Electron Configuration Orbital Diagrams Worksheet Answer Key
Ever felt like you're staring at a puzzle, but the pieces are tiny, invisible, and have their own quirky personalities? That's often how electron configuration and orbital diagrams can feel. But what if I told you there's a secret society of these little electron guys, and their living arrangements are actually kind of hilarious and surprisingly heartwarming?
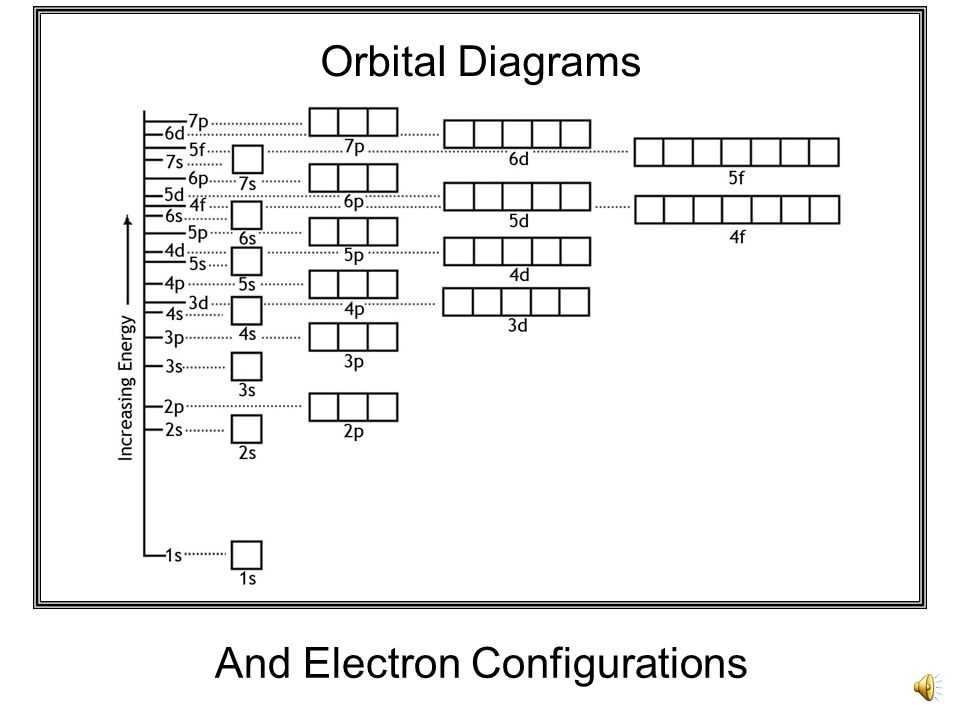
Think of atoms as bustling little apartment buildings. Each building has floors, and on those floors, there are different types of rooms where the tiny residents – the electrons – hang out. These rooms are called orbitals. Now, these electrons aren't exactly the tidiest tenants. They have rules, and understanding those rules is what electron configuration is all about.
Imagine the first floor is pretty basic. It's got one cozy little room, the 1s orbital. It's like a studio apartment for electrons, and it can only fit two. If you've got more than two electrons trying to cram into this little space, well, things get a bit awkward. It’s a bit like trying to fit your entire family into a Mini Cooper – it’s just not going to work out smoothly.
Then, you move up to the second floor. This floor is a bit more spacious and has more variety. There's another s orbital, just like the one on the first floor, but it's a bit bigger. And then, things get interesting! You get a whole set of three new rooms called p orbitals. These are like having a few extra bedrooms, perfect for sharing. Each of these p orbitals can hold two electrons, so the second floor can comfortably house a few more residents.
Now, the really fun part, and where things get a little wild, is when you start to look at how these electrons actually behave. They don't just randomly pick a room. Oh no, they have preferences and follow strict etiquette, thanks to the Pauli Exclusion Principle and Hund's Rule. It's like a very polite, but sometimes competitive, game of musical chairs.

Let's talk about Hund's Rule first. Imagine those three p orbitals on the second floor. If you have three electrons trying to move in, they won't pair up in one room. Instead, they'll each take their own p orbital first. It's like saying, "Let's all get our own space before we start sharing." They're giving each other a little elbow room. Only when you have a fourth electron do they start pairing up. It's a surprisingly considerate way to handle things, almost like they're saying, "Okay, this one's a bit crowded, but you can join me."
And then there's the Pauli Exclusion Principle. This one's a bit like a strict landlord's rule: each orbital can only hold two electrons, and they have to have opposite "spins." Think of spins as electrons having their own unique way of twirling. One spins clockwise, and the other spins counter-clockwise. They can't both spin the same way in the same room. It's the ultimate form of social distancing in the atomic world!
So, when we talk about orbital diagrams, we're essentially drawing little boxes to represent these orbitals and using arrows to show the electrons and their spins. An empty box means no electron is home. A box with one arrow is like a single occupant. And a box with two arrows, one pointing up and one down, means the orbital is full and those two electrons are cohabiting peacefully (or as peacefully as electrons can!).

The joy of an Electron Configuration Orbital Diagrams Worksheet Answer Key isn't just about getting the right answers. It's about seeing this complex, invisible world come to life. It’s about understanding the delightful little quirks of these fundamental building blocks of everything. You start to see how even the smallest things have their own intricate social structures and rules, and it’s surprisingly beautiful.
Think about it: the same rules that govern the electrons in your phone, the water you drink, and even the stars in the sky are being played out in these tiny atomic apartments. It’s a universal language of order and interaction. And when you finally crack the code on an orbital diagram, and see all those boxes filled with perfectly paired and unpaired arrows, there's a little thrill of understanding. You've peered into the heart of matter and seen its charmingly organized chaos.

So, the next time you encounter an electron configuration problem or an orbital diagram, don't just see it as a chore. See it as a peek into a microscopic world filled with quirky residents, surprising etiquette, and a fundamental dance of attraction and repulsion. It’s a story of how everything, from the smallest atom to the largest galaxy, is built on these fascinating, often humorous, and undeniably heartwarming principles.
And the answer key? That’s just your friendly guide, helping you navigate the apartment complex and understand which electron is crashing where and how they’re all getting along (or not!). It’s a little map to the most fundamental neighborhood in the universe, and it’s more interesting than you might ever have imagined.
It’s a bit like learning the secret handshake of the universe.
