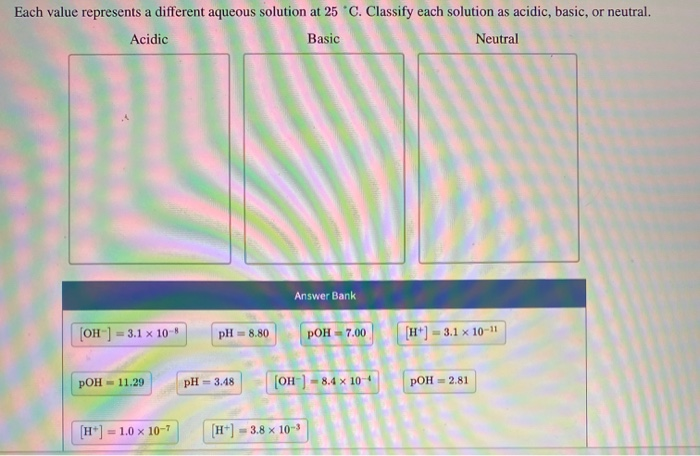
Each Value Represents A Different Aqueous Solution At

Ever find yourself staring at a bunch of labels, maybe on cleaning supplies or even just different kinds of bottled water, and wondering what all those technical-sounding words mean? You’ve probably seen things like “pH,” “alkaline,” or “acidic” tossed around. It can feel a bit like trying to decipher an ancient scroll, right? Well, guess what? It’s actually way simpler and, dare I say, more interesting than it sounds! Think of each of those terms as a little secret code that tells you something special about water. And why should you care? Because this secret code pops up in places you might not even realize, affecting everything from how your coffee tastes to how your laundry gets clean.
Let's break it down. At its core, we're talking about how "happy" or "grumpy" the water is, if you will. It all boils down to something called pH. Imagine a little dial, like the volume knob on your stereo, that goes from really low to really high. That dial is our pH scale, and it's usually measured from 0 to 14.
The sweet spot, the perfectly balanced, just-right-Goldilocks zone, is right in the middle at 7. This is considered neutral. Think of it like a calm, peaceful lake on a sunny day. Pure water, like the stuff that falls from the sky (before it picks up anything from the air, of course), hangs out here. It’s not trying to be anything it’s not; it’s just chilling.
Now, when that pH dial dips below 7, things start getting a little more… zesty. We call these solutions "acidic." Think of things that make your mouth pucker, like a squeeze of lemon juice into your water or the tang of vinegar. Acids have a sort of "bite" to them. In the chemical world, they're eager to give away tiny particles called hydrogen ions. It sounds complicated, but picture it like a kid who’s overflowing with energy and just wants to share it – sometimes a little too enthusiastically!
On the flip side, when the pH dial climbs above 7, we enter the realm of "alkaline" or "basic" solutions. These guys are the opposite of acids; they're more about accepting those eager hydrogen ions. Think of them as being a bit more… soothing or slippery. Baking soda, for instance, is a classic alkaline substance. It’s what your grandma might have used to clean her oven, and it’s a lot gentler than, say, drain cleaner (which is super alkaline!).

Why Does This pH Thing Even Matter in My Everyday Life?
Okay, so we've got neutral, acidic, and alkaline. Cool. But why should you, while you're trying to decide which dishwasher detergent to buy or what to drink, care about this? Because it’s everywhere!
Let’s start with the most obvious: drinking water. Bottled waters often market themselves as alkaline. Some people believe that drinking slightly alkaline water can help balance out the acidity in our bodies, though the scientific jury is still out on whether that makes a huge difference for most people. But hey, if drinking water that feels a little smoother and tastes a bit less sharp makes you happy, go for it! Conversely, highly acidic water can taste sour and might not be the most pleasant to sip all day.
Then there’s your morning cup of coffee. Did you know that coffee itself is slightly acidic? That’s part of why it tastes so robust! If you add milk or cream (which are slightly alkaline), you’re actually neutralizing some of that acidity. It's a little chemistry experiment happening in your mug every single morning, and you didn't even need a lab coat!

Kitchen Chemistry: From Cleaning to Cooking
Your kitchen is a pH playground! When you’re cleaning, different pH levels are good for different jobs. For tough grease, you might reach for something alkaline, like a degreaser or even that baking soda paste. The alkaline nature helps break down the fats. But for something like tackling rust stains, an acidic cleaner, like a vinegar-based solution, might be more your speed. It’s all about using the right tool for the job, and sometimes, that tool is a specific pH level.
Ever baked a cake? If you've ever used baking soda in a recipe, you've indirectly played with pH. Baking soda is alkaline. When it reacts with acidic ingredients in your batter (like buttermilk or brown sugar), it creates carbon dioxide gas. This is what makes your cake fluffy! Without that little pH dance, your cake would be a sad, flat pancake. Who knew a little bit of chemistry could lead to such deliciousness?
And what about your swimming pool? Keeping the water at the right pH is crucial! If it’s too acidic, it can irritate your eyes and skin, and it can damage the pool lining. If it’s too alkaline, the sanitizer (like chlorine) won't work as effectively, and the water might get cloudy. So, the pool folks are constantly monitoring that pH dial to keep everyone happy and healthy.
Your Body: A pH Balancing Act
Now, this is where it gets really personal. Our bodies are incredible, and they have a finely tuned system for maintaining a very specific pH balance, especially in our blood. Our blood normally hovers around a super-tight range of 7.35 to 7.45. That’s only a tiny difference on the pH scale, but it's vital for our cells to function correctly. It’s like a perfectly calibrated instrument; even a small deviation can cause big problems.
Your lungs and kidneys are the superheroes of this pH balancing act, working constantly to keep things just right. Think of them as the body’s internal pH police, making sure everything stays in the optimal zone. When you exhale, you’re actually releasing carbon dioxide, which is acidic, helping to adjust your body’s pH. Pretty neat, right?
Even things like digestion involve pH. Your stomach, for instance, is incredibly acidic (pH 1.5 to 3.5!) thanks to stomach acid. This extreme acidity is essential for breaking down food and killing harmful bacteria. It's like a tiny, highly efficient acid bath in there, perfectly designed for its job.

The Big Takeaway: It's All About Balance
So, next time you see those terms – acidic, alkaline, neutral, pH – don't just glaze over. Remember that it’s simply a way of describing how "friendly" or "reactive" a water-based solution is. It's a fundamental concept that pops up in your morning routine, your cleaning cabinet, your garden, and even within your own body.
Understanding these basic ideas can make you a more informed consumer and a more curious observer of the world around you. You can start to see the subtle chemistry at play in so many everyday things. It’s not about becoming a chemist overnight; it’s about appreciating the science that makes our lives work, sometimes with a little bit of zing, and sometimes with a lot of soothing calm.
It’s a reminder that even in the simplest things, like water, there's a whole world of fascinating properties just waiting to be understood. And who knows, maybe a little knowledge about pH will make you feel just a bit more empowered, a little bit more in control, and maybe, just maybe, a little bit more amazed by the ordinary.
