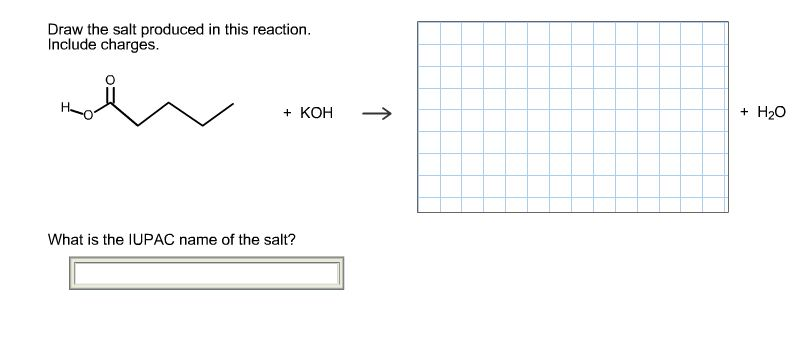
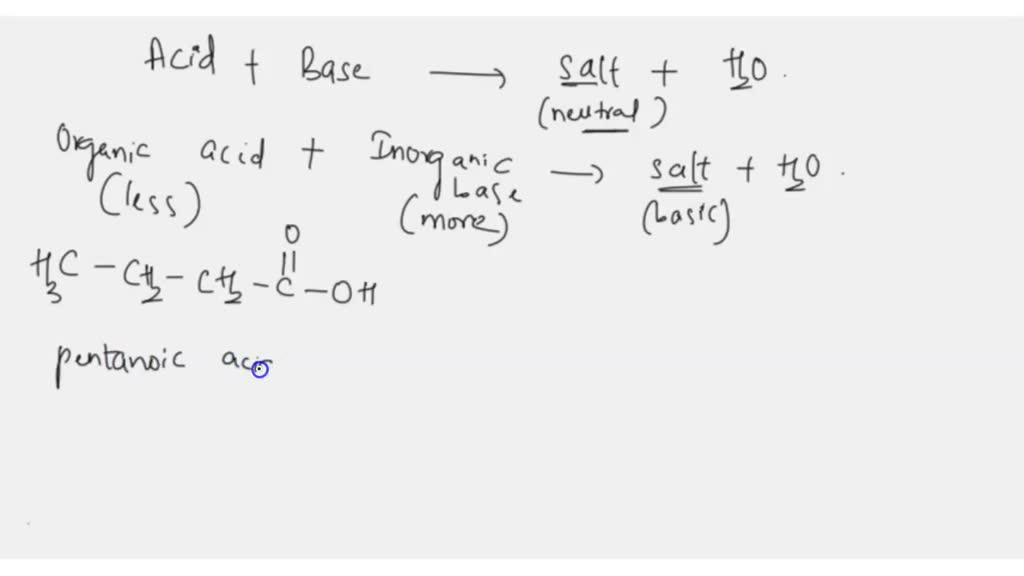
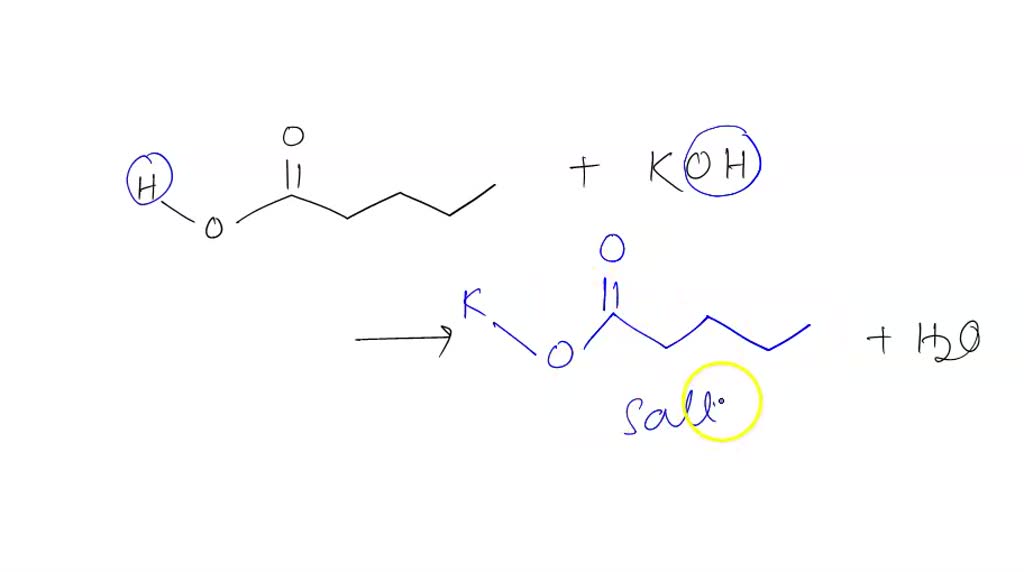
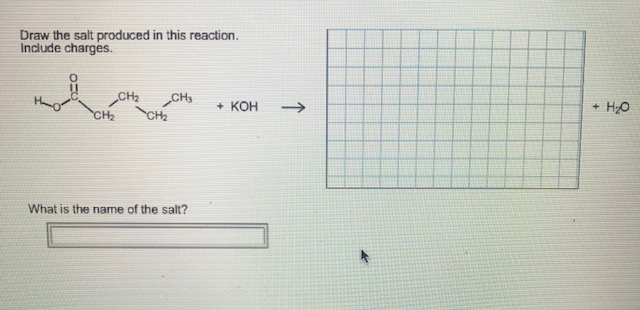
Draw The Salt Produced In This Reaction Include Charges

So, I was rummaging through my old science textbooks the other day, the kind with the smell of aged paper and forgotten experiments. You know, the ones that made you feel like a bona fide lab coat wearer, even if your most exciting experiment involved mixing baking soda and vinegar. Anyway, I stumbled across this diagram. It was of a chemical reaction, specifically one that produced a salt. And honestly, it looked like a kindergarten drawing gone wild. Little circles with pluses and minuses, arrows going everywhere… I swear, my first thought was, "Who drew this, a squirrel on caffeine?"
It got me thinking, though. How do we even know what these things look like? Or rather, how do we represent them so everyone else can understand? It's not like you can whip out a microscope and see an ion chilling with its buddies. We rely on these drawings, these symbolic languages, to make sense of the invisible world of atoms and molecules. And when it comes to salts, specifically the ones we produce in a reaction, there's a whole extra layer of "what's what."
You see, when you mix certain things together, magic (well, chemistry) happens. You get new stuff. And often, that new stuff is a salt. But what is a salt, really? Is it just the stuff you shake on your fries? (Spoiler alert: not entirely, though it is related!). And more importantly, what does it look like in terms of its electrical charge? Because, let me tell you, those little pluses and minuses aren't just for show. They're kind of the whole point.
So, buckle up, my fellow curious minds. Today, we're diving into the wonderfully weird world of drawing the salts produced in a reaction, complete with their charges. Don't worry, you don't need a lab coat, just your imagination and a willingness to embrace a little scientific scribbling. Think of me as your friendly neighborhood guide, leading you through the electrolyte jungle. No actual jungle animals involved, promise!
The Case of the Missing Charge (and How to Find It)
