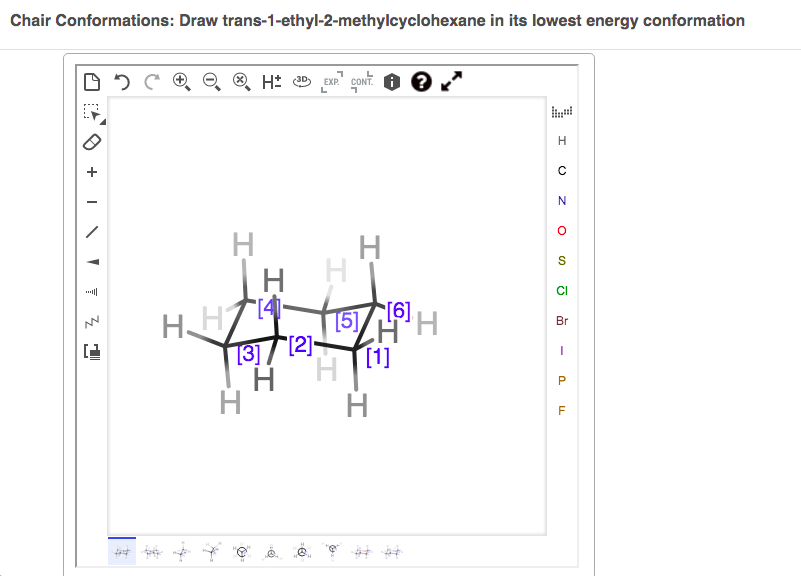
Draw Trans-1-ethyl-2-methylcyclohexane In Its Lowest Energy Conformation.
Ever looked at a molecule and wondered what makes it tick? For many, exploring the world of chemistry can seem a bit intimidating, but understanding how molecules are shaped, especially in their most comfortable, lowest energy form, is actually a surprisingly fun and rewarding puzzle. Think of it like finding the most relaxed way for your body to sit on the couch – molecules have their preferred postures too! This exploration is a big deal in chemistry because it helps us predict how molecules will behave, and that's incredibly useful for everything from designing new medicines to creating better materials.
So, what’s the big deal about trans-1-ethyl-2-methylcyclohexane and its lowest energy conformation? For beginners, it’s a fantastic way to start visualizing 3D structures and understanding the concept of spatial arrangement. It's like building with molecular LEGOs, where you learn how different pieces (atoms) fit together. For families, this can be a great way to spark curiosity about science, perhaps using models or drawings to explain to kids how atoms connect. Hobbyists in fields like molecular modeling or even organic gardening might find this knowledge useful as it can influence how plants interact with different chemical compounds.
Let’s break down what trans-1-ethyl-2-methylcyclohexane means. We have a cyclohexane ring, which is like a six-membered ring of carbon atoms. Then, we have an ethyl group (two carbons) and a methyl group (one carbon) attached. The '1' and '2' tell us where they are on the ring, and 'trans' is the crucial part – it means these two groups are on opposite sides of the ring. Now, for the "lowest energy conformation," think about that comfy couch posture again. A cyclohexane ring can exist in different shapes, but the most stable one is the "chair" conformation. In this chair, the ethyl and methyl groups want to be in the least crowded positions to minimize repulsion. For a trans arrangement, this usually means both substituents are in equatorial positions, pointing outwards from the ring, rather than axial, pointing up or down along the ring's axis.
Getting started is simpler than you might think! You don’t need fancy equipment. You can start by drawing simple cyclohexane rings. Then, practice adding substituents. For visualization, consider using molecular model kits. They are readily available and make it easy to see how different groups interact in 3D space. Even using toothpicks and marshmallows can be a fun, DIY approach to building these structures. Look up diagrams and examples of cyclohexane chair conformations online; there are tons of helpful resources that show how to draw them clearly.
Understanding the lowest energy conformation of molecules like trans-1-ethyl-2-methylcyclohexane isn't just an academic exercise. It’s about appreciating the elegance and efficiency of nature at a molecular level. It’s a small window into the vast and fascinating world of chemistry that’s accessible and genuinely enjoyable to explore. So, next time you encounter a molecule, remember it's not just a jumble of letters and numbers, but a structure with its own preferred way to relax!
