Draw The Product Of The Hydrogenation Of Ethyne

Ever wondered what happens when you take a super simple building block of chemistry and give it a little makeover? Well, get ready for some fun because we're diving into the world of ethyne, also known as acetylene, and its transformation through a process called hydrogenation. It might sound a bit science-y, but trust us, it's fascinating and has some really cool real-world applications. Think of it like a chemical glow-up, turning something with a triple bond into something even more useful and stable. It's like taking a very energetic, slightly wild molecule and making it a bit more chill and adaptable.
So, why is this particular chemical reaction so interesting to talk about? For starters, ethyne itself is a pretty exciting molecule. It's the simplest alkyne, boasting a triple bond between two carbon atoms. This triple bond is packed with energy and ready to react, making ethyne a versatile starting material in many chemical processes. However, that high reactivity also means it can be a bit tricky to handle. Hydrogenation is our way of taming that energy, adding hydrogen atoms to break those triple bonds and create new, more stable compounds. It’s a controlled transformation that unlocks new potential.
The beauty of hydrogenation lies in its ability to precisely modify molecules. When we hydrogenate ethyne, we’re essentially adding hydrogen atoms across its triple bond. This process can occur in stages. The first stage adds one molecule of hydrogen, converting the triple bond into a double bond, resulting in ethene (or ethylene). If we keep going and add another molecule of hydrogen, we completely saturate the molecule, breaking the double bond and forming a single bond. This leads us to ethane, a very stable and common hydrocarbon.
The purpose of drawing the product of ethyne's hydrogenation is to visualize this molecular transformation. It's a fantastic way to understand how chemical structures change and how those changes lead to different properties. By sketching out the molecules before and after the reaction, you can literally see the bonds being added and the molecule becoming more saturated. This visual learning is incredibly powerful, especially when you’re first getting to grips with organic chemistry. It bridges the gap between abstract chemical formulas and the tangible arrangement of atoms.
Visualizing the Transformation
Let's get down to the fun part: drawing! When we talk about the product of ethyne hydrogenation, it’s important to consider how far the reaction goes. As mentioned, we can stop at the intermediate stage or go all the way to the final, saturated product. Each stage has its own unique structure and hence its own unique drawing.
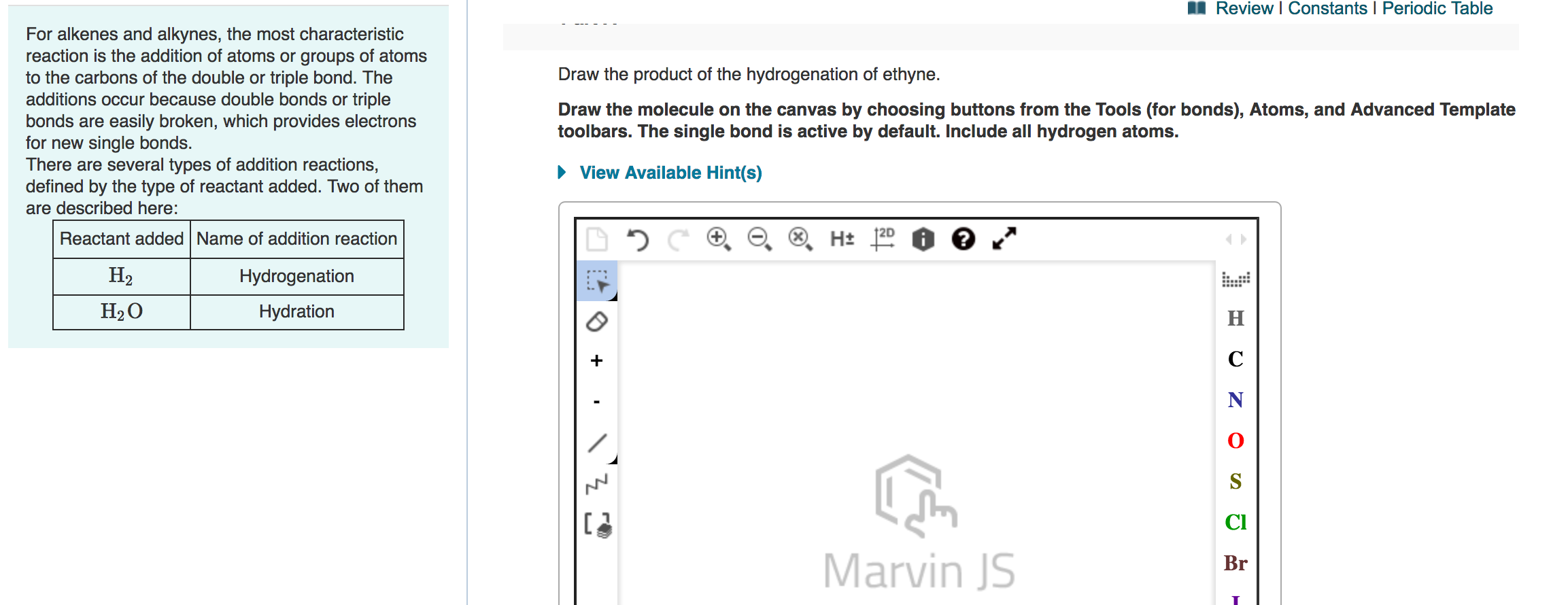
The initial molecule, ethyne, is represented by two carbon atoms each bonded to one hydrogen atom, with a prominent triple bond connecting the two carbons. Imagine them holding hands very, very tightly!
