden or even visit a neighbor's house!
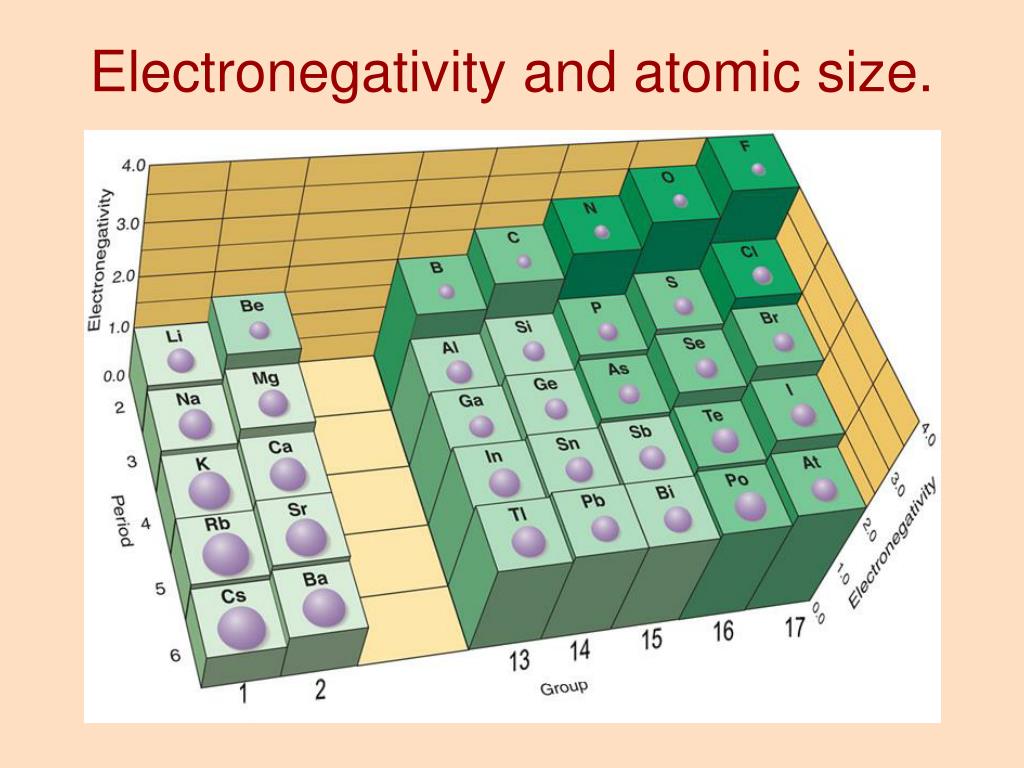
So, even though the big atom has more "stuff" and seems more imposing, the pull on its outermost electrons is actually weaker. It's like a parent trying to keep track of kids spread out across a huge park versus a small backyard.
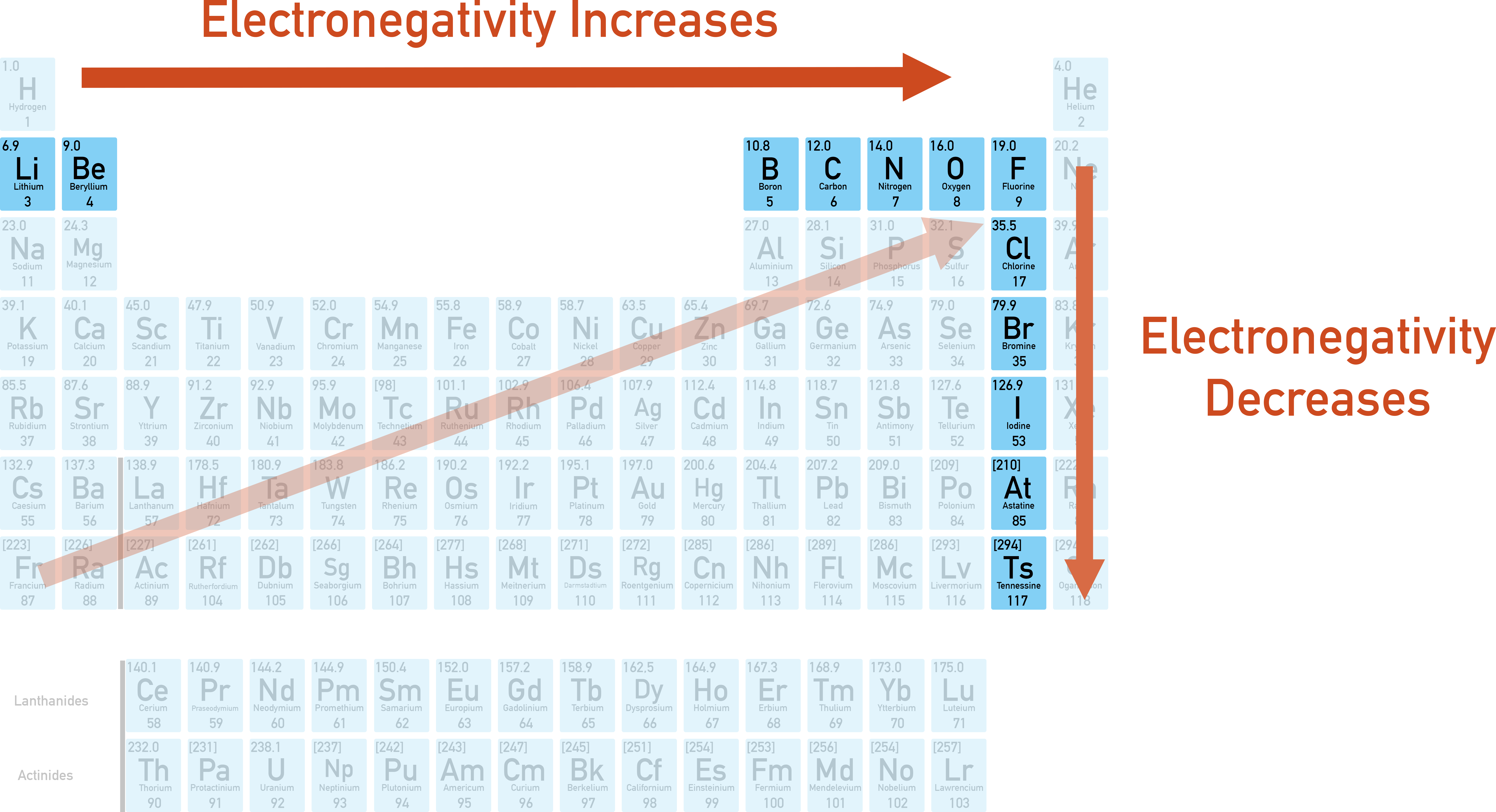
This is why smaller atoms are generally more electronegative. They have a stronger grip on their electrons because they're so close together. They're the ones who are more likely to say, "Hey, come back here! Don't go wandering off!"
Think of the tiniest, most energetic puppy. It wants to play with all the toys and keep them close. That's kind of like a small, electronegative atom.
Then you have a big, older dog. It might have a favorite toy, but it's less concerned about chasing down every single bouncy ball that rolls away. That’s more like a large atom with lower electronegativity.
It's a beautiful example of how appearances can be deceiving in the world of chemistry. We might expect bigger things to have a bigger impact, but in the case of electronegativity, it's the compact, tightly-knit atoms that pack the strongest punch when it comes to attracting electrons.
So, the next time you hear about electronegativity, remember this little story. It’s not about who’s the biggest, but about how close you are to the core of things.
It’s a reminder that sometimes, the most intense connections are found in the most intimate settings, not in the vast expanses.
The Fluorine atom, for instance, is famously tiny and incredibly electronegative. It’s like the ultimate electron-hoarder of the atomic world!
Compare that to an atom like Cesium, which is one of the largest and has very low electronegativity. It's much more laid-back about its electrons.
