Draw The Structure Of A Molecule With 7 Bonding Domains
Let's talk about molecules. Specifically, the ones that are a bit… extra. You know, the ones that seem to have too many friends at the party. Today, we’re diving into the wild world of drawing molecules with a whopping 7 bonding domains.
Now, I know what you’re thinking. “Bonding domains? Is this a chemistry class?” Relax, folks! We’re not here to ace any exams. We’re here to have a little fun with the universe’s building blocks. Think of it like assembling a very, very complicated LEGO set.
When we talk about bonding domains, we're basically talking about the spots where atoms decide to hold hands. It's where the chemical magic happens, you see. More domains mean more hand-holding. Simple, right?
But 7 bonding domains? That's like a molecule that decided to host a speed-dating event and everyone brought a plus-one. It’s a lot of connections. It’s a lot of… business.
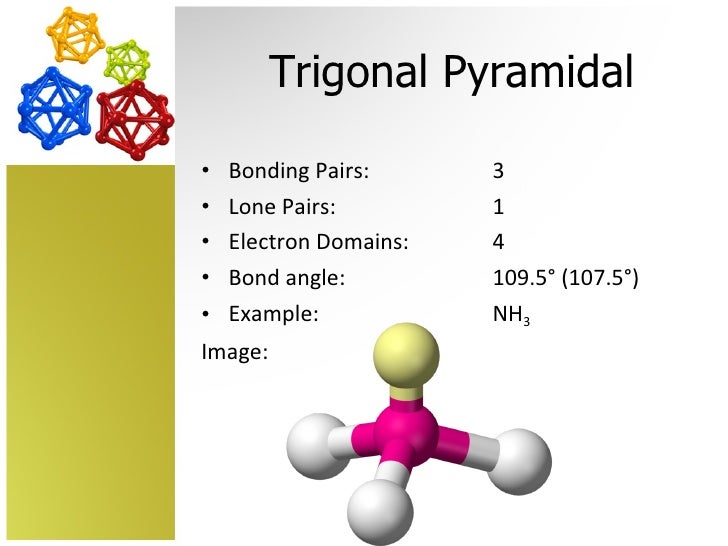
So, how do you even begin to draw such a thing? Imagine a central atom. This is our party host. It's the star of the show, holding court. Everything revolves around this little guy (or gal).
Now, this central atom needs to make friends. And not just one or two. It needs to make seven! That’s a social butterfly of an atom. It’s practically a celebrity in the molecular world.
When we draw these molecules, we often use something called VSEPR theory. Sounds fancy, doesn't it? But it's just a clever way to figure out how these atoms arrange themselves. They like their personal space, you see.
Even with all those hands being held, the atoms still want to be as far apart as possible. It’s like a group of friends trying to sit on a crowded bus. They’ll spread out as much as they can, right?
For our 7 bonding domains, this leads to some pretty interesting shapes. It's not your average, everyday triangle or square. Oh no. This is molecular architecture at its finest, or perhaps, its most chaotic.
One of the most common arrangements for 7 domains is called pentagonal bipyramidal. Try saying that five times fast! It’s a mouthful, and it looks like a whole lot of geometry happening.
