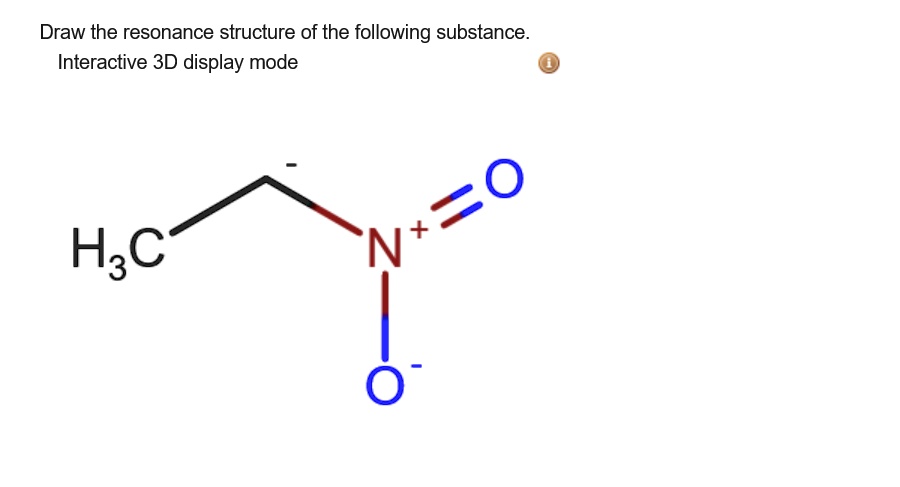
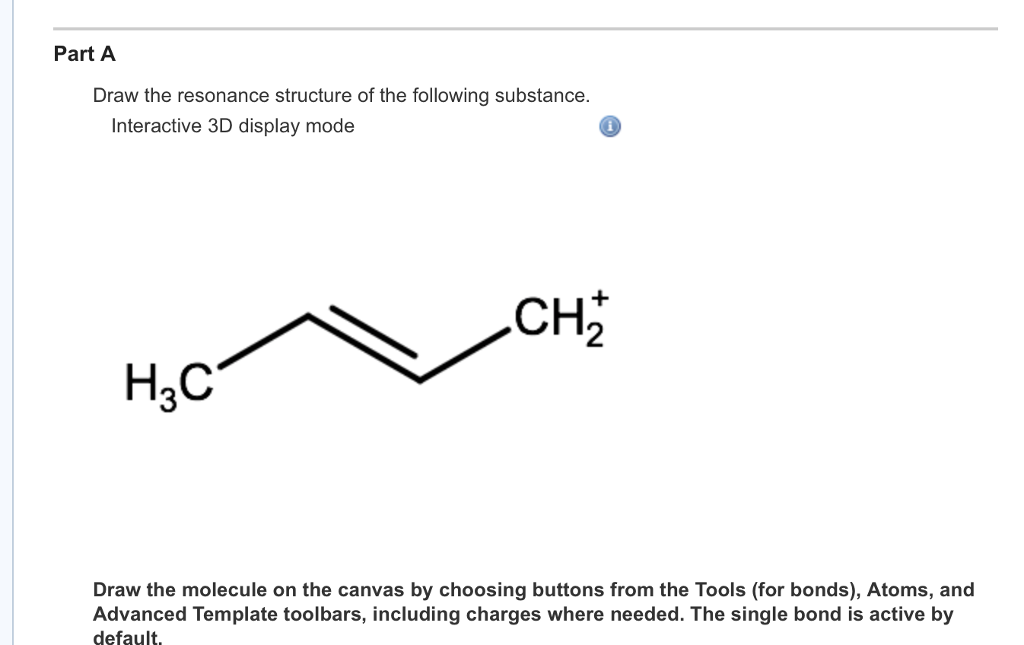
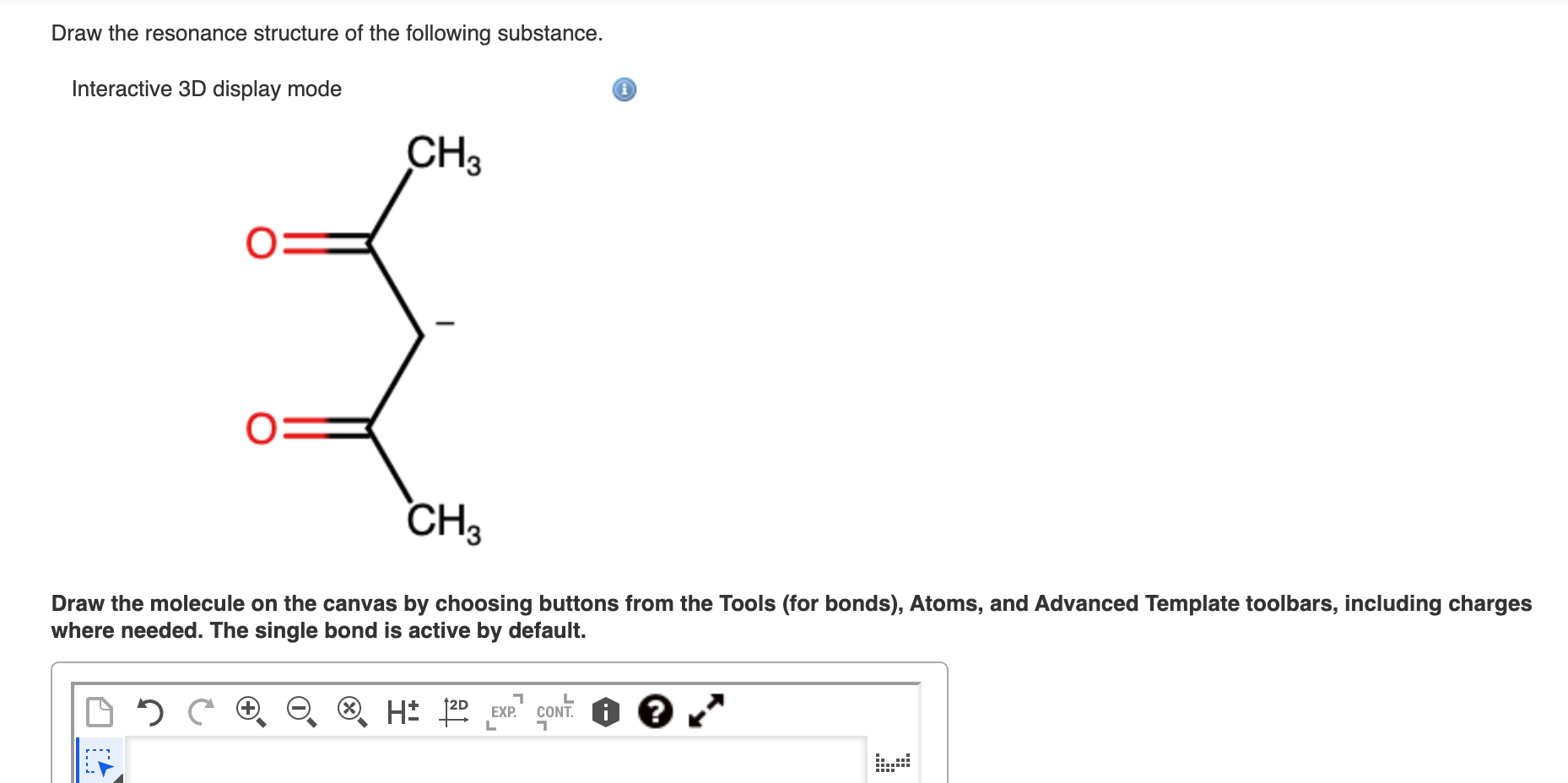
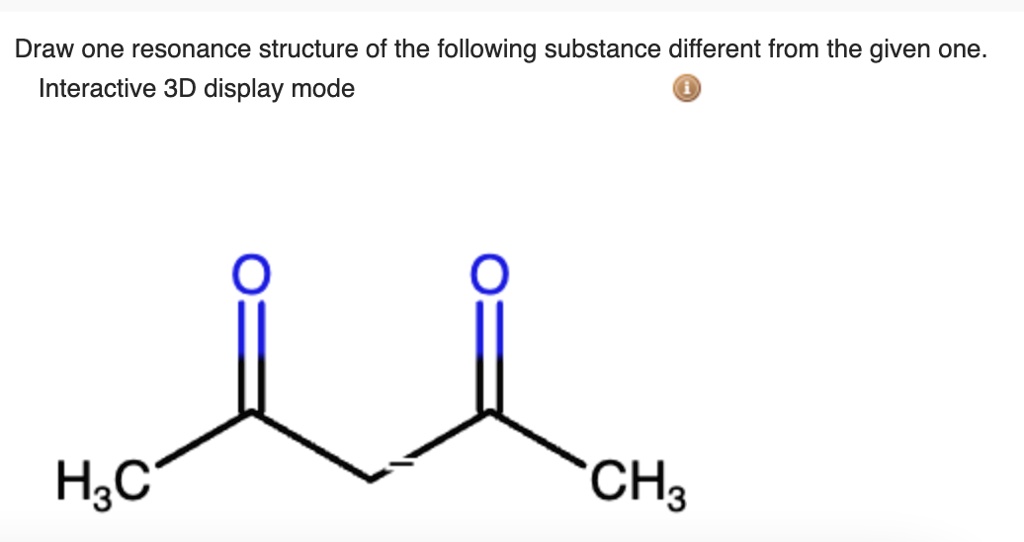
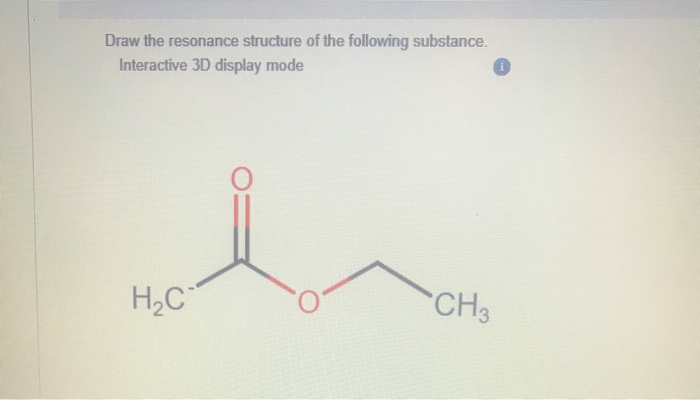
Draw The Resonance Structure Of The Following Substance
Hey there, fellow curious minds! Ever found yourself staring at a chemical formula and thinking, "What's the real story behind this molecule?" Well, buckle up, because we're about to dive into a super cool concept that’s not just for the lab coat crowd. We're talking about resonance structures, and trust me, it’s way more fun than it sounds!