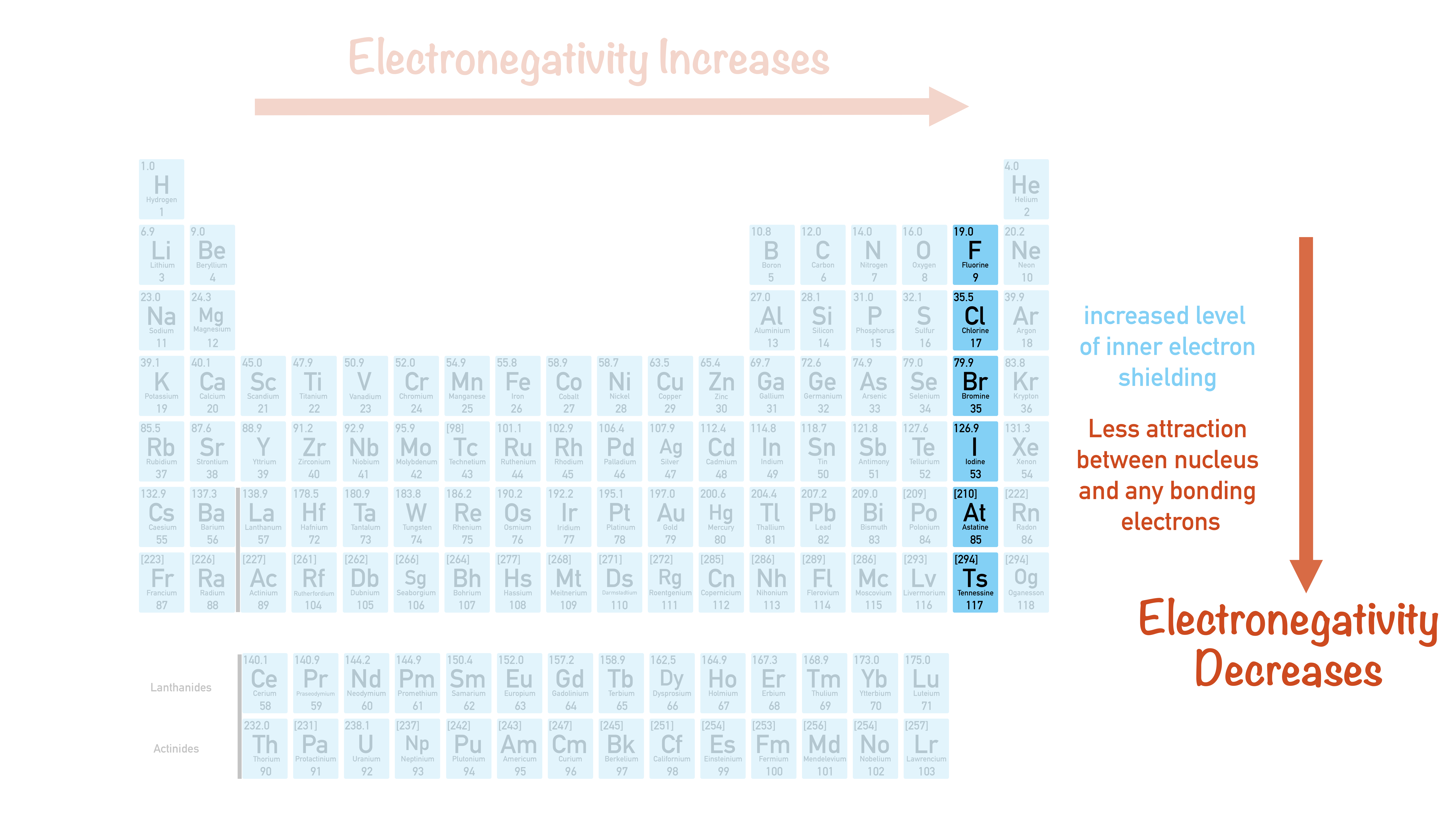
The trend is so consistent, it’s like a fundamental rule of nature, even if it seems a little counterintuitive at first glance.
This whole concept of electronegativity is super important because it helps us understand how atoms interact with each other to form molecules. It’s like knowing which kids on the playground are the most likely to share their toys (or, in this case, attract them).
It explains why water (H₂O) is such a great solvent, and why certain chemical reactions happen the way they do. It’s all thanks to these little electron tug-of-wars.
So, to recap: Electronegativity Increases As The Size Of The Atom Increases. This is False!
It’s the opposite! Smaller atoms, with their electrons nestled close to the nucleus, are the ones with the stronger pull and thus higher electronegativity.
Isn’t science wonderfully surprising? It’s full of little mind-benders that make you see the world in a whole new light.
Next time you’re holding a cup of coffee or looking at a leaf, remember the tiny, energetic atoms working together, each with their own unique brand of "electron desire."
It’s a beautiful dance of attraction and repulsion, all happening on a scale too small for our eyes to see, but with consequences that shape everything we experience.
So, let’s give a cheer for the small but mighty atoms and their surprisingly strong grips!
They remind us that power isn't always about size, but about proximity and intensity.
And that, my friends, is a truly heartwarming thought in the grand, complex, and often humorous universe of atoms.
