Electron Configuration And Orbital Notation Worksheet
Alright, space cadets and curious minds! Ever looked at the universe and thought, "Wow, that's a lot of stuff!"? Well, guess what? That stuff is made of teeny-tiny building blocks called atoms. And inside these atoms, things get even crazier, like a microscopic rave party happening all the time!
Today, we're diving headfirst into the dazzling world of electron configuration and its super-cool sidekick, orbital notation. Think of it as the cosmic choreograph of electrons, dictating exactly where these energetic little guys hang out within their atomic homes. It's like figuring out the seating chart for the universe's most exclusive nightclub!
So, what's this magical "electron configuration" we're talking about? Imagine an atom is a mansion, and electrons are the party guests. Electron configuration is basically the address book that tells you which floor and which room each guest is staying in. No more electrons wandering the halls aimlessly, right?
And then there's orbital notation. If electron configuration is the address book, orbital notation is the detailed floor plan with little arrows showing which way each guest is facing. It's all about those tiny, invisible "rooms" where electrons can chill. These rooms are called orbitals, and they're not your grandma's dusty attics, oh no!
These orbitals have some funky shapes, like little balloons, dumbbells, or even more complicated pretzels. And each orbital can only hold a maximum of two electrons. It's like they have a strict "two-person per room" policy at the atomic party. No overcrowding allowed!
Now, you might be thinking, "This sounds complicated!" And honestly, at first glance, it can seem a bit like deciphering an alien language. But that's where our trusty Electron Configuration and Orbital Notation Worksheet comes swooping in like a superhero in training!
This worksheet is your personal atomic cheat sheet, your Rosetta Stone to the electron universe. It breaks down the seemingly complex rules into bite-sized, chewable pieces. We're talking about making this whole electron-addressing business feel as easy as ordering pizza!

Let's talk about those orbital shapes for a sec. We have the super simple, spherical ones called 's' orbitals. Imagine a perfect, cozy little sphere. These are the bachelor pads of the electron world, always ready to welcome up to two guests.
Then we have the 'p' orbitals. These are a bit more flamboyant, shaped like dumbbells. They come in sets of three, pointing in different directions, like a tiny, atomic yoga pose studio. They're perfect for those electrons who like a bit more personal space.
And don't even get me started on the 'd' and 'f' orbitals. These are the penthouses and private jets of the atomic mansion, with even more intricate shapes and multiple rooms. They're where the really sophisticated electrons hang out, probably discussing quantum physics over tiny electron cocktails.
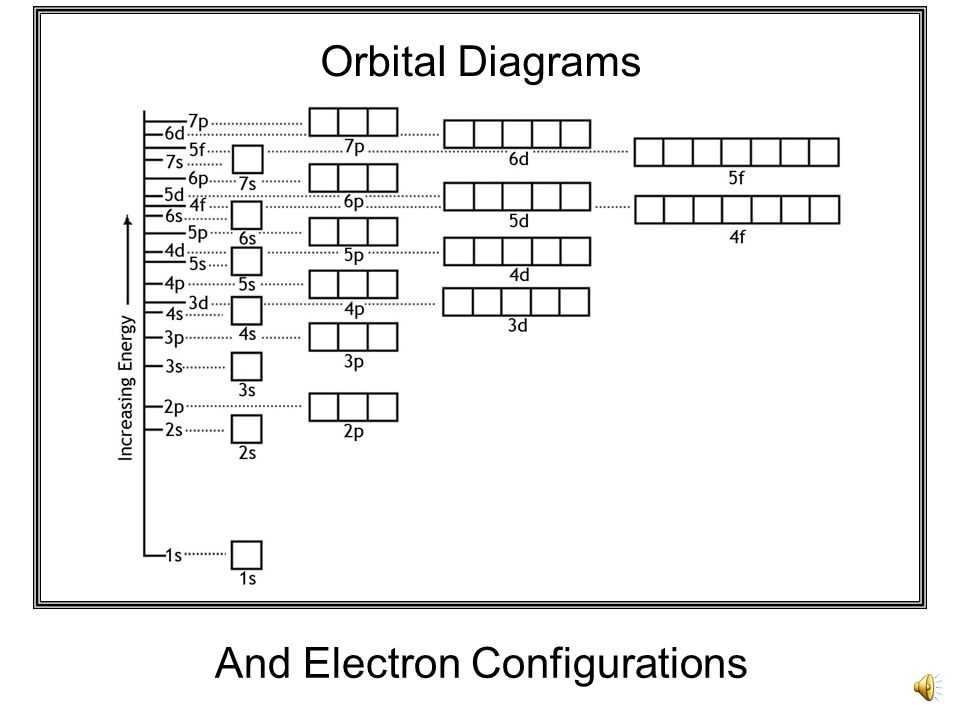
The fun part is figuring out the order these orbitals get filled. It's not just a free-for-all; there's a definite hierarchy! Think of it like boarding an airplane. The closest seats to the front get filled first, then the next row, and so on. Electrons follow a similar principle, filling the lowest energy levels first.

