Draw The Products Formed From The Ester Hydrolysis Reaction Shown.

Hey there, coffee buddy! So, you wanna talk about ester hydrolysis? Sounds fancy, right? But honestly, it's not as intimidating as it sounds. Think of it like this: we're basically taking something that's a little bit stuck together, and we're giving it a good old shake-up with some water. Boom! New things appear. Pretty neat, huh?
Imagine you've got this ester. What's an ester, you ask? Well, think of a delicious fruity smell. That often comes from esters! They're kind of like the flavor-makers of the world. They have this special little bond, this 'ester linkage,' that's just begging to be broken. And guess what loves to break things? Water! Yes, good old H₂O, our best friend in the lab and in life.
So, when we do ester hydrolysis, it's like we're inviting water over for a party with the ester. Water's a bit of a party animal, you see. It crashes the ester linkage and splits it right down the middle. And when it splits, things change. Magic happens, or at least, chemistry happens. It's all about making new friends, you know?
Let's get a little more specific, shall we? We're going to draw the products formed from the ester hydrolysis reaction. Sounds like homework, but it's more like a little puzzle. And who doesn't love puzzles, especially when there's coffee involved? This is where you grab your pen, your imaginary paper, or your actual tablet, and let's get sketching. Don't worry, I'll guide you through it. We're a team here!
First off, let's visualize our starting ester. It's got this characteristic part. Think of it like a little yin-yang symbol, but with atoms. You've got a carbonyl group (that's a carbon double-bonded to an oxygen, like a grumpy face) attached to an oxygen atom. This oxygen is then attached to another carbon group. That's your ester linkage right there. That's the spot we're targeting, the weak spot, if you will. It's like the Achilles' heel of the ester molecule. And water is ready to exploit it!
Now, when water (H₂O) comes in, it's not just a passive observer. It's an active participant. Water, as you know, is made of two hydrogen atoms and one oxygen. So, it's got these little bits that it can offer up. Think of it like a generous friend at the party, handing out favors.
