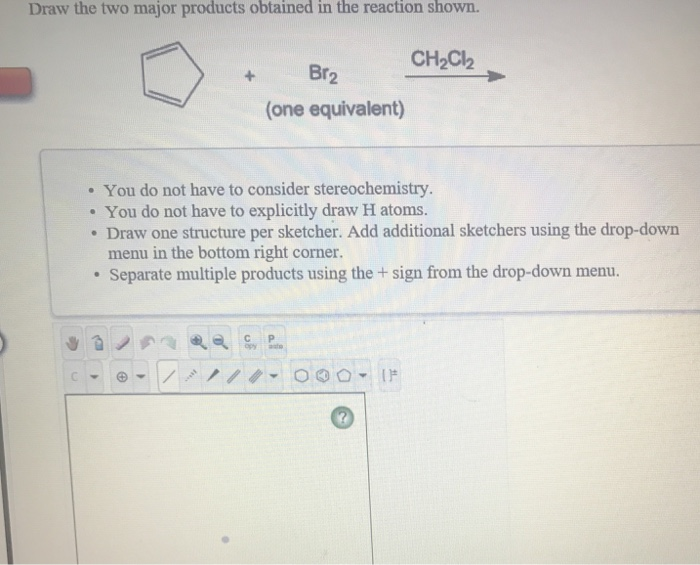
Draw The Two Major Products Obtained In The Reaction Shown
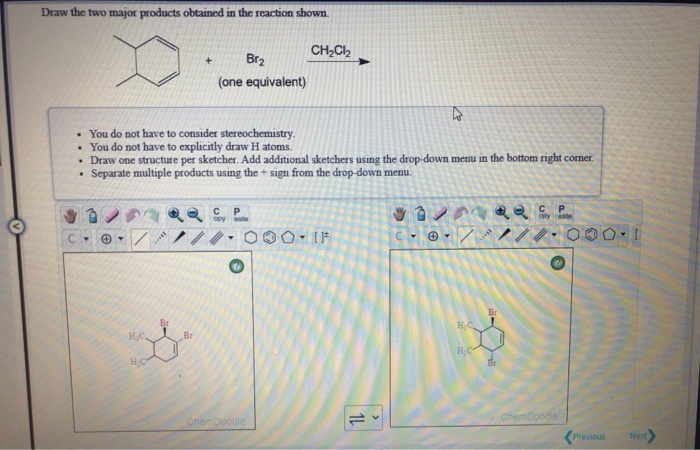
Get ready for a little bit of scientific magic! We're about to dive into a reaction that's pretty neat. It's like a tiny chemical dance, and the results are quite interesting.
Think of it like baking a cake, but with molecules! You start with some ingredients, mix them up, and poof! You get something totally new. In this case, the starting materials are quite specific, and the way they interact is what makes it all so cool.
The star of our show is a molecule that’s a bit of a celebrity in the chemical world. It’s called Cyclohexene. Ever heard of it? It’s a ring of six carbon atoms with a double bond in the middle. This double bond is like an open invitation for other molecules to join the party!
And who’s invited to this party? Well, one of the key guests is Bromine. Bromine is a bit of a dramatic element, often seen as a reddish-brown liquid. When it meets Cyclohexene, things start to happen!
So, what’s the big deal? It’s all about what happens when these two meet. The Bromine molecule, which is made of two bromine atoms stuck together, sees that double bond in Cyclohexene. It’s like a handshake, but much more involved.
The double bond in Cyclohexene is a place where electrons are easily shared. It’s a bit like a friendly, open space. Bromine, seeing this, decides to break its own bond and reach out to both sides of the double bond.
This is where the first major product comes into play. Imagine the Bromine molecule splitting into two. Each bromine atom then attaches itself to one of the carbon atoms that were part of the double bond in Cyclohexene.
The result? A new molecule is formed. This molecule still has the six-carbon ring, but now, instead of a double bond, it has two bromine atoms attached. These bromine atoms are on neighboring carbon atoms. This molecule has a rather fancy name: 1,2-Dibromocyclohexane.
It sounds complicated, but think of it this way: the Cyclohexene ring was a bit like a bicycle with two wheels side-by-side. The Bromine molecule was like two people wanting to hold onto those wheels. They grabbed on, one on each wheel, and now they're stuck there, holding on tight!
This 1,2-Dibromocyclohexane is one of the main things we get out of this reaction. It's a stable, solid molecule. You can actually see it and study it. It’s a direct result of the Bromine adding itself across that double bond.