This is where you start seeing cool patterns. You'll learn about the Aufbau principle, which basically means "build up." You fill the lowest energy orbitals first. It's like stacking LEGOs, you start with the bottom layer!
Then there's Hund's rule. Imagine electrons are like kids in a classroom. They prefer to have their own seat before they start sharing. So, in a set of orbitals like the 'p' orbitals, each one gets one electron before any orbital gets a second one. They're a bit shy, these electrons!
And finally, the Pauli exclusion principle. This one's all about those two electrons per orbital. They have to have opposite "spins," like two dancers spinning in opposite directions. It's a cosmic dance of electrons, and they can't both be spinning the same way in the same orbital. Think of it as an atomic "no-fly zone" for identical electron pairs in the same spot.
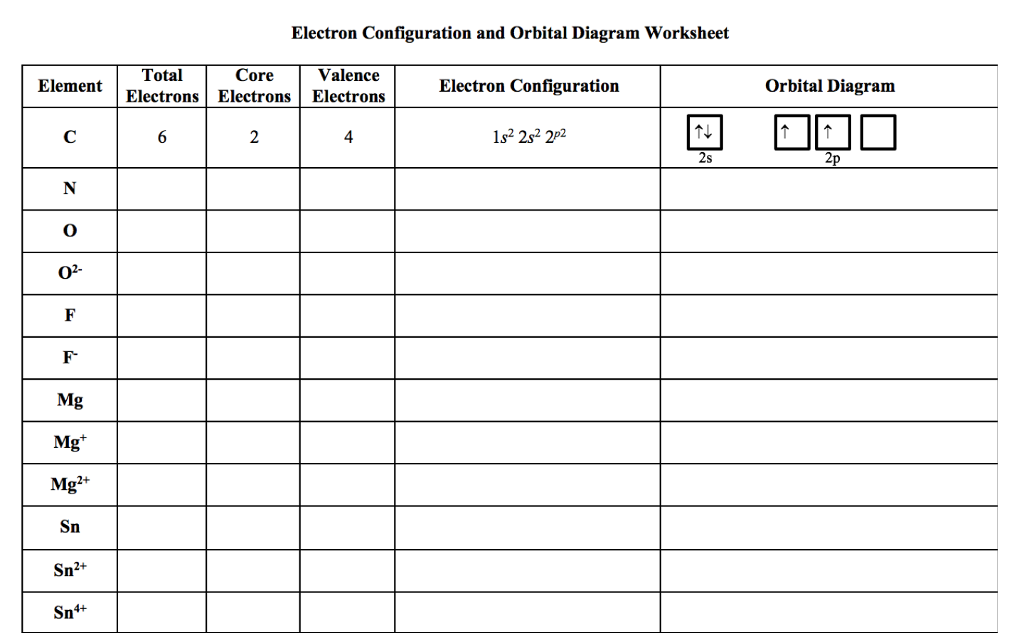
Our worksheet guides you through all of this. You'll be writing out electron configurations like a pro in no time. For example, for a simple atom like Oxygen (which has 8 electrons), its electron configuration might look like this: 1s² 2s² 2p⁴. See? It's like a secret code, and you're about to crack it!

The '1s²' means the first energy level, in the 's' orbital, has two electrons. The '2s²' means the second energy level, in its 's' orbital, also has two electrons. And the '2p⁴' means the second energy level, in its 'p' orbitals, has four electrons spread out.
Then, when you move to orbital notation, you'll draw little boxes for each orbital and arrows for the electrons. For Oxygen, that '2p⁴' would have three 'p' boxes, with arrows showing one electron in each of the first three boxes, and then the fourth electron pairing up in the first 'p' box. It's like drawing out the occupancy of those atomic dance floors!
This might sound like a lot, but trust me, the worksheet makes it ridiculously easy. It has plenty of examples, step-by-step instructions, and practice problems that will make you feel like a true electron whisperer. You’ll go from scratching your head to confidently scribbling these notations.
Why is this even important, you ask? Well, understanding electron configuration and orbital notation is like knowing the secret handshake of chemistry. It explains why atoms bond the way they do, why elements have certain properties, and essentially, why the universe behaves the way it does.
It's the foundation for everything from building amazing new materials to understanding how life itself works. Think of it as the atomic blueprint for reality! Without this knowledge, we'd be fumbling in the dark, trying to build the universe with no instructions.
And the best part? You don't need a PhD or a supercomputer to grasp these concepts. With a good worksheet and a sprinkle of enthusiasm, anyone can become an electron configuration ninja! It’s like learning to ride a bike – a little wobbly at first, but soon you’ll be cruising!
So, grab your pencils, unleash your inner scientist, and get ready to conquer the electron universe. This Electron Configuration and Orbital Notation Worksheet is your ticket to understanding the fundamental building blocks of everything you see, touch, and even imagine. It’s going to be an atomic adventure you won't forget!
Prepare to be amazed as you unravel the mysteries of electron placement, becoming a maestro of atomic organization. It’s a journey that’s both incredibly educational and surprisingly fun. Let the electron configuration quest begin!
