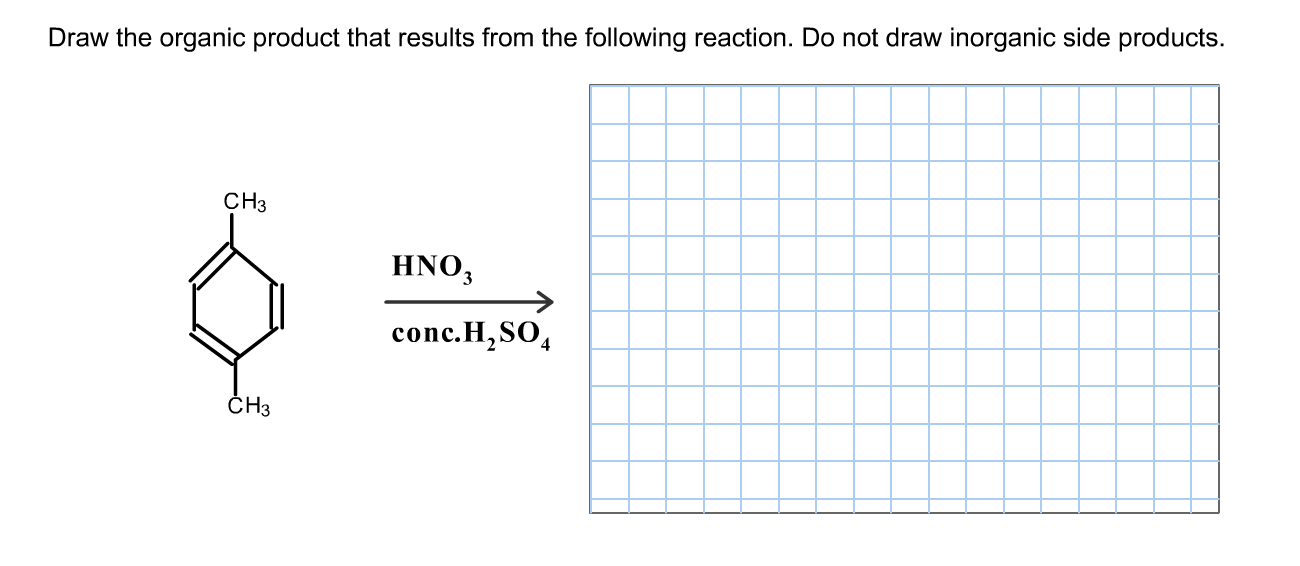
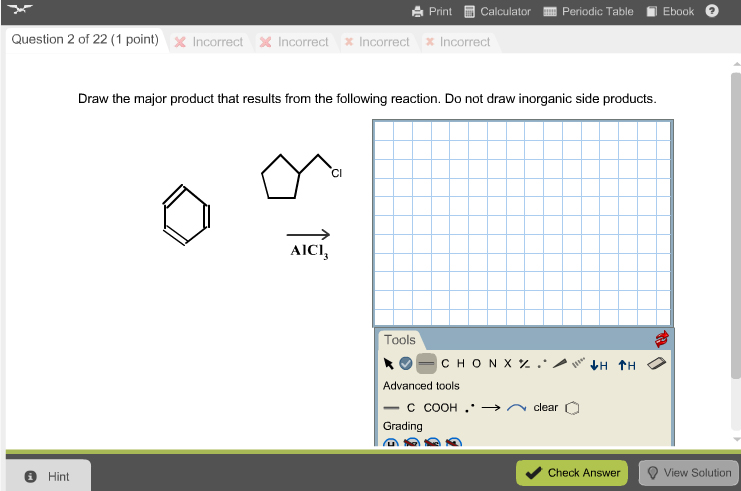
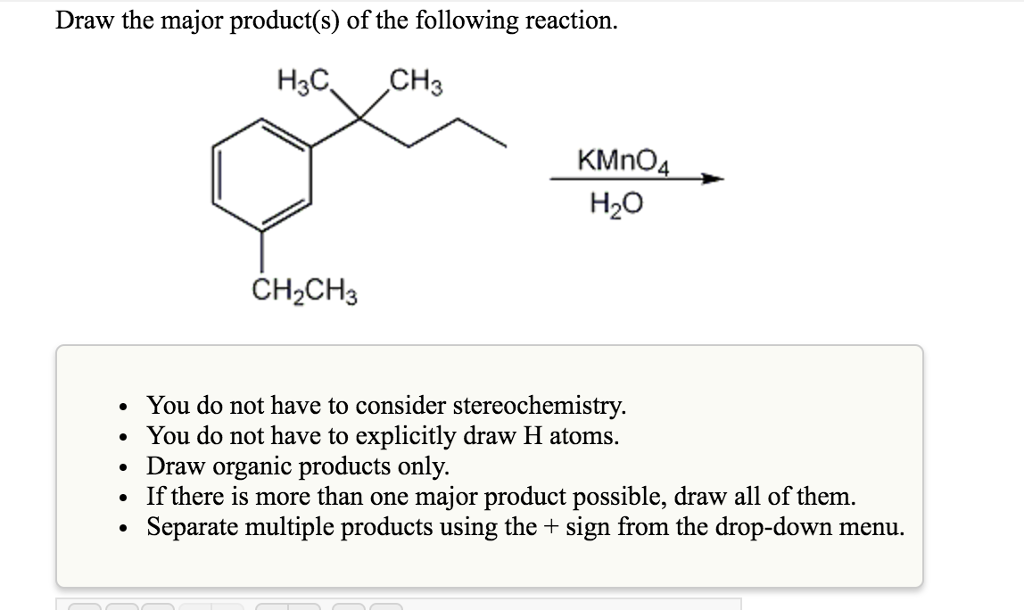
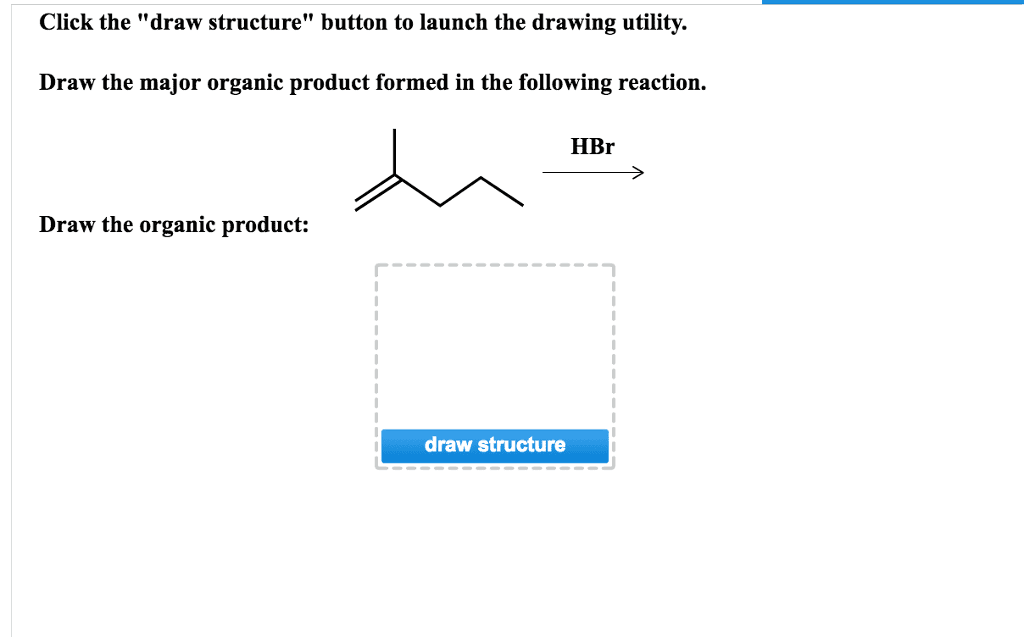
Draw The Product That Results From The Following Reaction.
Imagine you're a chef, but instead of a kitchen, your workspace is a microscopic laboratory, and your ingredients are tiny, invisible building blocks. Today, we're going to peek behind the scenes of a really cool chemical reaction, like watching a magic trick unfold! We're going to see what happens when we mix some familiar characters together and what amazing new thing they create.
Think of it like this: you know how LEGOs snap together to make something new? Chemistry is a bit like that, but with atoms and molecules. They bump into each other, rearrange themselves, and poof – something entirely different emerges. It’s a world of constant transformation, and we get to witness one such transformation right now.
Our story today involves some very special "ingredients." We're going to be looking at something called a Grignard reagent. Now, that might sound a bit fancy, but think of it as a super-powered worker molecule. It’s got a bit of a "get things done" attitude, ready to join forces with others.
The other main player in our little chemical drama is something called an aldehyde. Aldehydes are like the friendly neighborhood shopkeepers of the molecule world. They’re quite common and often have a pleasant smell, like the scent of a ripe apple or a fragrant flower. They’re happy to offer up a part of themselves to a willing partner.
So, what happens when our enthusiastic Grignard reagent meets our accommodating aldehyde? It’s a bit like a shy handshake turning into a warm hug. The Grignard reagent, with its energetic personality, reaches out to the aldehyde.
