Effective Nuclear Charge For 3d Electron In Vanadium
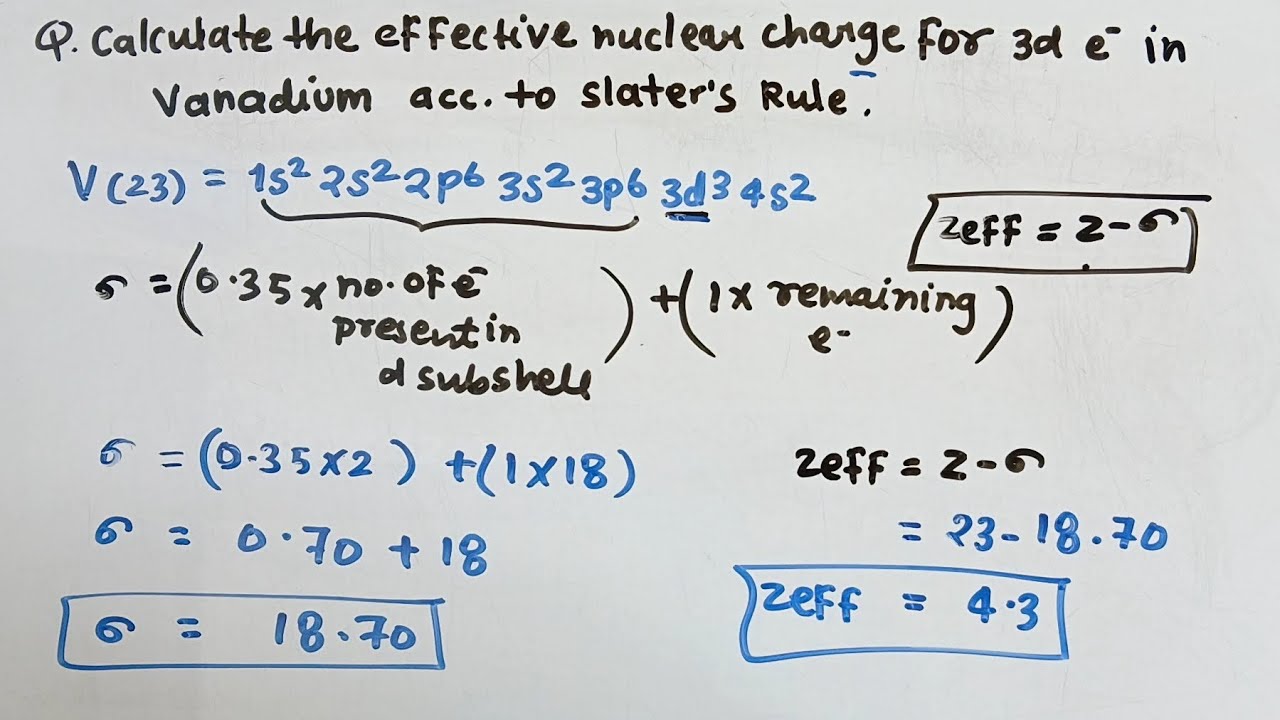
So, picture this. I’m wrestling with a particularly stubborn chemistry textbook, the kind that feels like it was written by a committee of super-intelligent but profoundly joyless robots. We’re talking about electron configurations, and for some reason, the example is Vanadium. Vanadium! This element, which sounds vaguely like a brand of fancy soap or maybe a superhero sidekick, is suddenly the star of my academic misery. And there it is, buried deep within a paragraph that’s drier than a desert in August: the concept of effective nuclear charge.
I blinked. Effective nuclear charge? What’s so effective about it? Is the regular nuclear charge just… lounging around, not doing much? It felt like a trick question, a linguistic prank designed to make me doubt my sanity. But then, as I kept reading, a tiny spark of understanding flickered. It wasn’t about a lazy nucleus; it was about how that nucleus’s pull is actually felt by its electrons, especially the ones that aren’t exactly living in the nucleus’s penthouse suite. And for Vanadium, with its more complicated electron arrangement, this idea starts to get really interesting.
The Nuclear Charge Hustle: What’s Really Going On?
Let’s zoom out for a sec. You know the nucleus, right? It’s that dense, positively charged core of an atom, packed with protons (which are positive) and neutrons (which are, well, neutral, bless their hearts). The number of protons is what defines an element – that’s your atomic number. For Vanadium (V), the atomic number is 23. So, in its neutral state, a Vanadium atom has 23 protons, giving its nucleus a grand total charge of +23.
Now, if only things were that simple! If every electron in the atom felt the full, unadulterated +23 pull from the nucleus, chemistry would be a lot less exciting. But we have electrons, and they’re not exactly living in a vacuum. They’re zipping around the nucleus in various energy levels and shapes – those orbitals we love to draw with squiggly lines. And here’s the kicker: there are other electrons in the atom, too. Lots of them.
