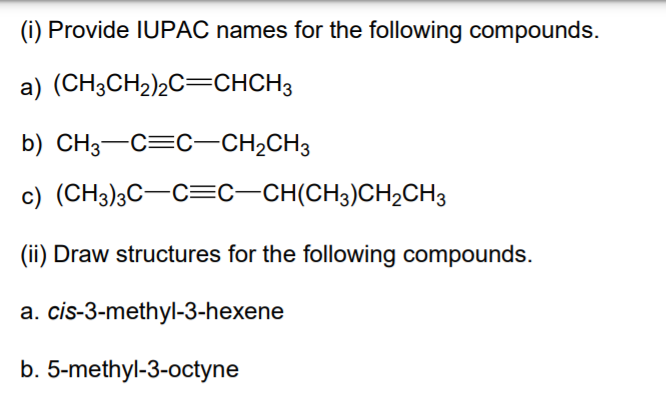
Draw The Structural Formula Of 5 Ethyl 3 Octyne

Ever looked at a jumble of letters and numbers and wondered what it all meant? If you've ever dabbled in chemistry, or even just seen those fancy molecular diagrams in movies, you've probably encountered structural formulas. But these aren't just scribbles for scientists! Understanding them is like having a secret decoder ring for the building blocks of everything around us. And today, we're going to unlock the mystery of drawing one specific, rather interesting molecule: 5-ethyl-3-octyne. Trust us, it's more fun than it sounds, and it’s a fantastic way to get a handle on how atoms link up to create the diverse world of organic compounds!
Why Draw Structural Formulas? It’s All About Seeing the Structure!
So, why bother drawing out these sometimes intricate maps of molecules? Think of it this way: a chemical name like "5-ethyl-3-octyne" tells us what's in it and roughly where things are, but it doesn't show us the complete picture. A structural formula, on the other hand, is like a blueprint. It reveals the exact arrangement of atoms and the bonds connecting them. This is incredibly important because even tiny differences in how atoms are arranged can lead to vastly different properties. For instance, a drug molecule's effectiveness, a plastic's flexibility, or even a flavor's intensity can hinge on its precise structural formula.
The benefits of being able to visualize these structures are huge. For students, it’s fundamental to understanding chemical reactions and predicting outcomes. For chemists, it’s the basis for designing new materials, discovering medicines, and even developing sustainable energy sources. For the curious mind, it’s simply a fascinating glimpse into the intricate dance of atoms that forms our universe. It’s a way to move beyond abstract concepts and see the tangible, three-dimensional shapes that make up everything from the air we breathe to the food we eat. And drawing them yourself is a powerful way to solidify that understanding.
Understanding structural formulas is like learning the alphabet of chemistry. Once you know the letters and how they combine, you can start reading and writing the language of molecules!
Let's Decode "5-Ethyl-3-Octyne"
Now, let's break down our target molecule, 5-ethyl-3-octyne, and get ready to draw it. The name itself is a treasure trove of information. We’ll dissect it piece by piece:

- "Octyne": This is our anchor. The "oct" part tells us we have a chain of eight carbon atoms. The "yne" ending signifies the presence of a triple bond (a very strong connection between two carbon atoms where they share three pairs of electrons). In an "octyne," there's at least one triple bond.
- "3-octyne": This tells us where that triple bond is located. The "3-" indicates that the triple bond starts on the third carbon atom in the eight-carbon chain. We number the carbon chain from either end, and we choose the numbering that gives the triple bond the lowest possible number.
- "5-ethyl": This tells us we have a side chain attached to our main eight-carbon chain. The "ethyl" group is a small chain consisting of two carbon atoms (represented as -CH₂CH₃). The "5-" tells us where this ethyl group is attached: to the fifth carbon atom of our octyne chain.
The Drawing Process: Step-by-Step!
Ready to grab your virtual pencil? Here’s how we’ll construct the structural formula of 5-ethyl-3-octyne. We’ll use a simplified skeletal structure, which is common and much easier to draw!
Step 1: Draw the Main Carbon Chain
Start by drawing a zig-zag line representing the eight-carbon chain. Each peak and valley in the zig-zag represents a carbon atom. Remember, hydrogen atoms are implied to fill out the remaining bonds for each carbon, so we don't usually draw them in skeletal structures unless they are attached to a heteroatom (like oxygen or nitrogen).
Imagine drawing eight dots connected by lines, then arrange them in a zig-zag. Each point or end of a line is a carbon.
Step 2: Number Your Carbon Chain
Now, let's number our carbon atoms from left to right (or right to left, it doesn't matter for now). This is crucial for placing our triple bond and our ethyl group correctly. Let’s assume we number from left to right, 1 through 8.
Step 3: Place the Triple Bond
The name "3-octyne" tells us the triple bond is between the 3rd and 4th carbon atoms. In a skeletal structure, we represent a triple bond by drawing three parallel lines connecting the two carbon atoms. So, between your 3rd and 4th carbon, draw ===.
Your zig-zag line now has a triple bond on the third segment.
Step 4: Attach the Ethyl Group
The "5-ethyl" tells us an ethyl group is attached to the 5th carbon. An ethyl group is a two-carbon chain (-CH₂CH₃). In a skeletal structure, this is drawn as a short zig-zag line branching off the 5th carbon atom. Imagine the 5th carbon atom having a little two-segment arm extending from it.
So, on the 5th carbon of your main chain, draw a small zig-zag with two points.

Step 5: Fill in Implicit Hydrogens (Mentally!)
In skeletal structures, we don't draw the hydrogen atoms. However, it's good to remember that each carbon atom typically forms four bonds. The skeletal structure automatically accounts for this by implying the necessary hydrogens. For example, a carbon at the end of a chain (forming one bond to the next carbon) will have three implicit hydrogens. A carbon in the middle forming two bonds to other carbons will have two implicit hydrogens. A carbon involved in a triple bond and one single bond will have zero implicit hydrogens.
The Final Picture!
When you put it all together, you'll have an eight-carbon zig-zag chain. On the third and fourth carbons, you'll see the three lines of the triple bond. And branching off the fifth carbon, you'll see a two-carbon side chain (the ethyl group). This visual representation is the structural formula of 5-ethyl-3-octyne!
Drawing structural formulas might seem daunting at first, but with a little practice, it becomes second nature. It’s a fantastic skill that opens up a deeper understanding of the molecular world. So, the next time you hear a chemical name, remember you have the tools to visualize its structure. Happy drawing!