This isn't a violent clash; it's a cooperative dance. The Grignard reagent has a special bit, a magnesium-carbon bond, that's eager to connect. The aldehyde has a spot, a carbon-oxygen double bond, just waiting for a new friend.
When they meet, the Grignard reagent practically leaps at the opportunity. It shares its carbon atom, forming a brand new connection with the carbon atom of the aldehyde. It’s a bit like a baker adding a special ingredient to a cake batter.
And what’s the result of this little molecular rendezvous? It’s a brand new molecule! This new creation is a type of molecule called an alkoxide. Think of it as the happy couple after their first dance, now a new unit.
But our story doesn't quite end there. This alkoxide is a little unstable, like a newborn baby needing some care. It’s got a negative charge hanging around, and it’s looking for something to balance it out.
This is where a simple proton, basically a tiny positively charged hydrogen atom, comes in. Imagine it as a kind nurse coming to soothe the alkoxide. The alkoxide readily accepts this proton.
And voilà! The alkoxide accepts the proton and transforms into something even more special: an alcohol! Yes, that’s right, the same family of compounds that give us our favorite refreshing drinks and are used in everyday cleaning products.
So, in essence, our Grignard reagent and our aldehyde, through this gentle chemical exchange, have given birth to an alcohol. It’s a bit like two ingredients combining to make a delicious sauce for a meal.
Now, the exact alcohol that is formed depends on the specific aldehyde we start with. If we begin with a very simple aldehyde, like formaldehyde (which sounds a bit serious, but it's quite small!), we end up with a very simple alcohol, called methanol. Methanol is the simplest of the alcohols.
Methanol is a clear liquid and is used in many industrial processes. It’s a workhorse molecule, but its creation from such a dynamic reaction is pretty neat.
If we use a slightly more complex aldehyde, like acetaldehyde (imagine it as formaldehyde's slightly older sibling), we get a different alcohol. This time, we create ethanol. This is the alcohol found in alcoholic beverages, but it’s also used for much more, like as a disinfectant.
It’s quite a journey from the initial handshake between the Grignard reagent and the aldehyde to the formation of these useful alcohols. Each step is a little piece of molecular choreography.
The beauty of this reaction, known as the Grignard reaction, is its versatility. By simply changing the aldehyde we use, chemists can create a whole pantry full of different alcohols. It’s like having a master key that unlocks many doors.
This reaction is a cornerstone of organic chemistry, meaning the chemistry of carbon-based molecules that make up life. It’s a fundamental tool for building larger and more complex molecules from simpler ones. Think of it as a crucial step in building a detailed LEGO model, adding essential pieces to create intricate structures.
And the best part? This reaction is often carried out under relatively mild conditions. It doesn't require extreme heat or pressure, making it a bit more gentle and energy-efficient. It’s a bit like baking a cake at a comfortable oven temperature, rather than trying to cook it over a volcano.
So, when you see a bottle of rubbing alcohol, or even enjoy a responsibly consumed beverage containing ethanol, remember the fascinating journey it took to get there. It involved a dynamic duo – the Grignard reagent and the aldehyde – performing a little molecular magic.
The Grignard reagent, with its inherent reactivity, and the aldehyde, with its welcoming nature, come together. They form an intermediate, a temporary structure, that is then stabilized.
This transformation is a testament to the elegant simplicity and profound power of chemical reactions. It shows how even the smallest building blocks can be manipulated to create substances that are essential to our world.
It's a process that's both scientifically significant and surprisingly accessible when you break it down. We’re not just mixing chemicals; we’re witnessing a creative act at the molecular level.
From the initial spark of reactivity to the final, stable product, it's a story of partnership and transformation. A story that plays out every day in laboratories around the globe.
So, next time you encounter an alcohol, whether for practical purposes or a moment of enjoyment, take a moment to appreciate the clever chemistry that brought it into existence. It’s a small wonder of the molecular world.

The Grignard reaction is a beautiful example of how chemists can precisely build molecules, much like an artist paints a masterpiece or a sculptor shapes clay.
It's a reminder that even the most common substances have a remarkable origin story, rooted in the fascinating interactions of atoms and molecules.
And the product? It's an alcohol, a versatile and important compound, brought to life through a delightful dance of chemical bonds.
It's a process that highlights the ingenuity and artistry inherent in chemistry, making the familiar seem wonderfully new.
The Grignard reagent, the aldehyde, and the resulting alcohol are all part of a grand molecular narrative.
This reaction shows us that even at the smallest scales, there's a world of creativity and transformation waiting to be discovered.