Let's start with a classic. Imagine you've got some hydrochloric acid (HCl) chilling in a beaker. Scary name, right? Sounds like it could dissolve your lunch. And it kinda can. Now, let's introduce it to some sodium hydroxide (NaOH). This is a base. Think of bases as the opposite of acids, like a friendly handshake after a tense argument. When these two meet, something pretty neat happens.
They react! And the main things you get out of this little shindig are water (H₂O) – that's the stuff you drink – and… you guessed it… a salt! In this case, it's sodium chloride (NaCl). Yep, the very same stuff that makes popcorn addictive and pickles, well, pickly. Pretty cool that something so mundane comes from such a dramatic-sounding reaction, wouldn't you say?
But here's where the drawing part gets interesting. We can't just draw a little box labeled "NaCl." That would be like describing a person by saying "they have a head and limbs." It's technically true, but it's missing all the details. And in chemistry, those details are often about charge. Because, you see, while NaCl as a whole compound is neutral, it's made up of tiny particles that aren't neutral. They have electrical charges. These are called ions.
So, how do we represent sodium chloride as ions with their charges? We break it down. Sodium (Na) is a metal. Metals tend to give away electrons when they react. Losing a negatively charged electron leaves them with a positive charge. So, sodium becomes Na⁺. See that little plus sign? That's its happy little charge. It's lost one electron, so it's got one more proton (positive charge) than electrons (negative charge). Simple, right? Almost too simple, makes you wonder what else is going on.
Chlorine (Cl), on the other hand, is a non-metal. Non-metals tend to gain electrons. It's like they're collecting them to feel complete. When chlorine gains an electron, it gets a negative charge. So, it becomes Cl⁻. That little minus sign means it's gained one extra electron, making it have one more electron than protons. See how the positive charge of sodium and the negative charge of chloride balance each other out? That's why the whole compound, NaCl, is neutral. It's all about electrical balance!
So, when we draw the salt produced in this reaction, we don't just draw "NaCl." We draw it as its constituent ions, showing their charges. You'd typically see something like: Na⁺ and Cl⁻. These are the building blocks of our table salt, and understanding their individual charges is key to understanding how they behave and interact.