The water molecule is going to attack that ester linkage. Specifically, it's the oxygen in the water molecule that's going to be the main aggressor, or rather, the main helper. It's going to swoop in and, with a little help from either an acid or a base (we'll get to that!), it's going to break that bond.
Imagine the ester linkage as a little bridge. Water comes along and, with a bit of a shove, it breaks that bridge. And when the bridge breaks, what's on either side? Two separate pieces! These are our new products. Ta-da! It's like a molecular divorce, but a friendly one. Everyone gets something to take home.
So, what are these two pieces? One of them is going to be an alcohol. Yep, just like the stuff you might find in, well, certain beverages. This alcohol part comes from the part of the ester that was attached to the oxygen atom in the ester linkage. It's usually got an -OH group hanging off it. Pretty recognizable, right?
The other piece? That's going to be a carboxylic acid. Now, this one's a bit different. It also has an -OH group, but it's attached to a carbonyl group. So, it's got that grumpy face (C=O) right next to an -OH. This is the "acid" part of "carboxylic acid." It's got a certain zing to it, you know? It's the component that gives vinegar its sourness, for example. So, it's not just abstract chemistry; it's stuff we encounter every day!
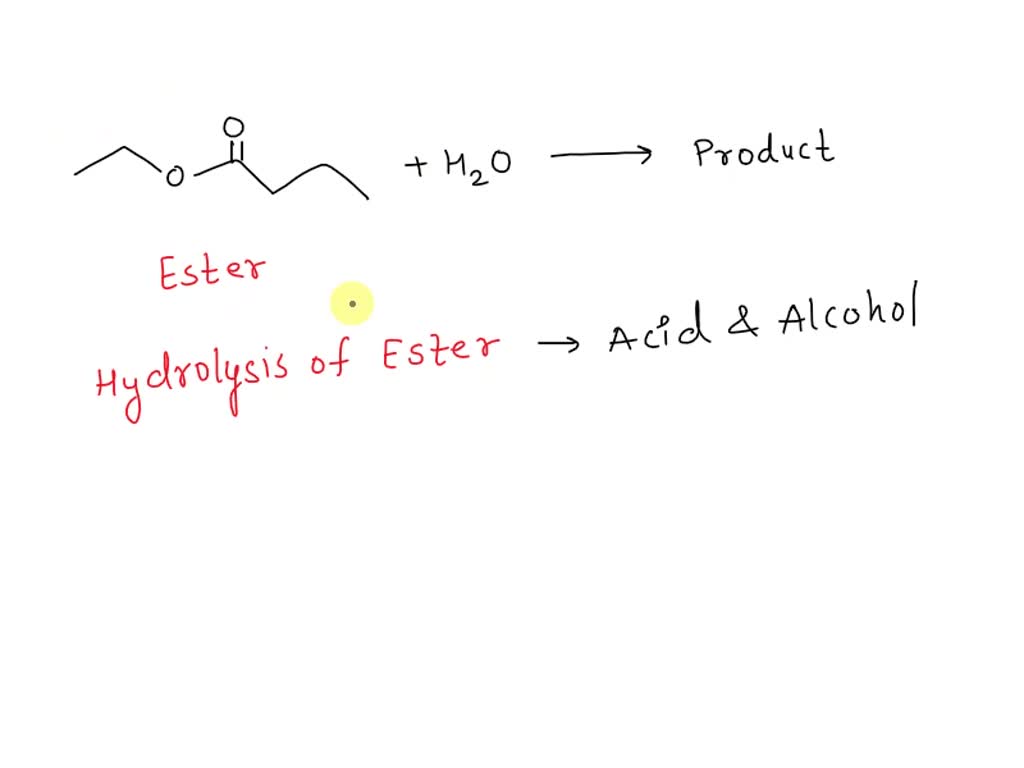
Let's break down the drawing part. Say you have a generic ester. Let's call it R-COO-R'. The 'R' and 'R'' are just placeholders for different carbon chains or even just single hydrogen atoms. It's like saying "some stuff" and "other stuff." So, R is one part, and R' is the other part. The -COO- is the ester linkage, the heart of the matter.
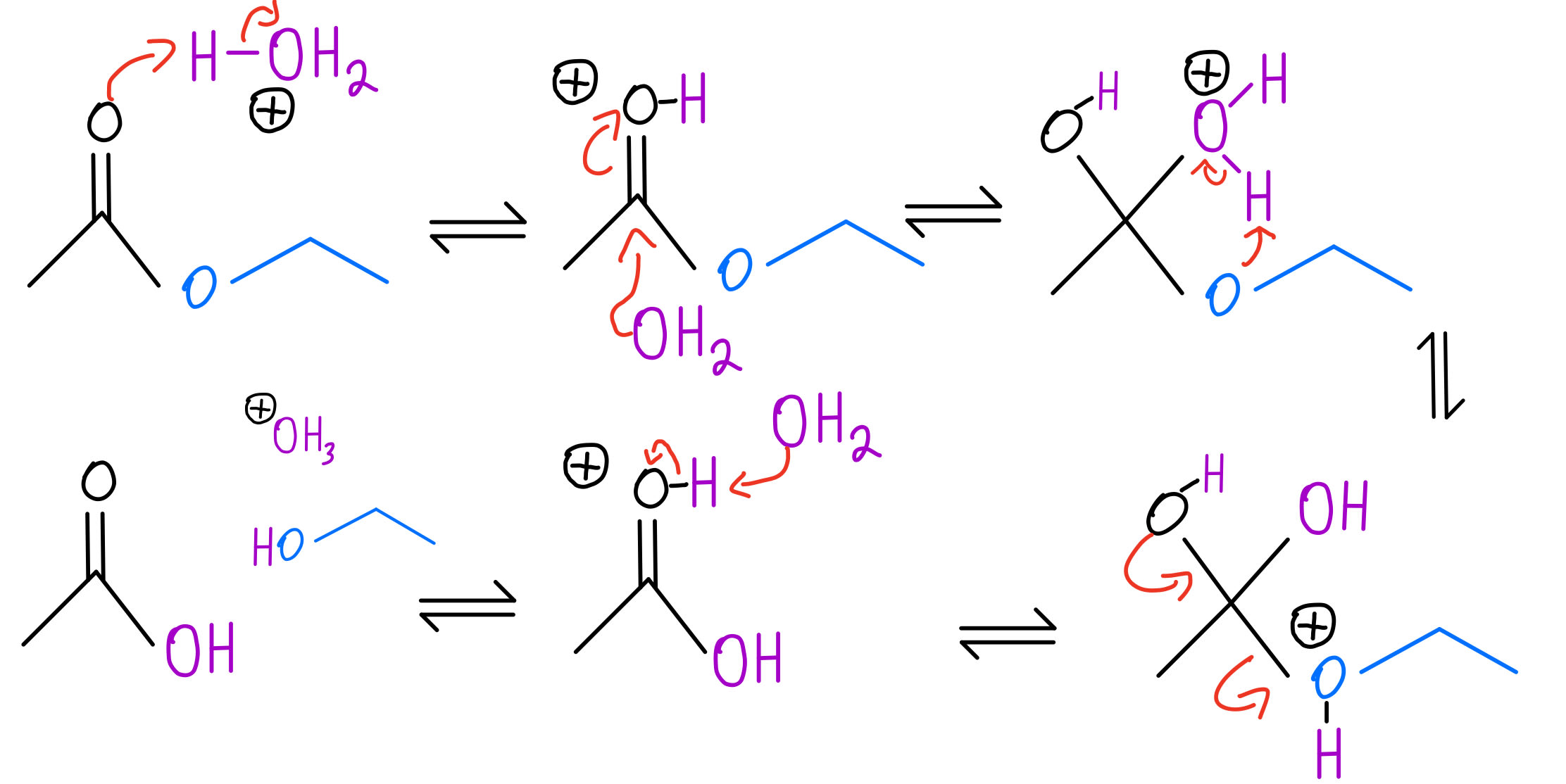
When water, H₂O, comes along, it's like a two-pronged attack. One hydrogen from the water molecule is going to attach itself to the oxygen that was originally double-bonded to the carbonyl carbon. And the other part of the water molecule, the hydroxide ion (OH⁻), is going to grab onto the carbon atom of the carbonyl group. Or, depending on the mechanism, the water molecule can attack the carbonyl carbon directly, and then a proton transfer happens. Either way, the result is the same!
