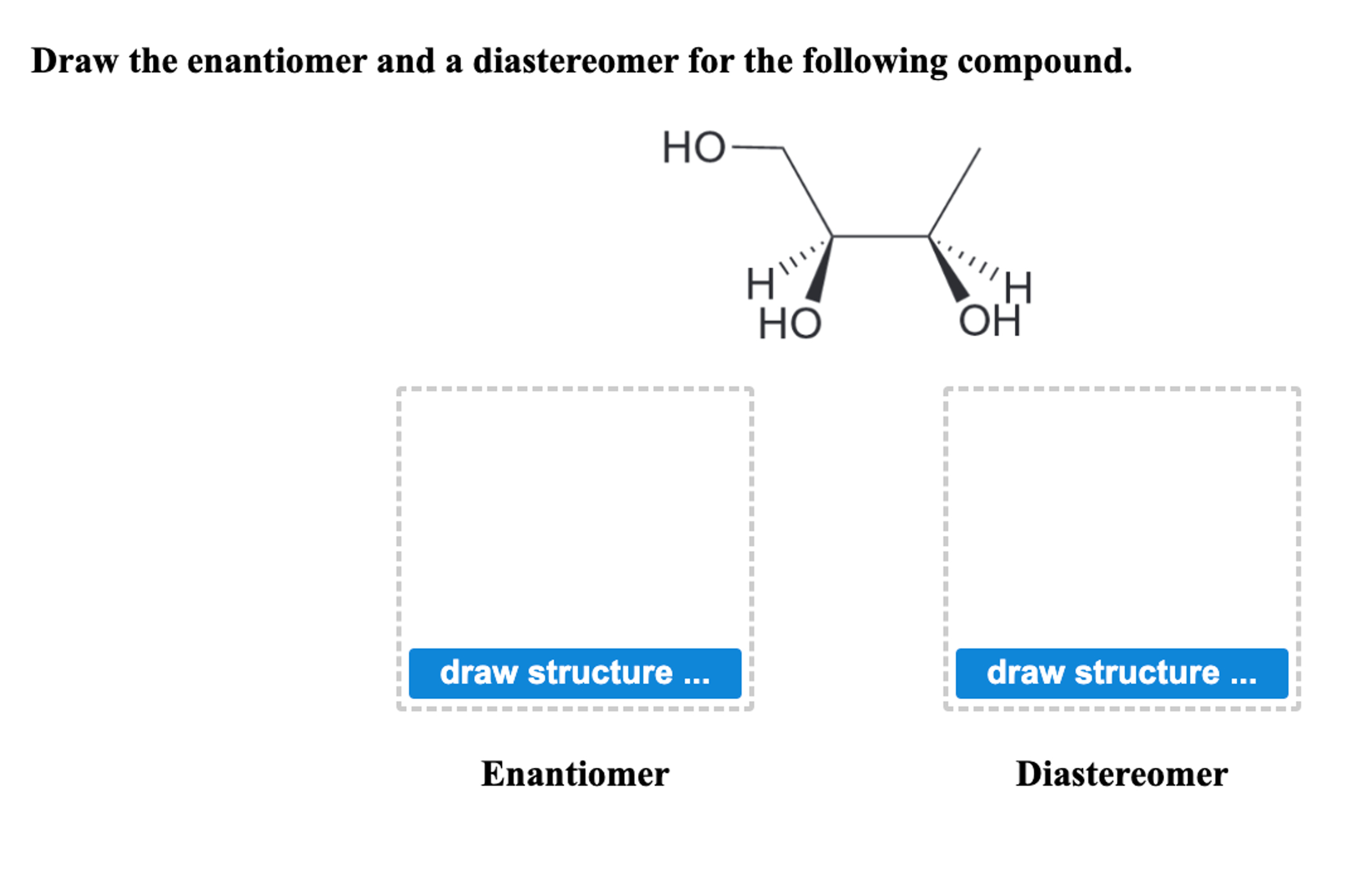
Draw The Structure Of The Enantiomer Of Glucose
Hey there, science adventurers and lovers of all things sweet! Ever stopped to think about sugar? Like, really think about it? We're talking about that wonderful stuff that makes cookies magical and coffee just right. Well, today, we're diving into a tiny, fascinating corner of the sugar universe, and it’s going to be a blast! Get ready to meet the super-cool, slightly-misunderstood twin of our beloved glucose.
You know how sometimes you have a best friend, and they have a twin? They look almost exactly alike, right? Maybe they even have the same amazing taste in music or a killer sense of humor. But then, there are these tiny, almost imperceptible differences that make them, well, them. They might do a little handshake differently, or one might prefer their pizza with pineapple (don't worry, we won't judge!). That's kind of what we're talking about in the world of molecules, and our star player today is glucose. It's a sugar, a super important one for… well, pretty much everything that needs energy. Think of it as the energizer bunny of your body!