Think of it like this: sometimes, one picture just doesn't capture the whole essence of something, right? Like trying to describe your best friend with just one emoji. It’s just… incomplete. Molecules are kind of the same way! They can be a bit of a shape-shifter, or rather, they can be represented as a shape-shifter.
So, what exactly are these "resonance structures" we keep hearing about? Imagine a molecule is like a rockstar. This rockstar has multiple amazing poses, right? One day they're shredding on guitar, the next they're belting out a ballad, and another they're doing a mic drop. Resonance structures are like those different, equally valid "poses" of a molecule. They help us understand its true nature, which is often a blend of these poses.
The substance we’re going to "draw the resonance structures of" – don't let the fancy phrasing scare you! – is actually pretty common and incredibly important. It’s something you encounter all the time, even if you don't realize it. We're talking about the molecule that makes your car run, that you breathe in (and hopefully out!), and that’s a fundamental building block of so many things: carbon dioxide. Yep, CO2!
Now, you might think, "Carbon dioxide? That's the stuff we're trying to reduce for the planet!" And you'd be absolutely right! But understanding its molecular structure, and especially its resonance, is a key step in figuring out how it interacts with other things, and ultimately, how we can manage it. So, in a way, learning about resonance is like gaining a superpower to better understand and perhaps even help our world!
Let's Get Visual (Sort Of!)
So, how do we actually "draw" these resonance structures? It's all about understanding where electrons hang out. Electrons are the super energetic, often mischievous, little particles that hold atoms together in molecules. In resonance, these electrons aren't stuck in just one place. They’re a bit more… delocalized.
Imagine you have a group of friends playing tug-of-war. In one snapshot, it looks like one team is winning. But the game is constantly shifting, and the rope is moving back and forth. Resonance structures are like taking multiple snapshots of that tug-of-war, showing the rope in slightly different positions. None of those individual snapshots is the entire story, but together, they give you a much better idea of the dynamic energy of the game.
For carbon dioxide (CO2), we have one carbon atom and two oxygen atoms. The carbon atom is in the middle, and the oxygens are on either side. Now, the simplest way you might draw CO2 is with double bonds between the carbon and each oxygen. That looks like O=C=O. Seems pretty straightforward, right?
But here’s where the magic of resonance comes in. It turns out that CO2 isn't just that O=C=O picture. The electrons can rearrange themselves. We can also have structures where there's a triple bond on one side and a single bond on the other. So, you might see something like
-O≡C-O+
+O-C≡O-
.What's the Big Deal?
Why do we need to consider these different "drawings"? Because the actual molecule of carbon dioxide isn't perfectly represented by any single one of these structures. It's like a hybrid, a blend of all of them. The real CO2 molecule is somewhere in between these different electron arrangements. This blending is called the resonance hybrid.
Think of it like mixing paint colors. If you mix red and blue, you don't get just red or just blue. You get purple! The purple is the resonance hybrid, and the red and blue are like the contributing resonance structures. The hybrid is often more stable and has different properties than any of the individual contributing structures alone.
In the case of CO2, this resonance means the bonds between the carbon and oxygen aren't perfectly double bonds or perfectly triple/single bonds. They have characteristics of both. This subtle electron dance makes CO2 a very stable molecule, which is why it hangs around in our atmosphere. Pretty neat, huh?
Making Chemistry Fun (Yes, Really!)
So, why is this fun? Because it’s like being a detective! You’re not just memorizing facts; you’re uncovering the hidden life of molecules. When you look at a chemical reaction, understanding resonance helps you predict what might happen. It’s like knowing your rockstar friend has different moves – you can anticipate their performances!
It adds a layer of depth and intrigue to the world around you. Every time you see something made of molecules – which is everything – you can imagine these fascinating electron arrangements at play. It’s like having a secret decoder ring for the universe!
And the best part? This concept of resonance isn't limited to carbon dioxide. It applies to so many other molecules, from the DNA in your cells to the flavors in your food. Understanding resonance is like unlocking a fundamental language of chemistry.
Learning to draw these resonance structures might seem a bit daunting at first, like learning a new dance step. But with a little practice, you'll start to see the patterns. You’ll begin to appreciate the elegance and complexity of even the simplest-seeming substances.
So, the next time you hear about "drawing resonance structures," don't groan! Instead, think of it as an invitation to explore, to visualize, and to understand the dynamic, energetic world of molecules. It’s a journey into the heart of matter, and believe me, it’s an adventure worth taking. Keep that curiosity alive, and you’ll find endless wonder in the unseen!
