Draw The Structural Formula Of 1 2 Dimethylcyclopentene
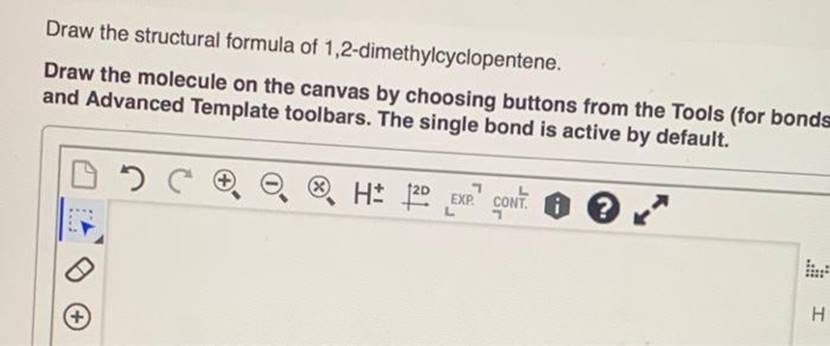
Hey there, science curious folks! Ever find yourself staring at a chemical name and wondering what on earth it looks like? Like, "1,2-dimethylcyclopentene"? Sounds like a mouthful, right? But what if I told you that behind that slightly intimidating name is a pretty neat little molecule, almost like a tiny, ring-shaped dancer with some friends attached?
Today, we're going to chill out and explore exactly that. We're going to draw the structural formula of 1,2-dimethylcyclopentene, and more importantly, we're going to understand what that even means. No need to break out the textbooks or cram for a pop quiz. Think of this as a friendly chat over a cup of tea, where we just happen to be talking about atoms and bonds.
So, what's the big deal about drawing a chemical structure? Well, it's like giving a molecule its own personal portrait. It tells us exactly where every atom is and how they're connected. It's the difference between knowing someone's name and actually seeing their face and knowing their quirky habits. For chemists, this is everything. It helps them predict how a molecule will behave, what it can do, and how it fits into the grand, molecular puzzle of the universe.
Let's break down our name: "1,2-dimethylcyclopentene." It's like a set of instructions, and each part gives us a clue. First off, we have "cyclopentene." What does that tell us? The "penta" part usually means five. So, we're dealing with a ring that has five corners, or in chemistry terms, five carbon atoms. Think of it like a pentagon, a familiar shape, right?
Now, the "ene" ending is super important. It means there's a double bond somewhere in that ring. Imagine two of the carbon atoms in our pentagon decided to hold hands extra tightly, sharing not one, but two pairs of electrons. This double bond is like the special feature, the little sparkle, that makes alkenes (that's what "ene" signifies) behave in certain ways. It’s a point of interest, a place where reactions love to happen!
So, we've got a five-membered ring, a pentagon shape, with one double bond. Pretty straightforward so far, huh? But what about the "1,2-dimethyl"? The "di" means two. And "methyl" refers to a –CH₃ group, which is basically a carbon atom with three hydrogen atoms attached. It's like a tiny, friendly little arm sticking out from the main structure.

The "1,2-" part tells us where these methyl groups are attached to our cyclopentene ring. We number the carbon atoms in the ring, and the double bond gets priority, usually starting the numbering from one of the carbons involved in the double bond. So, "1,2-" means we have one methyl group attached to the carbon atom labeled '1' and another methyl group attached to the carbon atom labeled '2'. These are right next to each other!
Imagine our pentagon ring. Let's say the double bond is between carbon 1 and carbon 2. Then, we'd put a methyl group (that –CH₃ thing) on carbon 1 and another methyl group on carbon 2. They're like twins, sitting side-by-side on the double bond!
So, how do we actually draw this? In organic chemistry, we often use what are called skeletal structures. This is where we get fancy and simplify things. We don't draw every single carbon and hydrogen atom. Instead, each corner or endpoint of a line represents a carbon atom, and the hydrogen atoms are assumed to be there to fill up the bonding capacity of each carbon (usually four bonds). It’s like a secret code for chemists!