Now, imagine you've got a perfectly drawn picture of glucose. It's a beautiful, intricate structure, like a tiny molecular sculpture. It has all these arms and legs – well, chemists call them functional groups and carbon atoms – all arranged in a very specific way. It’s got a special little twist, a handedness if you will, that’s super important for how it interacts with everything else. It's like the way you'd draw a right hand. You know exactly where all the fingers and thumbs go, and it fits perfectly into a right-handed glove.
Glucose is like the star athlete of the sugar world, always ready to power you up!
So, what happens when we want to draw its twin? Its enantiomer? This is where things get really fun! Think of it like looking at that right hand in a mirror. What do you see? You see a left hand! It's the same basic shape, the same number of fingers, the same thumb, but everything is flipped. It's like a perfectly symmetrical reflection. It's still a hand, but it's definitely the other hand.
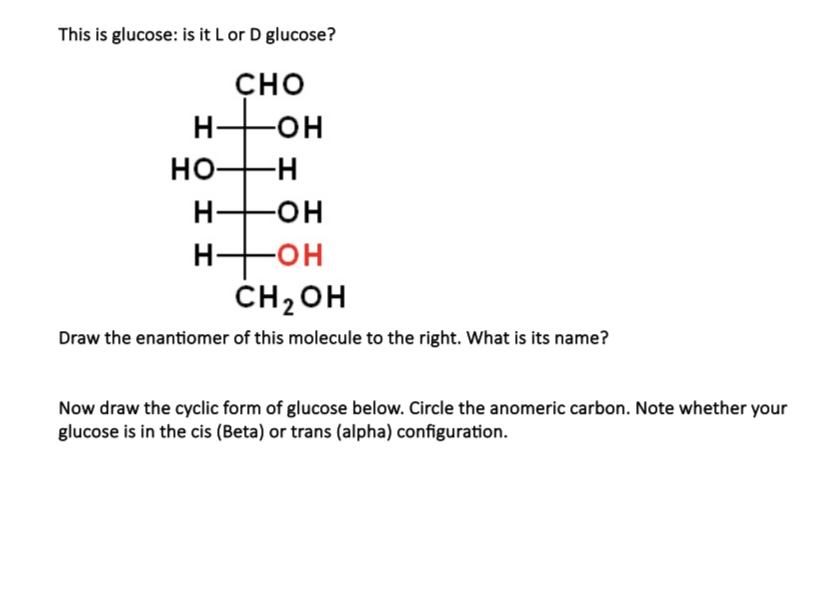
And that's precisely what an enantiomer is in chemistry! It's a molecule that is the mirror image of another molecule, but they aren't identical. They are like those fraternal twins who look alike but have a few distinct differences. They're related, they're family, but they are their own unique selves. When it comes to glucose, its mirror-image twin is called D-glucose. Now, the 'D' part is just a chemist's way of saying it's oriented in a certain direction, like saying "right-handed." Its enantiomer, the mirror image, is called L-glucose. So, L-glucose is the enantiomer of D-glucose!
Let's get our imaginary art supplies out! We’re going to draw the structure of this fabulous enantiomer. Don't worry, this isn't a scary chemistry exam. This is like doodling with molecular shapes. You've probably seen diagrams of sugars before, looking like little houses or abstract art pieces. They have a main chain of carbon atoms, and then little arms sticking out. For glucose (and therefore for its enantiomer too!), we're usually talking about a six-carbon sugar. It’s shaped like a ring, often drawn as a hexagon, with some extra bits and bobs sticking out.
Now, if you had a picture of D-glucose, and you wanted to draw its enantiomer, L-glucose, you'd basically take that picture and hold it up to a mirror. Or, even better, imagine a tiny molecular pretzel and you flip it inside out. All the little arms that were sticking out on one side will now be sticking out on the other side. It’s that simple! It’s a bit like having a Lego castle and then flipping the whole thing over. The bricks are still there, but their positions relative to the base have changed in a mirrored way. It’s a completely different orientation, even though it’s made of the exact same building blocks.
Think of it this way: you might have a favorite pair of sneakers, the kind that are super comfy and make you feel like you can run a marathon. Now, imagine you have a perfect mirror image of those sneakers. They're still sneakers, they look the same size, they have the same laces, but one is clearly designed for your left foot and the other for your right foot. You can't wear the right sneaker on your left foot and expect it to feel right, can you? It just won't fit properly. That's a great analogy for how enantiomers work. Even though they're mirror images, they interact differently with other molecules in our bodies, which are also made of chiral (that's the fancy word for "handed") molecules.
So, when we’re talking about drawing the structure of the enantiomer of glucose, we’re essentially redrawing the glucose molecule but flipping all the asymmetrical parts to their opposite sides. It's like a fun molecular makeover, giving it a whole new perspective! It’s a beautiful dance of atoms, a little twist here, a little flip there, and voila! You have its perfectly mirrored twin, ready to embark on its own molecular adventures. Isn't that just the coolest? Science is full of these amazing, subtle differences that make all the difference in the world!