The Art of the Ion: More Than Just Squiggles
Let's try another example, just to solidify this. Imagine we react sulfuric acid (H₂SO₄) with potassium hydroxide (KOH). Again, scary names, but trust me, it's more about the outcome. This reaction will produce water and a salt called potassium sulfate (K₂SO₄).
Now, let's break down potassium sulfate. Potassium (K) is another metal, part of the same family as sodium on the periodic table. What do you think that means for its charge? You got it! It likes to lose an electron, so it becomes K⁺. But here's a twist: in potassium sulfate, there are two potassium atoms for every sulfate group. So, we actually have two K⁺ ions.
What about the sulfate part? That's the SO₄ group. This is a bit more complex because it's a polyatomic ion. That means it's a group of atoms (one sulfur and four oxygens) that stick together and carry a charge as a unit. In the case of sulfate, it has a charge of 2⁻. So, it's SO₄²⁻. This group of atoms collectively has gained two extra electrons.
So, to draw the salt produced here, we'd represent it as: two K⁺ ions and one SO₄²⁻ ion. You can see how the charges balance out again: (+1) + (+1) + (-2) = 0. It's like a chemical balancing act! And this is crucial for understanding how these salts dissolve in water, conduct electricity, and participate in other reactions. The charges are the engine that drives their behavior.
When you're asked to draw these, it's not about artistic skill, but about accurately representing the chemical species. You'd typically use element symbols, with the charge as a superscript next to it. For polyatomic ions, the entire group is enclosed in parentheses before the charge is written, like (SO₄)²⁻. This is a universally understood shorthand in chemistry. Think of it as the alphabet and grammar of the molecular world. And if you mess up the grammar, you might end up with a very different molecule!
Why Do We Bother With All This Charge Stuff?
Okay, I get it. Sometimes, drawing little boxes with pluses and minuses can feel a bit tedious. You might be thinking, "Can't we just say 'potassium sulfate' and be done with it?" Well, you can, but you'd be missing a huge chunk of the story.
Understanding the charges of ions is fundamental to chemistry. It tells us: * How ions attract each other: Opposite charges attract, like magnets! This is why Na⁺ and Cl⁻ stick together to form NaCl. It's a powerful electrostatic attraction. * How salts dissolve in water: When a salt dissolves, it breaks apart into its individual ions. Water molecules, which are polar (they have a slightly positive and slightly negative end), surround these ions, keeping them separated. This is why salt water conducts electricity – the mobile ions act as charge carriers. * How salts react with other substances: The reactivity of a salt is heavily dependent on the charges and sizes of its constituent ions. * The properties of the salt: Melting point, solubility, and even color can be influenced by the ionic interactions.
So, when you're asked to draw the salt produced in this reaction and include charges, it's not just a drawing exercise. It's a way of visually representing the fundamental nature of the substance. It's showing that it's not just a collection of atoms, but a structured arrangement of charged particles.
Think about it this way: if you were describing a superhero, would you just say "they can fly"? Or would you mention their super strength, their invulnerability, their laser eyes? The charges on ions are like those superpowers. They define what the salt is and what it can do. And trust me, in the chemical world, these powers are pretty impressive.
Putting It All Together: Your Turn!
So, the next time you encounter a chemical reaction that produces a salt, take a moment. Identify the cation (the positive ion, usually a metal) and the anion (the negative ion, often a non-metal or polyatomic ion). Then, recall (or look up!) their typical charges. Draw them out, making sure those little pluses and minuses are clear.
For instance, if you react magnesium hydroxide (Mg(OH)₂) with nitric acid (HNO₃), you'll get water and magnesium nitrate (Mg(NO₃)₂). Magnesium (Mg) is in the same group as calcium, and it likes to lose two electrons, so it's Mg²⁺. The nitrate ion (NO₃) is another polyatomic ion, and it carries a charge of 1⁻, so it's NO₃⁻.
Since magnesium has a 2⁺ charge and nitrate has a 1⁻ charge, you need two nitrate ions to balance out one magnesium ion. So, the salt produced, magnesium nitrate, would be drawn as: one Mg²⁺ and two NO₃⁻ ions.
It might seem a little quirky at first, this whole drawing-charged-particles thing. But it's the language of chemistry, and once you get the hang of it, it opens up a whole new understanding of the world around you. From the salt on your table to the minerals in the earth, they all owe their existence and properties to these fundamental charged entities. So go forth, draw those ions, and embrace the electrifying world of chemical salts! And maybe, just maybe, your drawings will be a little less squirrel-on-caffeine and a lot more scientifically sound.