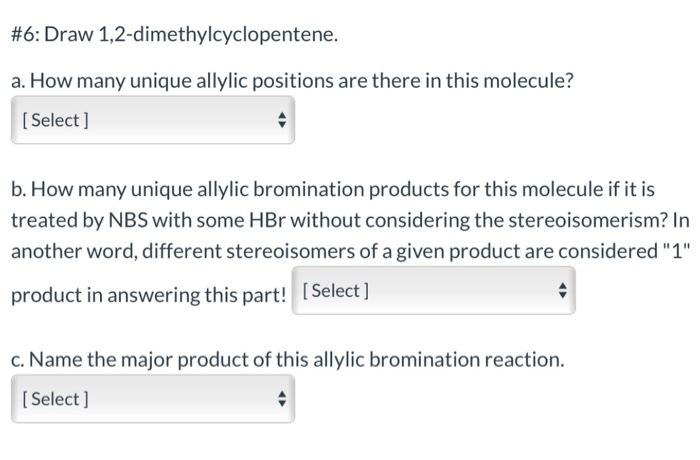
Let's start with our five-membered ring. We draw a pentagon. Easy peasy. Now, we need to show that double bond. We can pick any two adjacent sides of the pentagon and draw an extra line between them. That's our double bond, and the two carbons at the ends of those sides are now carbons 1 and 2.
Next, we add our methyl groups. Remember, a methyl group is a –CH₃. In a skeletal structure, we draw a short line sticking out from the carbon atom where the methyl group is attached. The end of that short line represents the carbon atom of the methyl group, and we understand there are three hydrogens attached to it. So, we draw a short line sticking out from carbon 1 and another short line sticking out from carbon 2.
And voilà! You've just visualized 1,2-dimethylcyclopentene. It's a five-membered ring with a double bond, and two little –CH₃ arms sticking out from adjacent carbons involved in that double bond. Pretty neat, right?
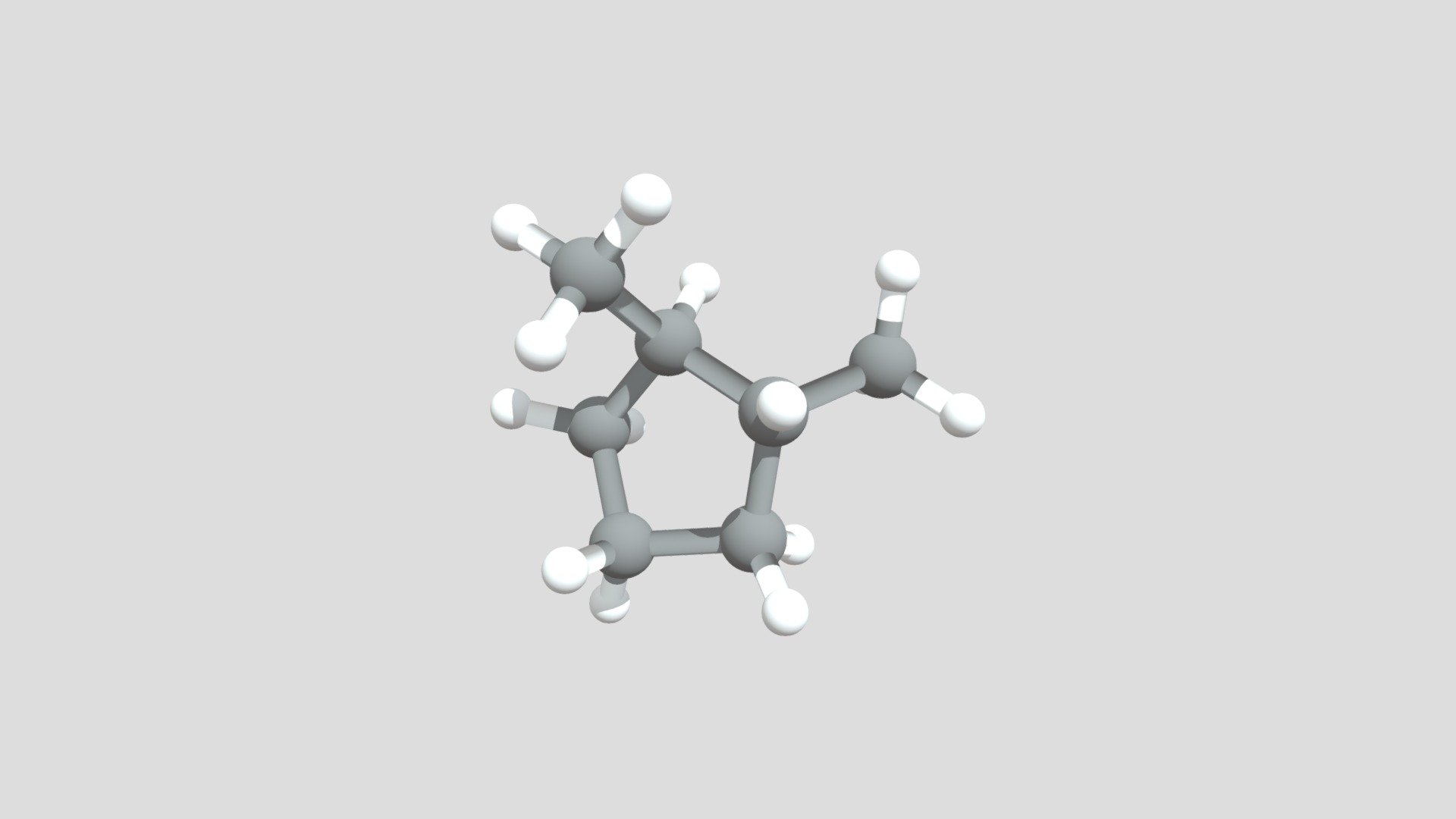
Think of it like this: the cyclopentene ring is the main stage, the performers. The double bond is the spotlight, the most exciting part of the show. And the two methyl groups are like the lead dancers, positioned right where the spotlight hits. They're important players, adding their own flavor to the performance.
Why is this interesting? Well, molecules like this are the building blocks of so many things. They're found in fuels, in perfumes, in medicines, and even in the natural compounds that make up living organisms. Understanding their structure is key to understanding their function and how we can use them.