These other electrons, especially the ones closer to the nucleus, get in the way. They’re like a bunch of gossipy neighbors standing in front of your window, partially blocking your view of the beautiful cityscape (which, in this analogy, is the attractive nucleus). They repel the outer electrons, essentially shielding them from the full force of the positive charge. This reduction in the nuclear attraction felt by an electron is what we call the effective nuclear charge, often abbreviated as Zeff. It’s the actual experience of the nucleus’s hug, not the theoretical maximum.
So, while the nucleus of Vanadium has a charge of +23, the electrons on the outside don’t feel that full +23. It’s like a celebrity walking through a crowd – they’re famous (+23), but they can’t directly shake hands with everyone because there are security guards and fans (the inner electrons) in the way.
Vanadium: The Electron Configuration Tango
To really get a handle on the effective nuclear charge for a 3d electron in Vanadium, we need to talk about its electron configuration. Remember how I mentioned that confusing textbook example? This is where it gets spicy. Vanadium is in the fourth period (n=4) and is the fifth element in the d-block. Its electron configuration is:
1s2 2s2 2p6 3s2 3p6 4s2 3d3
Let’s break this down, because it’s a bit of a dance. You’ve got your core electrons, neatly tucked away in the 1s, 2s, 2p, 3s, and 3p orbitals. Then you have your valence electrons, those outermost ones that are involved in bonding and generally making chemistry interesting. In Vanadium’s case, the 4s2 electrons are technically the furthest out, but the 3d electrons are also very important and often considered part of the valence shell, especially when it comes to transition metals.
The question is, what’s the effective nuclear charge felt by one of those 3d electrons? This is where the shielding effect really comes into play. The 3d electrons are being pulled by the +23 nucleus, but they’re also being repelled and shielded by all the electrons that are closer to the nucleus.

The Shielding Effect: Like a Fuzzy Blanket Around the Nucleus
Think of the inner electrons as creating a sort of fuzzy blanket of negative charge around the nucleus. This blanket doesn’t perfectly cancel out the positive charge, but it definitely softens the blow for the outer electrons. The more electrons you have packed in the inner shells, the more effective this shielding becomes.
For Vanadium, the electrons in the 1s, 2s, 2p, 3s, and 3p orbitals are all contributing to shielding the 3d electrons. These are a lot of electrons!
- 1s: 2 electrons
- 2s, 2p: 2 + 6 = 8 electrons
- 3s, 3p: 2 + 6 = 8 electrons
So, we have a total of 2 + 8 + 8 = 18 electrons in orbitals that are definitely closer to the nucleus than the 3d orbitals. These are the primary shielders.
But wait, there’s more! The 3d electrons themselves also shield each other. This is where things get a bit nuanced. Electrons within the same subshell don’t shield each other as effectively as electrons in inner shells. It’s like people standing in a circle – they can bump into each other, but they’re not really blocking the view of what’s happening in the center as much as someone standing directly in front of you.
Calculating (and Understanding) Zeff
So, how do we get a number for this effective nuclear charge? There are various methods, but a common one is using Slater’s rules. They’re not perfect, but they give us a good approximation. Slater’s rules assign a shielding constant (S) to each electron, based on the orbital it’s in and the orbitals of the other electrons.
For an electron in a 3d orbital, the rules (simplified for our purposes) are:
- Electrons in the same (3d) subshell contribute 0.35 each to the shielding constant.
- Electrons in inner shells (n-1) contribute 0.85 each.
- Electrons in shells further out (n-2 and beyond) contribute 1.00 each (but these don’t apply here as we’re looking at the pull from the nucleus).
Let’s apply this to our 3d electron in Vanadium. We have 3 electrons in the 3d subshell. So, the shielding from within the 3d subshell is 3 * 0.35 = 1.05. Uh oh, wait. If we have 3 electrons in 3d, and we pick one to calculate Z_eff for, there are 2 other 3d electrons. So, the shielding from other 3d electrons is 2 * 0.35 = 0.70.

Now, let's consider the inner electrons. These are all the electrons in shells n=1, n=2, and n=3 (except the 3d ones we just accounted for). We have:
- 1s2: 2 electrons. These are in the n-1 shell relative to 3d (if we consider n=3 to be the principal shell for 3d, and n=2 for 2p, etc. This can get confusing with d-orbitals). Let's use a more standard Slater rule interpretation for clarity.
A clearer way to think about Slater's rules for a 3d electron in Vanadium:
Zeff = Z - S
Where Z is the nuclear charge (23 for Vanadium) and S is the shielding constant.
For a 3d electron, Slater's rules say:
- Electrons in the 3d orbital: 3 electrons total. For one 3d electron, the other 2 electrons in the 3d orbital shield it with a value of 0.35 each. So, 2 * 0.35 = 0.70.