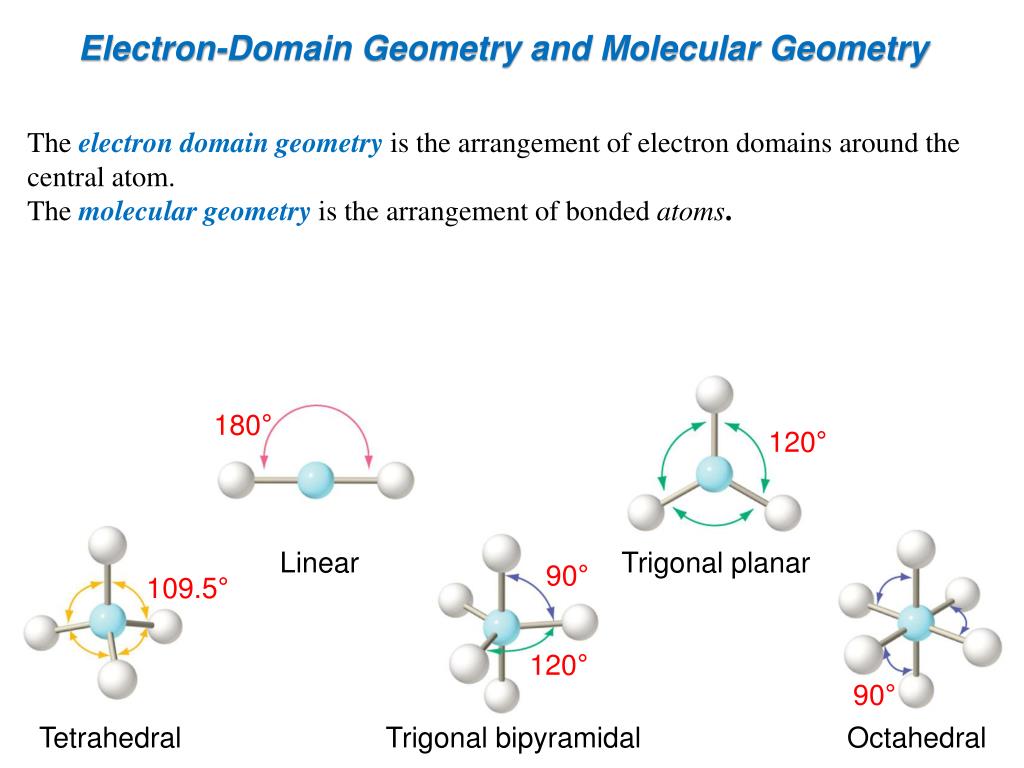
Imagine a pentagon. You know, five sides? Now, stick a pointy hat on top and another pointy hat on the bottom. That’s kind of the vibe we’re going for.
The central atom is right in the middle of that pentagon. Then you have five other atoms sitting around the equator of our imaginary sphere. And then, two more atoms are chilling at the north and south poles.
It’s like a planetary system, but instead of planets, we have atoms. And instead of gravity, we have the irresistible urge to bond.
Drawing this can feel like a bit of a puzzle. You’ve got your central atom, let’s call it ‘X’. Then you have your seven friends, let’s call them ‘Y’. So, ‘XY7’. Simple enough to write, right?
But the drawing? That’s where the adventure begins. You draw your ‘X’ in the middle. Then you start placing your ‘Y’s. Five of them forming a pentagon around ‘X’.
These five ‘Y’s are all kind of in the same plane. They’re the “equator crew.” They’re doing their thing, keeping things balanced.
Then, you have your two special ‘Y’s. These are the “axial twins.” One goes up, one goes down. They are perfectly positioned above and below the plane of the pentagon.
It looks a bit like a kite, or maybe a weird, futuristic spacecraft. It's definitely not something you'd doodle on a napkin during a boring meeting. Unless your meetings are really boring.

Now, here’s where things get even more interesting. Sometimes, not all of those 7 bonding domains are actually forming new bonds. Sometimes, one of them might be a lone pair of electrons.
A lone pair is like the introverted cousin at the family reunion. It’s there, it’s part of the group, but it’s not actively mingling with everyone else. It’s just… existing.
When you have a lone pair in one of those 7 spots, the shape of the molecule changes. It’s like taking away one of the guests from our party, and suddenly everyone else shifts around a bit.
If you have one lone pair, the shape might be called pentagonal bipyramidal distorted. Which is a fancy way of saying, “it’s not quite the perfect shape anymore.”
The lone pair, being a bit shy, tends to push the other atoms around. It’s like that one person who takes up more space than they need. The other atoms have to adjust.
So, instead of a perfect pentagonal bipyramid, you might get something that looks a little lopsided. It’s still got the basic structure, but it's got character. It's got… quirks.
What if you have two lone pairs? Well, that’s even more interesting. The molecule starts to look even more peculiar. It’s like the party guests have now formed little cliques.