When we perform the first step of hydrogenation, adding one molecule of H₂, we break one of the pi bonds in the triple bond. The product here is ethene. Now, each carbon atom is bonded to two hydrogen atoms, and the carbons are linked by a double bond. It's like they’ve let go of one hand each and are now holding one hand.
If the hydrogenation continues, adding a second molecule of H₂, both remaining pi bonds are broken. This leads to the formation of ethane. In this final product, each carbon atom is bonded to three hydrogen atoms, and the two carbon atoms are connected by a simple, strong single bond. They’ve let go of all extra hands and are now just holding one each, with plenty of room for those hydrogen buddies.

Drawing these structures is a fundamental skill in chemistry. You'll typically see them represented using Lewis structures, where dots represent valence electrons and lines represent bonds (one line for a single bond, two for a double bond, and three for a triple bond). Alternatively, skeletal structures are often used, especially for larger molecules, where carbon atoms are implied at the vertices and ends of lines, and hydrogen atoms bonded to carbon are not explicitly drawn.
Benefits and Applications
The benefits of understanding and performing ethyne hydrogenation are numerous. For chemists, it's a cornerstone reaction that helps build more complex organic molecules. Many industrial processes rely on controlled hydrogenation. For example, ethene, the product of partial hydrogenation of ethyne, is a massive industrial chemical. It's the monomer used to create polyethylene, the most common plastic in the world! Think of all the plastic bottles, bags, and toys – they all start with something like this.
Furthermore, the complete hydrogenation of ethyne to ethane leads to a very stable alkane. Alkanes are the primary components of natural gas and are extensively used as fuels. So, in a way, this simple reaction is linked to how we power our homes and industries.
The ability to control the extent of hydrogenation is key. Catalysts, such as palladium, platinum, or nickel, are often used to speed up these reactions and guide them towards specific products. For instance, using a poisoned catalyst can help stop the hydrogenation at the ethene stage, preventing further conversion to ethane. This level of control is what makes chemical synthesis so powerful and allows us to create a vast array of materials with specific properties.
So, the next time you see a plastic item or even think about the natural gas that heats your home, remember the humble ethyne molecule and its transformative hydrogenation. Drawing the product isn't just an academic exercise; it's a visual passport into understanding the building blocks of our material world and the ingenious ways chemists manipulate them to create the products we use every day. It’s a journey from a simple triple bond to the complex materials that shape our lives!