- Electrons in the (n-1) shell: This includes the 4s electrons! Yes, that’s right. Electrons in the immediately preceding principal shell contribute 0.85. So, the 4s2 electrons shield the 3d electrons. That’s 2 * 0.85 = 1.70.
- Electrons in the (n-2) shell and lower: These are the 3s, 3p, 2s, 2p, and 1s electrons. These contribute 1.00 each.
- 3s2, 3p6: 8 electrons * 1.00 = 8.00
- 2s2, 2p6: 8 electrons * 1.00 = 8.00
- 1s2: 2 electrons * 1.00 = 2.00
- Total shielding from electrons in shells n-2 and lower = 8.00 + 8.00 + 2.00 = 18.00
So, the total shielding constant (S) for a 3d electron in Vanadium would be approximately:
S = (0.70 from other 3d electrons) + (1.70 from 4s electrons) + (18.00 from inner shells)

S = 0.70 + 1.70 + 18.00 = 20.40
Therefore, the effective nuclear charge (Zeff) for a 3d electron in Vanadium is:
Zeff = Z - S = 23 - 20.40 = 2.60
Whoa. So, even though the nucleus has a charge of +23, a 3d electron in Vanadium only feels a pull of about +2.60. That’s a significant difference! It’s like the nucleus is screaming its love (+23) but only about 10% of that message is actually getting through to the 3d electron.
Why Does This Matter? The Big Picture (and More Chemistry Jargon)
Okay, so we’ve done the math, and it might feel a bit abstract. Why should we care about this seemingly small number, 2.60? Well, Zeff is a fundamental concept that helps us understand a bunch of important chemical properties. It's the real boss behind the scenes, dictating how atoms behave.
Atomic Radius: A higher Zeff means the nucleus has a stronger pull on the outermost electrons. This pulls the electron cloud closer to the nucleus, resulting in a smaller atomic radius. If you compare Vanadium to elements earlier in the periodic table with fewer protons (and less shielding), Vanadium will be smaller. Conversely, elements later in the same period will have a higher Zeff and be even smaller.
Ionization Energy: This is the energy required to remove an electron from an atom. If an electron feels a strong pull from the nucleus (high Zeff), it's going to be harder to yank it away. So, higher Zeff generally correlates with higher ionization energies. Vanadium's 3d electrons, while not as tightly bound as, say, a 2p electron, still experience a significant pull.

Electronegativity: This is an atom's ability to attract electrons in a chemical bond. Again, a stronger pull from the nucleus (higher Zeff) means the atom is better at attracting electrons. This is why transition metals, including Vanadium, tend to have moderate electronegativities.
The fact that the 3d electrons in Vanadium have an effective nuclear charge of around 2.60 is crucial for understanding its chemical behavior. It explains why Vanadium can form multiple oxidation states. These 3d electrons are close enough to the nucleus to be involved in bonding but also shielded enough that they can be more easily removed or shared than if they were feeling the full +23 charge.
The 4s vs. 3d Tango: A Nuance to Ponder
You might be thinking, "Wait a minute! The 4s electrons are further out than the 3d electrons, so shouldn't they have a lower Zeff?" That’s a great question, and it highlights the complexity of transition metals. According to Slater's rules, a 4s electron in Vanadium would have a different Zeff. The 3d electrons would shield the 4s electrons, and the 3d electrons are in the n-1 shell relative to 4s.
Let's quickly estimate for a 4s electron:
- Other 4s electron: 1 * 0.35 = 0.35
- 3d electrons: 3 * 0.85 = 2.55
- 3s, 3p electrons: 8 * 0.85 = 6.80
- 2s, 2p electrons: 8 * 1.00 = 8.00
- 1s electrons: 2 * 1.00 = 2.00
Total shielding for 4s electron = 0.35 + 2.55 + 6.80 + 8.00 + 2.00 = 19.70
Zeff for 4s electron = 23 - 19.70 = 3.30
Interesting, right? The 4s electron feels a slightly higher effective nuclear charge than a 3d electron in Vanadium! This is a key reason why, despite being in the outermost shell, the 4s electrons are still quite tightly bound and involved in ionization. This interplay between the 3d and 4s electrons is what makes transition metals so chemically versatile. The energy levels are very close, and the shielding makes them behave in ways that aren't always intuitive based on simple distance from the nucleus.
So, the next time you see Vanadium in a chemical reaction, remember that its nucleus is playing a complex game of tug-of-war with its electrons. The effective nuclear charge isn't just a number; it's a measure of the real-world forces shaping how this element interacts with the world. It’s the reason Vanadium can contribute to alloys that make airplanes stronger, or act as a catalyst in chemical processes. It all comes down to how that nucleus’s charge is effectively experienced by its electrons. Pretty neat, huh? And much more engaging than just memorizing numbers!