So, the ester linkage -COO- gets broken. The R- group, which was attached to the carbonyl carbon, will end up attached to the original carbonyl oxygen plus a hydrogen. That gives you R-COOH, your carboxylic acid. And the R'- group, which was attached to the other oxygen of the ester linkage, will end up with the rest of the water molecule attached, which is the -OH group. So, you get R'-OH, your alcohol.
Acidic Hydrolysis: The Strong Arm of the Law
Now, sometimes, this reaction needs a little nudge. It can happen on its own, but it's often slow. So, we bring in some reinforcements. One way to speed it up is by using an acid. Think of an acid as a very persuasive negotiator. It's like, "Come on, ester, let's break this bond! I'll help you out!"
In acidic hydrolysis, we usually add a strong acid like hydrochloric acid (HCl) or sulfuric acid (H₂SO₄). The acid's job is to protonate the carbonyl oxygen. That makes the carbonyl carbon even more positive, making it a prime target for the water molecule to attack. It's like making the grumpy face even grumpier, so it's more attractive to new friends.
Once the water attacks and the bond breaks, you get your alcohol (R'-OH) and your carboxylic acid (R-COOH). But here's a little twist in acidic conditions: carboxylic acids are, well, acids. And alcohols are pretty neutral. If there's enough acid present, the carboxylic acid can actually get protonated too, but the primary products we're usually concerned with are the alcohol and the free carboxylic acid. It's like the alcohol is the chill one, and the carboxylic acid is the one that still needs to sort itself out.
The key thing to remember in acidic hydrolysis is that it's often a reversible reaction. That means the alcohol and carboxylic acid can actually come back together to form the ester again. We call this esterification. So, it's a bit of a back-and-forth. To get the ester to break completely, you often need to use a large excess of water or remove one of the products as it forms. It's like trying to get a stubborn couple to split up – sometimes you need to take drastic measures!

So, when you're drawing, make sure to show the starting ester, the water molecule (H₂O), and then your two products: the alcohol and the carboxylic acid. And don't forget to indicate that there's an acid catalyst present, usually written above the arrow of the reaction. It's like a little note saying, "This happened with a little help!"
Basic Hydrolysis: The Helping Hand of a Base
Now, let's talk about the other side of the coin: basic hydrolysis. This is like having a really supportive friend, a base, who's going to help break things apart. Instead of an acid, we use a base, like sodium hydroxide (NaOH) or potassium hydroxide (KOH).
In basic hydrolysis, the hydroxide ion (OH⁻) from the base is the star of the show. It directly attacks the carbonyl carbon of the ester. This is a much more direct approach than the acidic method. It's like, "Okay, let's just get this done!" The hydroxide ion is a strong nucleophile, meaning it's attracted to positive charges, and that carbonyl carbon is definitely looking a little positive.
When the hydroxide ion attacks, it forms a tetrahedral intermediate – fancy name for a temporary structure where the carbon is temporarily bonded to five things. Then, the ester linkage breaks. One part goes off as the alcohol (R'-OH), just like before. But the other part, the R-COO⁻, doesn't immediately become a carboxylic acid. Instead, it becomes a carboxylate anion. It's like the carboxylic acid lost its acidic hydrogen, and it's now negatively charged. It's got a bit of an attitude, you could say.