The arrangement of these methyl groups can even matter! In some molecules, if you have groups attached to a double bond, they can be on the same side (called 'cis') or on opposite sides (called 'trans'). For 1,2-dimethylcyclopentene, the way it's drawn implies a specific arrangement, but in reality, there could be different possibilities depending on how those methyl groups are oriented. It’s like a molecule having different dance poses!
This is where stereochemistry comes in, but don't worry, we're not going down that rabbit hole today. The point is, even small differences in how atoms are arranged in space can lead to big differences in how a molecule behaves. It's like the difference between a waltz and a tango – same dancers, different moves, totally different feel.
So, when you see a chemical name like 1,2-dimethylcyclopentene, don't just see a jumble of letters and numbers. See a story. See a shape. See a molecule with its own unique personality and potential. You’ve just decoded it, and hopefully, it feels a little less mysterious and a lot more cool.
Drawing these structures is a fundamental skill, but it's also a way to engage with the molecular world in a tangible way. It’s like sketching your thoughts – you’re bringing an idea to life visually. And for chemists, this visual language is essential for communicating complex ideas clearly and efficiently.
Next time you hear a chemical name, give it a try. Break it down. See if you can picture the shape. You might be surprised at how much you can understand, just by following the clues within the name itself. It’s a little bit of detective work, a little bit of art, and a whole lot of chemistry!