But wait, there’s more! Chemical reactions are rarely that simple. Sometimes, under certain conditions, a slightly different story unfolds. What if the reaction isn't just about Bromine and Cyclohexene alone?
Let’s add another player to the mix. Imagine we’re doing this reaction in water. Water might seem like just a bystander, but in chemistry, water can be quite active. It can act as a solvent, but it can also participate in the reaction itself.
So, when Bromine and Cyclohexene get together in the presence of water, the Bromine still wants to add across the double bond. However, there’s now a new competitor for that open spot: a water molecule.
The water molecule can also approach the double bond. It’s not as forceful as Bromine, but it’s there. So, after the Bromine has started its move, the water molecule can jump in and attach itself to one of the carbon atoms.
This leads to our second major product. This molecule is also based on the Cyclohexene ring. It still has one bromine atom attached, just like before. But instead of a second bromine atom, it now has a part of the water molecule attached.

This part from water is a hydroxyl group, which is basically an oxygen atom bonded to a hydrogen atom (-OH). So, this new molecule has a bromine atom on one carbon and a hydroxyl group on the neighboring carbon.
This second product is called 2-Bromocyclohexanol. Again, a fancy name, but it tells us exactly what’s there. It has a bromine and it has an alcohol group (the -OH part) attached to the cyclohexane ring.
This reaction, where water also gets involved, is super interesting. It shows us how even seemingly simple reactions can have multiple outcomes depending on the exact environment. It's like a branching path in a choose-your-own-adventure book.
The beauty of this is seeing how different molecules behave. Bromine is a halogen, and it loves to do what’s called an "electrophilic addition." It’s attracted to electron-rich areas like double bonds. Water, on the other hand, can act as a nucleophile, meaning it’s attracted to electron-deficient areas.
When both Bromine and water are present, there’s a competition. The Bromine usually gets there first, creating a temporary intermediate. Then, the water molecule can attack that intermediate, leading to the formation of 2-Bromocyclohexanol.
So, to sum it up, in the reaction of Cyclohexene with Bromine, we get two main products:
Product 1: 1,2-Dibromocyclohexane
This is what happens when Bromine just adds to itself across the double bond. Two bromine atoms end up on adjacent carbons of the ring.
Solved Draw the two major products obtained in the reaction | Chegg.com
Product 2: 2-Bromocyclohexanol
This is what happens when Bromine and water both get involved. One bromine atom attaches, and a hydroxyl group from water attaches to the neighboring carbon.
Why is this so entertaining? Because it’s a great example of how chemistry isn't always a single, straightforward answer. It shows us the power of different reagents and conditions to steer a reaction in different directions.
Imagine you're a chef. You have your ingredients, and you want to make a dish. Depending on what else you add to the pot, you can end up with two entirely different, yet delicious, meals. That's what's happening here, but with molecules!
The visual of the Bromine molecule breaking apart and the two bromine atoms happily attaching themselves to the Cyclohexene ring is quite satisfying. It’s like a puzzle piece fitting perfectly into place.
And then, the twist with water! It’s like a surprise guest at the party who also brings a gift. The fact that water can incorporate itself into the final molecule is a testament to its versatility.
What makes it special is that it's a fundamental reaction that chemists learn about. It’s a building block for understanding more complex chemical transformations. It’s like learning your ABCs before you can write a novel.
Seeing these structures, 1,2-Dibromocyclohexane and 2-Bromocyclohexanol, come to life from simple starting materials is pretty remarkable. It's a peek into the molecular world, where atoms and bonds are constantly rearranging.
It’s a visual treat for the mind. You can picture the ring, the double bond, the Bromine molecule approaching, and then the final structures. It’s like watching a tiny, intricate ballet unfold.
The names might sound intimidating, but the concepts are quite accessible. It’s all about addition reactions, where new things are added to an existing molecule.
This reaction is a fantastic way to illustrate the concept of regioselectivity and stereoselectivity in organic chemistry, though we won't get into those super technical terms here! Just know that the way things attach matters, and it's dictated by the molecules themselves.
Think about the journey of the atoms. They start in one arrangement, and through a chemical interaction, they find a new, stable arrangement. It's a process of transformation and creation.
This is the kind of science that sparks curiosity. It makes you wonder, "What else can molecules do?" The possibilities are endless, and reactions like this are the keys to unlocking them.
So, the next time you hear about a chemical reaction, remember this one. Remember the dance of Cyclohexene and Bromine, and how water can sometimes join in the fun. It’s a little bit of chemistry that’s both understandable and wonderfully intriguing!