So, in basic hydrolysis, you get an alcohol and a carboxylate salt. For example, if you used NaOH, you'd get R-COO⁻Na⁺. This is often a precipitate or something that's easily separated. This is actually a really useful reaction because the carboxylate salt is usually stable and doesn't readily react to reform the ester. It's like a permanent separation!
The beauty of basic hydrolysis is that it's generally irreversible. Once you've made that carboxylate salt, it's pretty much stuck that way. You can then add an acid later to protonate the carboxylate anion and get your carboxylic acid, if that's what you need. But the initial hydrolysis itself is a one-way street, which is often super convenient for synthetic chemists. No fiddling around with equilibrium!
When you're drawing the products of basic hydrolysis, you'll show the starting ester, the base (like NaOH), and then the alcohol (R'-OH) and the carboxylate salt (R-COO⁻Na⁺). Remember, that R-COO⁻ part has a negative charge on one of the oxygen atoms, and the positive ion from the base (like Na⁺) is there to balance it out. It's like a little ionic dance.
Let's Get Visual: Drawing Time!
Alright, enough talk, let's draw! Imagine you have methyl acetate. It's a simple ester, like the stuff that gives nail polish remover its smell (don't try that at home, though!). Methyl acetate has the structure CH₃-COO-CH₃.
So, our R group is CH₃ (a methyl group), and our R' group is also CH₃ (another methyl group). We're going to break that -COO- bond in the middle.
Acidic Hydrolysis of Methyl Acetate
If we do this with acid and water (let's say, H⁺/H₂O), the CH₃-COO- part will become CH₃-COOH. That's acetic acid. And the -OCH₃ part will become CH₃-OH. That's methanol. So, your products are acetic acid and methanol. You'd draw CH₃-COO-CH₃ + H₂O (with H⁺ above the arrow) → CH₃-COOH + CH₃-OH.
Look at it! The carbonyl carbon is still there, with its grumpy face. It's now bonded to an -OH. The methyl group that was attached to that carbonyl carbon is now part of the acetic acid. The other methyl group, the one that was attached to the oxygen, is now a standalone methanol molecule with its own -OH.
Basic Hydrolysis of Methyl Acetate
Now, if we do the same with a base, like NaOH, and water (NaOH/H₂O), the CH₃-COO- part becomes CH₃-COO⁻Na⁺. That's sodium acetate. And the -OCH₃ part becomes CH₃-OH. That's still methanol. So, your products are sodium acetate and methanol. You'd draw CH₃-COO-CH₃ + NaOH → CH₃-OH + CH₃-COO⁻Na⁺.

See the difference? In the basic hydrolysis, that acetate part has a negative charge on one of the oxygens, and the sodium ion is hanging out with it. It's a salt, not a free acid. The methanol is the same in both cases, which makes sense because it's the R' group that's becoming the alcohol. The R group, which was attached to the carbonyl, is the one that becomes either the carboxylic acid or the carboxylate salt.
It's like a family reunion where one side of the family splits off and forms a stable partnership (the salt), while the other side goes off on its own adventure (the alcohol). And in the acidic case, it's more like a temporary separation where they could potentially get back together.
Why Does This Even Matter?
You might be thinking, "Okay, cool, I can draw some molecules. But why should I care?" Well, ester hydrolysis is super important in so many areas! Think about digestion. Our bodies use enzymes that are basically catalysts for hydrolysis reactions to break down fats (which are esters) into fatty acids and glycerol. It's how we get energy from the food we eat. How amazing is that?
In the industry, it's used to make soaps! Saponification, which is basically basic hydrolysis of fats, produces soap (salts of fatty acids) and glycerol. So, the next time you wash your hands, you can thank ester hydrolysis. You're literally washing away with the products of this reaction!
It's also crucial in the synthesis of other organic compounds. Need a specific acid or alcohol? Hydrolysis can be a key step in getting there. It's a fundamental reaction that unlocks a lot of possibilities in the world of organic chemistry.
So, when you're faced with a drawing problem for ester hydrolysis, don't panic. Just remember the basics: water is involved, the ester linkage breaks, and you end up with an alcohol and either a carboxylic acid (in acidic conditions) or a carboxylate salt (in basic conditions). Think of it as two different ways to achieve a similar outcome, each with its own unique flavor and mechanism. Just like choosing between a strong espresso and a creamy latte – both get you going, but in slightly different ways!
Keep practicing, keep sketching, and most importantly, keep enjoying your coffee and the fascinating world of chemistry. It's a journey, and with a little understanding and a lot of drawing, you'll master these reactions in no time. You've got this!