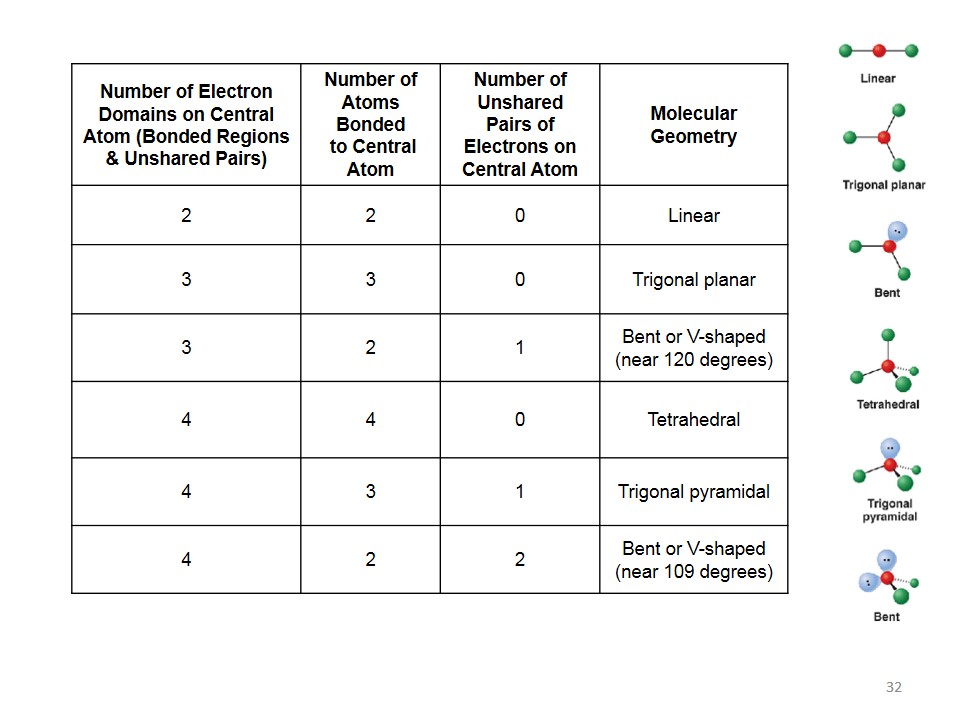
The specific shape depends on where those lone pairs decide to hang out. They try to get as far away from each other and the bonded atoms as possible, of course.
This is where things can get a little mind-bending. The rules of geometry and electron repulsion are in full effect. It's a delicate dance of attraction and repulsion.
Honestly, sometimes I think chemists just invent these shapes to make us draw more. It's like a secret code, and we're the ones trying to crack it with our pencils.
But there's a certain beauty to it, isn't there? The fact that these tiny, invisible particles can arrange themselves in such complex and predictable ways.
Think about it. A molecule with 7 bonding domains is not just a random jumble of atoms. It's a carefully orchestrated arrangement, dictated by the fundamental forces of nature.
It’s a testament to the fact that even in the seemingly chaotic world of chemistry, there’s order. There’s structure. There’s a certain elegance.
So, next time you’re looking at a chemical structure, don’t just see lines and circles. Imagine the little atoms, holding hands, trying to find their personal space. Imagine the molecules throwing their own unique parties.
And when you encounter a molecule with 7 bonding domains, give it a little nod. It’s one of the overachievers. It’s one of the ones who decided to go all out.

Drawing it might feel like a challenge. But it’s also a chance to appreciate the incredible diversity and complexity of the molecular universe. It’s a little glimpse into how everything around us is put together.
So, grab your pen, sketch out that central atom, and start placing those seven friends. Don’t be afraid to get a little creative. After all, it's your drawing. Make it your own. Make it fun.
And if it looks a little funny, well, that’s just part of its charm. Just like us, molecules don’t always have to be perfectly symmetrical to be amazing.
The pentagonal bipyramidal shape, with or without those shy lone pairs, is a marvel. It’s a structure that allows for a whole lot of chemical interactions.
It’s a lot of bonding, a lot of potential reactions, and a lot of ways for nature to get creative. So, embrace the complexity. Enjoy the process. And smile, because you’re drawing a molecule with 7 bonding domains. That's pretty cool, no matter how you slice it.
It’s an “unpopular opinion,” perhaps, but I think these complex shapes are more interesting than the simple ones. They’ve got stories to tell. They’ve got personalities.
So, go forth and draw! Let your inner molecular architect shine. And remember, even with 7 domains, there’s always room for a little bit of fun.
